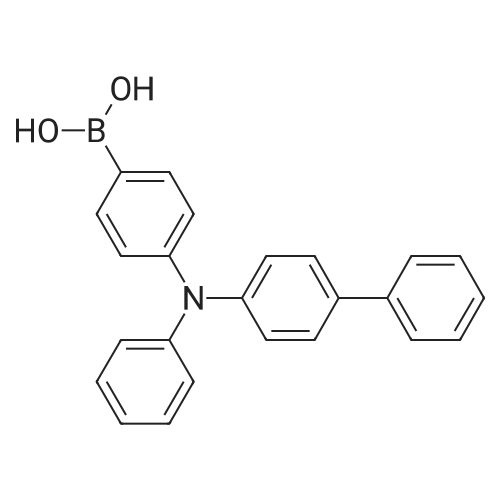
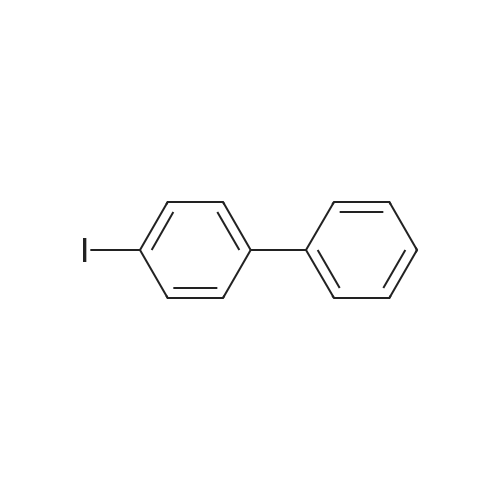
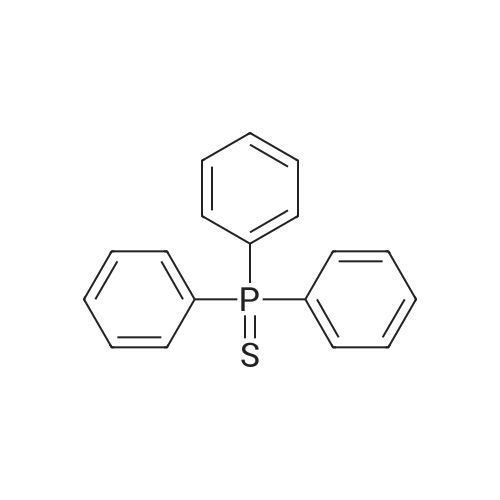
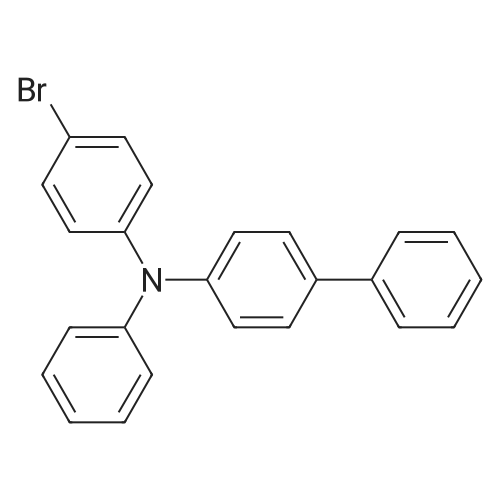
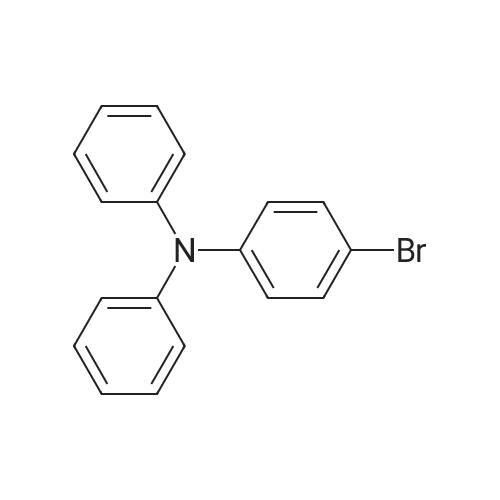
| 95.1% |
With sodium tert-pentoxide In toluene at 40 - 110℃; for 0.333333 - 2.33333h; |
1; 1a
Example 1; Batch Mode; To a 500 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.314 g, 2 mol %), CYTOP-216 (0.303 g, 2 mol %) and 45 mL of anhydrous toluene. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sequentially, 4-bromobiphenyl (32.8 g, 1.0 equiv), diphenylamine (26 g, 1.05 equiv), toluene (82 mL) and sodium t-pentoxide (29.1 g) are added with stirring. The reaction is heated to 90° C. over a 30 min period. A large exotherm is observed and the heating is shut off. After 1 hr 15 min the exotherm subsides and HPLC analysis confirms complete conversion of diphenylamine to N-biphenyl-diphenyl amine.Following completion of the reaction, toluene (50 mL) is added and the solution is filtered to remove insoluble materials. The solids are washed with toluene so as to have a final liqueur with a volume of 500 L. This solution is treated with Filtrol-24 (20 g) and Al2O3 (20 g) at 90° C. for 2 hours. The absorbents are filtered while the solution is hot. A second treatment with Al2O3 (4 g) at 90° C. is necessary to completely remove color (most likely residual palladium catalyst). The toluene solution is concentrated to 100 mL and isopropanol (150 mL) is added followed by methanol (250 mL) to complete precipitation of N-biphenyl-diphenylamine. The solid N-biphenyl-diphenylamine is filtered and washed with methanol (100 mL), air dried then finally vacuum dried (60° C./5 mmHg) overnight. The final yield of N-biphenyl-diphenylamine is 42.97 g (95.1%). HPLC, 1H NMR and elemental analysis confirm purity of N-biphenyl-diphenylamine at >99.5%. Ashing followed by ICP analysis does not detect any residual palladium present.When this reaction is run in the laboratory in batch mode it is heated to the point where it self-refluxes (from exothermic self-heating) and then upon cooling the reaction is complete. In determining isothermal conditions whereby N,N-diphenyl-4-biphenylamine was produced at a reasonable rate, it was found that the reaction proceeded well at 90° C. but did not proceed at all at temperatures lower than 90° C., thus indicating the reaction has an on-off point in addition to being exothermic. Three identical runs were performed at 90° C. to measure accurately the reaction conversion kinetics.It was found that the conversion profiles for the three runs are not exactly reproducible, as seen in FIG. 2. FIG. 2 shows a conversion plot of N,N-diphenyl-4-biphenylamine reaction isothermal at 90° C. (left) and temperature profiles of same reactions (right). The additional temperatures measured during the reaction indicated that, while the heating profiles for each reaction were identical, in one of the three cases, the reaction resulted in a large exotherm and an overheating of the reaction mixture (FIG. 2). The cause of the inconsistency is unknown. Complete conversion was seen for all three reactions based on the 10% excess of diphenylamine that is present in the reaction.; Example 1a; Continuous Mode with One Solvent; With satisfactory conversion results in batch mode, the identical reaction conditions (10 mol % excess diphenylamine, Pd(OAc)2/CYTOP-216 2 mol %, NaOtPen 1.2 mol equivalents, and toluene as solvent, isothermal at 90° C. were used with a continuous plug flow reactor (as shown in FIG. 1). The residence time was approximately 20 minutes and samples of approximately 10 mL were taken continuously.Plugging of the reactor was a constant problem that required pulsing of the pressure on the input pots. However, after approximately 30 samples, the reactor plugged entirely. The plugging is presumably due to the production of insoluble sodium bromide precipitate (a known by-product of the reaction). In order to counter the plugging, several re-pipings of the plug flow reactor were tried but did not resolve the problem. Subsequently, the use of a co-solvent along with toluene was tried. The conversion of this continuous reaction was on average around 20% to produce the N,N-diphenyl-4-biphenylamine even with the plugging of the reactor. |
| 95% |
With tri-tert-butyl phosphine; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In hexane; toluene at 80℃; for 5h; Inert atmosphere; |
1.2.i; C-2 (i)
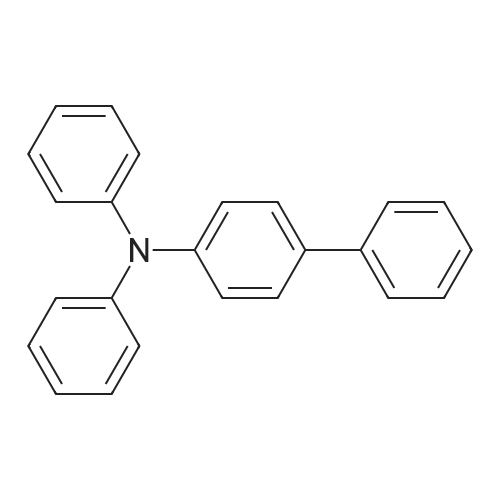
Synthesis of 4-phenyltriphenylamine
(i) Synthesis of 4-phenyltriphenylamine A synthesis scheme (C-2) of 4-phenyltriphenylamine is shown below. Initially, 14 g (59 mmol) of 4-bromobiphenyl, 10 g (59 mmol) of diphenylamine, 11 g (118 mmol) of sodium tert-butoxide, 0.30 g (0.52 mmol) of bis(dibenzylideneacetone)palladium(0) were put in a 100 mL three-neck flask, and the atmosphere of the flask was substituted by nitrogen. Then, 100 mL of toluene and 0.50 mL of tri(tert-butyl)phosphine (10 wt % hexane solution) were added to this mixture. This mixture was stirred under reduced pressure so as to be deaerated. After deaeration, this mixture was heated and stirred at 80° C. for 5 hours. After stirring, toluene was added to this mixture, and the suspension was washed with a saturated sodium hydrogen carbonate solution, and a saturated saline. Then, magnesium sulfate was added into the organic layer for drying. The mixture was subjected to suction filtration through Celite (produced by Wako Pure Chemical Industries, Ltd., Catalog No. 531-16855), alumina, and Florisil (produced by Wako Pure Chemical Industries, Ltd., Catalog No. 540-00135) and a filtrate was obtained. The obtained filtrate is concentrated to give a solid. The solid was recrystallized from a mixed solvent of chloroform and hexane to give 18 g of the target substance, powdery light brown solid of 4-phenyltriphenylamine in a yield of 95%. |
| 94% |
With tris-(dibenzylideneacetone)dipalladium(0); P-phenyl-2,2,6,6-tetramethyl-phosphorinan-4-ol; sodium t-butanolate In 1,4-dioxane at 100℃; for 12h; Inert atmosphere; |
|
| 91% |
With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; palladium 10% on activated carbon; sodium t-butanolate In 1,3,5-trimethyl-benzene for 24h; Inert atmosphere; Reflux; |
|
| 91% |
With potassium hydroxide In water for 24h; Reflux; Inert atmosphere; |
|
| 91% |
With bis(η3-allyl-μ-chloropalladium(II)); potassium hydroxide In water for 24h; Reflux; Inert atmosphere; |
|
| 89% |
With sodium t-butanolate In hexane; xylene at 130℃; for 3.5h; |
1
Step 1: Synthesis of 4-phenyltriphenylamine][0510]A synthetic scheme of 4-phenyltriphenylamine in Step 1 is shown in the following (L-I). [0511] In a 300-mL three-neck flask, 9.3 g (40 mmol) of 4-bromophenyl, 6.8 g (40 mmol) of diphenylamine, 5.O g (50 mol) of sodium te/t-butoxide, and 10 mg of bis(dibenzylideneacetone)palladium(0) were put, and the atmosphere in the flask was substituted by nitrogen. Then, 100 mL of xylene and 0.6 mL of tri(tert-butyl)phosphine (10 wt% hexane solution) were added to this mixture. [0513]This mixture was deaerated while being stirred under low pressure. After the atmosphere was substituted by nitrogen, the mixture was stirred at 130 0C for 3.5 hours. After the stirring, 250 mL of toluene was added to the reaction mixture, and this suspension was filtrated through Celite, alumina, and then Florisil. The obtained filtrate was washed with water and dried, and magnesium sulfate was added thereto. This mixture was filtrated through Celite, alumina, and then Florisil to obtain filtrate. The obtained filtrate was concentrated, and methanol was added thereto. The mixture was irradiated with supersonic and then recrystallized to obtain 11 g of an objective white powder at a yield of 89 %. |
| 89% |
With tri-tert-butyl phosphine; sodium t-butanolate In hexane; xylene at 130℃; for 3.5h; Inert atmosphere; |
1.1
In a 300-mL three-neck flask, 9.3g (40 mmol) of 4-bromobiphenyl, 6.8 g (40 mmol) of diphenylamine, 5.0g (50 mmol) of sodium tert-butoxide, and lOmg of bis(dibenzylideneacetone)palladium(0) were put, and 100 mL of xylene and 0.6 mL of tri(?ert-butyl)phosphine (a 10wt % hexane solution) were added to this mixture. This mixture was deaerated while being stirred under low pressure. After the deaeration, the mixture was stirred under a nitrogen atmosphere at 130 0C for 3.5 hours. After the stirring, 250 mL of toluene was added to this reaction mixture, and this suspension was filtrated through Celite (produced by Wako Pure Chemical Industries Ltd., Catalogue No. 531-16855, the same product was used hereinafter), alumina, and then Florisil (produced by Wako Pure Chemical Industries Ltd., Catalogue No. 540-00135, the same product was used hereinafter). The obtained filtrate was washed with water, and magnesium sulfate was added thereto to dry the filtrate. This mixture was filtrated through Celite, alumina, and then Florisil to obtain filtrate. The obtained filtrate was concentrated, methanol was added thereto, ultrasonic waves were applied thereto, and then recrystallization thereof was performed to obtain 11 g of an objective white powder at a yield of 89 %. The synthetic scheme of Step 1 is shown in (a-1) given below. |
| 86% |
With bis[2-(diphenylphosphino)phenyl] ether; sodium t-butanolate In toluene at 100℃; for 18h; |
|
| 84% |
With palladium diacetate; bis[2-(diphenylphosphino)phenyl] ether; sodium t-butanolate In toluene at 100℃; for 14h; |
|
| 75% |
With sodium t-butanolate In toluene at 70℃; for 72h; Inert atmosphere; |
|
| 73% |
With sodium t-butanolate at 104℃; for 2h; |
|
| 58 %Chromat. |
With potassium <i>tert</i>-butylate In toluene at 100℃; for 2h; |
|
| 29 %Chromat. |
With potassium <i>tert</i>-butylate In toluene at 100℃; for 2h; |
|
| 44 %Chromat. |
With potassium <i>tert</i>-butylate |
|
| 50 %Chromat. |
With potassium <i>tert</i>-butylate at 200℃; for 0.75h; |
|
| 62 %Chromat. |
With potassium <i>tert</i>-butylate for 1.5h; |
|
| 50 %Chromat. |
With potassium <i>tert</i>-butylate In toluene for 1h; |
|
|
In trihexyl(tetradecyl)phosphonium chloride; toluene at 90℃; for 2.75h; |
1
Example 1 Preparation of Arylamine Intermediate in an Ionic Liquid; To a 500 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.47 g, 0.01 mol %) and tri-t-butylphosphine stock solution (0.46 mL 2.7 mmol) and dissolved in 75 mL toluene. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sequentially, 75 mL of (tetradecyl)trihexyl phosphonium chloride, 4-bromobiphenyl (50 g, 224 mmol), diphenylamine (39.7 g, 235 mmol), and sodium t-butoxide (38.7 g, 403 mmol) are added with stirring. The reaction is heated to 90° C. over a 30 min period. A large exotherm is observed and the heating is shut off. After 1 hr 15 min the exotherm subsides and HPLC analysis confirms complete conversion of diphenylamine to N-biphenyl-diphenylamine. Following completion of the reaction, the reaction mixture is cooled to room temperature (about 20° C. to about 25° C.), and hexane and water are added to produce a triphasic medium. The top organic layer is removed, and is treated with Filtrol-24 and Al2O3 at 90° C. for 2 hours. The absorbents are filtered while the solution is hot. The hexane solution is concentrated and isopropanol is added followed by methanol to complete precipitation of N-biphenyl-diphenylamine. The solid N-biphenyl-diphenylamine is filtered and washed with methanol, air dried then finally vacuum dried (60° C./5 mmHg) overnight. Ashing followed by ICP analysis does not detect any residual palladium present. The middle ionic liquid phase is collected and dried. The reaction is repeated again with re-charging of the reactants, as described above. The process proceeds in the same manner, with the same results, indicating that the palladium catalyst is recycled. |
|
With sodium tert-pentoxide In CYPHOS IL-109; toluene at 90 - 110℃; for 0.633333 - 2.5h; |
2; 2a
Example 2; Batch Mode with Co-Solvent; To a 500 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.314 g, 2 mol %), tri-t-butylphosphine (0.310 g, 2 mol %) and 45 mL of anhydrous toluene. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sequentially, 4-bromobiphenyl (32.8 g, 1.0 equiv), diphenylamine (26 g, 1.05 equiv), CYPHOS IL-109 (63.5 mL), anhydrous toluene (18.5 mL) and sodium t-pentoxide (29.1 g) are added with stirring. The reaction is heated to 110° C. over a 30 minute period and the reaction was continued for 2 hours at 110° C. after which time. HPLC analysis showed a 67.9% conversion of diphenylamine to N-biphenyl-diphenylamine.; Example 2a; Continuous Mode with Co-Solvents; With satisfactory conversion results in batch mode, the identical reaction conditions (10 mol % excess diphenylamine, Pd(OAc)2/tri-t-butylphosphine 2 mol %, NaOtPen 1.2 mol equivalents, and a 50% mixture of toluene and CYPHOS IL-109 as the solvent, isothermal at 90° C. were used with a continuous plug flow reactor (as shown in FIG. 1). The desired outcome was high conversion of up to 89%, shown in FIG. 3, with no insoluble precipitates (sodium bromide solubilized) and no formation of side products.The results demonstrate that indeed Buchwald chemistry can be run to high conversion if used in continuous mode. It is believed that the variation in conversion was related to variation in the residence time due to uneven flow characteristics; use of a pump and better flow control should allow more consistent performance. The average residence time for this reaction was 38 minutes.Furthermore, if the residence time during the continuous process inside the reactor is considered, the processes in this example have better conversion at an equivalent time and at a lower temperature (Table 1). |
|
With sodium tert-pentoxide In 1,3-DIOXOLANE; toluene at 70 - 100℃; for 0.25 - 0.391667h; microreactor technology; |
1; 2; 3; 4
The commercial Cytos microreactor, manufactured by CPC Technologies, is configured with three residence time units (total volume 47 mL) and heated to 70° C. Toluene is pumped through the microreactor until the reagent streams are ready to be pumped.To a 1 L flask fitted with a mechanical stirrer, argon inlet and reflux condenser is added 9.5 g of tri-tert-butylphosphine into 1 L of anhydrous toluene to make Stock Solution A.To a 50 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.072 g, 1 mol %), Stock Solution A (6.93 mL, 1 mol % tri-t-butylphosphine), and 23 mL of 1,3-dioxolane. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sodium t-pentoxide (3.54 g, 1 mol equivalent) is added with stirring. This solution (Solution B) was not heated.To a second 50 mL fitted with mechanical stirrer, argon inlet and reflux condenser is charged 4-bromobiphenyl (7.5 g, 1.0 mol equivalent), diphenylamine (5.72 g, 1.05 mol equivalent) and 30 mL of 1,3-dioxolane. The solution is stirred for 10 minutes. This solution (Solution C) was not heated.Solution B and C are each pumped at 1.0 mL/min into the microreactor via high pressure liquid chromatography (HPLC) pumps. The microreactor is maintained at 70° C. The resulting residence time in the microreactor is 23.5 minutes. The outlet stream is collected in a vial, with samples taken over 20 minutes. Conversion averaged 92% over 18 minutes.The chemical reaction as was conducted in this example, and in the following examples, is represented as: Example 2To a 50 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.036 g, 1 mol %), Stock Solution A (3.47 mL, 1 mol % tri-t-butylphosphine) and 26.53 mL of anhydrous toluene. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sodium t-pentoxide (1.77 g, 1 mol equivalent) is added with stirring. This solution (Solution B) was not heated.To a second 50 mL fitted with mechanical stirrer, argon inlet and reflux condenser is charged 4-bromobiphenyl (3.75 g, 1.0 mol equivalent), diphenylamine (2.86 g, 1.05 mol equivalent) and 30 mL of 1,3-dioxolane. The solution is stirred for 10 minutes. This solution (Solution C) was not heated.Solution B and C are each pumped at 1.0 mL/min into the microreactor via HPLC pumps. The microreactor is maintained at 70° C. The resulting residence time in the microreactor is 23.5 minutes. The outlet stream is collected in a vial, with samples taken over 15 minutes. Conversion was about 51% over 20 minutes.Example 3The microreactor is configured with three residence time units (total volume 47 mL) and heated to 80° C. Toluene is pumped through the microreactor until the reagent solutions are ready to be pumped.To a 50 mL flask fitted with mechanical stirrer, argon inlet and reflux condenser is charged palladium(II)acetate (0.036 g, 1 mol %), Stock Solution A (3.46 mL, 1 mol % tri-t-butylphosphine) and 27 mL of anhydrous toluene. The solution is stirred for 1 hour to allow for dissolution of palladium acetate. Then sodium t-pentoxide (1.77 g, 1 mol equivalent) is added with stirring. The solution (Solution B) is heated to 40° C. over a 30 min period.To a second 50 mL fitted with mechanical stirrer, argon inlet and reflux condenser is charged 4-bromobiphenyl (3.75 g, 1.0 mol equivalent), diphenylamine (2.858 g, 1.05 mol equivalent) and 30 mL of 1,3-dioxolane. The solution is stirred for 10 minutes. This solution (Solution C) is heated to 40° C.Solution B and C are each pumped at 1.0 mL/min into the microreactor via HPLC pumps. The microreactor is maintained at 80° C. The resulting residence time in the microreactor is 23.5 minutes. The outlet stream is collected in a vial, with samples taken over 20 minutes. Conversion averaged 82% over the 20 minutes.Example 4The same procedure as followed in Example 1, except the two Solutions B and C are each pumped at 1.5 mL/min each. The resulting residence time was 15 minutes. Again, the outlet stream was collected with samples taken over 15 minutes. Conversion was lower, averaging 50% over 15 minutes. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping