Abstract
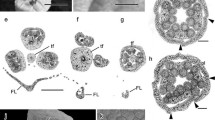
The genus Mimosa L. (Leguminosae; Caesalpinioideae; mimosoid clade), comprising more than 500 species, is an intriguing genus because, like other members of the mimosoid clade, it presents an enormous variation in floral characteristics and high merism lability. Thus, this study aimed to elucidate the floral development and identify which ontogenetic pathways give rise to merism variation and andromonoecy in Mimosa caesalpiniifolia, M. pudica, M. bimucronata, and M. candollei. Floral buds at various stages of development and flowers were collected, fixed, and processed for surface analysis (SEM). The development of the buds is synchronous in the inflorescences. Sepals appear simultaneously as individualized primordia in M. caesalpiniifolia and in reversed unidirectional order in M. bimucronata, with union and formation of an early ring-like calyx. Petal primordia appear in unidirectional order, with a noticeably elliptical shape in M. caesalpiniifolia. The wide merism variation in Mimosa results from the absence of organs from inception in the perianth and androecium whorls: in dimerous, trimerous, or tetramerous flowers, the additional organs primordia to compose the expected pentamerous flowers are not initiated. The haplostemonous androecium of M. pudica results from the absence of antepetalous stamens from inception. In the case of intraspecific variations (instabilities), there is no initiation and subsequent abortion of organs in the events of reduction in merosity. In addition, extra primordia are initiated in supernumerary cases. On the other hand, staminate flowers originate from the abortion of the carpel. Mimosa proved to be an excellent model for studying merism variation. The lability is associated with actinomorphic and rather congested flowers in the inflorescences. Our data, in association with others of previous studies, suggest that the high lability in merism appeared in clades that diverged later in the mimosoid clade. Thus, phylogenetic reconstruction studies are needed for more robust evolutionary inferences. The present investigation of ontogenetic processes was relevant to expand our understanding of floral evolution in the genus Mimosa and shed light on the unstable merism in the mimosoid clade.









Similar content being viewed by others
References
Almeida J, Rocheta M, Galego L (1997) Genetic control of flower shape in Antirrhinum majus. Instituto Superior De Agronomia Development 124:1387–1392
Arroyo MK (1981) Breeding systems and pollination biology in Leguminosae. In: Polhill RM, Raven PH (eds) Advances in legume systematics, part 2. Royal botanic gardens, Kew, pp 723–769
Barneby RC (1991) Sensitivae censitae: a description of the genus Mimosa Linnaeus (Mimosaceae) in the new world. Mem NY Bot Gard 65:1–835
Barros TC, Pedersoli GD, Paulino JV, Teixeira SP (2017) In the interface of caesalpinioids and mimosoids: Comparative floral development elucidates shared characters in Dimorphandra mollis and Pentaclethra macroloba (Leguminosae). Am J Bot 104:218–232
Beentje HJ (2010) The Kew plant glossary: an illustrated dictionary of plant terms. Royal Botanic Gardens, Kew
Bruneau AB, Klitgaard B, Prenner G, Fougere-Danezan M, Tucker SC (2014) Floral evolution in Detarieae (Leguminosae): phylogenetic evidence for labile floral development in an early diverging legume lineaege. Int J Plant Sci 175:392–417
Bull-Hereñu K, dos Santos P, Toni JFG, El Ottra JHL, Thaowetsuwan P, Jeiter J, Ronse De Craene LP, Iwamoto A (2022) Mechanical forces in floral development. Plants 11:661
Carles CC, Choffnes-Inada D, Reville K, Lertpiriyapong K (2004) ULTRAPETALA1 encodes a SAND domain putative transcriptional regulator that controls shoot and floral meristem activity in Arabidopsis. Development 132:897–911
Claßen-Bockhoff R, Bull-Hereñu K (2013) Towards an ontogenetic understanding of inflorescence diversity. Ann Bot 112:1523–1542
Corley SB, Carpenter R, Copsey L, Coen E (2005) Floral asymmetry involves an interplay between TCP and MYB transcription factors in Antirrhinum. Proc Natl Acad Sci USA 102:5068–5073
Daly DCDB, Harley MM, Martínez-Habibe MC, Weeks A (2011) Burseraceae. In: Kubitzki K (ed) The families and genera of vascular plants. Flowering plants. Eudicots: Sapindales, Cucurbitales Myrtaceae, vol 10. Springer-Verlag, Berlin, pp 76–104
Dellaporta S, Calderón-Urrea A (1993) Sex determination in flowering plants. Plant Cell 5:1241–1251
Dong J (2016) Morphological variation and floral development of major clades in Urticaceae -A focus on the female flowers. Dissertation, The University of Edinburgh.
Ellstrand NC, Mitchell RJ (1988) Spatial and temporal patterns of floral inconstancy in plants and populations of Ipomopsis aggregata (Polemoniaceae). Bot Gazette 149:209–212
Endress PK (2001) Evolution of floral symmetry. Plant Biol 4:86–91
Endress PK (2006) Angiosperm floral evolution: morphological developmental framework. Adv Bot Res 44:1–61
Endress PK (2008) The whole and the parts: relationships between floral architecture and floral organ shape, and their repercussions on the interpretation of fragmentary floral fossils. Ann Miss Bot Gard 95:101–120
Falcão MJ, Paulino JV, Kochanovski FJ, Figueiredo RC, Basso-Alves JP, Mansano VF (2020) Development of inflorescences and flowers in Fabaceae subfamily Dialioideae: an evolutionary overview and complete ontogenetic series for Apuleia and Martiodendron. Bot J Linn Soc 193:19–46
Fontquer P (1985) Diccionario de Botánica. Editorial Larbor, Barcelona
Galego L, Almeida J (2002) Role of DIVARICATA in the control of dorsoventral asymmetry in Antirrhinum flowers. Genes Dev 16:880–891
Gómez-Acevedo SL, Magállon S, Rio-Arce L (2007) Floral development in three species of Acacia (Leguminosae, Mimosoideae). Aust J Bot 55:30–41
Johansen DA (1940) Plant microtechnique. McGraw-Hill Book Co Inc, New York
Kochanovski FJ, Paulino JV, Teixeira SP, Tozzi AMGDA, Mansano VDF (2018) Floral development of Hymenaea verrucosa: an ontogenetic approach to the unusual flower of Fabaceae subfamily Detarioideae. Bot J Linn Soc 187:46–58
Koenen EJM, Kidner C, Souza ER, Simon MF, Iganci JR, Nicholls JA, Brown GK, Queiroz LP, Luckow M, Lewis GP, Pennington RT, Hughes CE (2020) Hybrid capture of 964 nuclear genes resolves evolutionary relationships in the mimosoid legumes and reveals the polytomous origins of a large pantropical radiation. Am J Bot 107:1710–1735
Kebert T, (2015) Floral diagram generator. http://ashipunov.info/shipunov/school/biol_154/kviti/napoveda_en.php Accessed 28 Feb 2023
Leite VG, Teixeira SP, Mansano VF, Prenner G (2015) Floral development of the early-branching papilionoid legume Amburana cearensis (Leguminosae) reveals rare and novel characters. Int J Plant Sci 176:94–106
Leite VG, Mansano VF, Teixeira SP (2018) Floral development of Moraceae species with emphasis on the perianth and adroecium. Sci Direct 240:116–132
Lewis G, Schrire B, Mackinder B, Lock M (2005) Legumes of the world. Royal Botanic Gardens, Kew, UK
LPWG [Legume Phylogeny Working Group] (2013) Legume phylogeny and classification in the 21st century: progress, prospects and lessons for other species-rich clades. Taxon 62:217–248
LPWG [Legume Phylogeny Working Group] (2017) A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny. Taxon 66:44–77
Luo D, Carpenter R, Vincent C, Copsey L, Coen E (1996) Origin of floral asymmetry in Antirrhinum. Nature 383:794–799
Mansano VF, Tucker SC, Tozzi AMGDA (2002) Floral Ontogeny of Lecointea, Zollernia, Exostyles, and Harleyodendron (Leguminosae: Papilionoideae: Swartzieae s.l.). Am J Bot 89:1553–1569
Matsunaga S, Kawano S (2001) Sex determination by sex cromosomes in dioeicious plants. Plant Biol 3:481–488
Meng A, Zhang Z, Li Z, Ronse de Craene LP, Wang H (2012) Floral development of Stephania (Menispermaceae): impact of organ reduction on symmetry. Inter J Plant Sci 173:861–874
Mitchell CH, Diggle PK (2005) The evolution of unisexual flowers: morphological and functional convergence results from diverse developmental transitions. Am J Bot 92:1068–1076
Moço MCDC, Pinheiro MCB (1999) Pollination ecology of Swartzia apetala Raddi var. apetala (Leguminosae-Papilionoideae). Brazil Archives Biol Technol 42:1–9
Naghiloo S, Dadpour MR, Movafeghi A (2012) Floral ontogeny in Astragalus compactus (Leguminosae: Papilionoideae: Galegeae): variable occurrence of bracteoles and variable patterns of sepal initiation. Planta 235:793–805
Nuraliev MS, Oskolski AA, Sokoloff DD, Remizowa MV (2010) Flowers of Araliaceae: structural diversity, developmental and evolutionary aspects. Plant Diver Evol 128:247–268
Nuraliev MS, Degtjareva GV, Sokoloff DD, Oskolski AA, Samigullin TH, Valiejo-Roman CM (2014) Flower morphology and relationships of Schefflera subintegra (Araliaceae, Apiales): an evolutionary step towards extreme floral polymery. Bot J Linn Soc 175:553–597
Paulino JV, Mansano VF, Teixeira SP (2013) Elucidating the unusual floral features of Swartzia dipetala (Fabaceae). Bot J Linn Soc 173:303–320
Paulino JV, Prenner G, Mansano VF (2014) Comparative development of rare cases of a polycarpellate gynoecium in an otherwise monocarpellate family Leguminosae. Am J Bot 101:572–586
Paulino JV, Mansano VF, Prenner G, Teixeira SP (2017) High developmental lability in the perianth of Inga (Fabales, Fabaceae): a Neotropical woody rosid with gamopetalous corolla. Bot J Linn Soc 183:146–161
Pedersoli GD, Teixeira SP (2016) Floral development of Parkia multijuga and Stryphnodendron adstringens, two andromonoecious mimosoid trees (Leguminosae). Inter J Plant Sci 177:60–75
Pedersoli GD, Paulino JV, Leite GV, Teixeira SP (2010) Elucidating enigmatic floral issues in Copaifera langsdorffii Desf. (Leguminosae, Caesalpinioideae). Inter J Plant Sci 171:834–846
Pedersoli GD, Leme FM, Leite VG, Teixeira SP (2019) Anatomy solves the puzzle of explosive pollen release in wind-pollinated urticalean rosids. Am J Bot 106:489–506
Pedersoli GD, Staedler YM, Schönenberger J, Teixeira SP (2022) A comparative approach reveals diversity of floral developmental processes in Urticaceae. Bot J Linn Soc 200:465–490
Pedersoli GD, Mansano VF, de Barros TC, Paulino JV, Teixeira SP (2023) Sympetaly in the mimosoid clade (Leguminosae, Caesalpinioideae): an unusual trait in the rosid group. Perspect Plant Ecol Evol Syst 60:125747
Prenner G (2004) Floral ontogeny in Calliandra angustifolia (Leguminosae: Mimosoideae: Ingeae) and its systematic implications. Inter J Plant Sci 165:417–426
Prenner G (2011) Floral ontogeny of Acacia celastrifolia: an enigmatic mimosoid legume with pronounced polyandry and multiple carpels. In: Wanntorp L, Ronse De Craene LP (eds) Flowers on the Tree of Life, vol 1. Cambridge University Press, pp 256–278
Prenner G, Bateman RM, Rudall PJ (2010) Floral formulae updated for routine inclusion in formal taxonomic descriptions. Taxon 59:241–250
Preston JC, Hileman LC (2009) Developmental genetics of floral symmetry evolution. Trends Plant Sci 14:147–154
Queiroz LP (2009) Leguminosas da Caatinga. Royal Botanic Gardens, Kew
Ramírez-Domenech JI (1989) Floral ontogeny of mimosoid legumes. Dissertation, Louisiana State University
Ramírez-Domenech JI, Tucker SC (1989) Phylogenetic implications of inflorescence and floral ontogeny of Mimosa strigillosa. Am J Bot 76:1583–1593
Ramírez-Domenech JI, Tucker SC (1990) Comparative ontogeny of perianth in mimosoid legumes. Am J Bot 77:624–635
Richards AJ (1997) Plant breeding system, 2nd edn. Garland Science, London
Ronse De Craene LP (2016) Meristic changes in flowering plants: how flowers play with numbers. Flora 221:22–37
Ronse De Craene L (2018) Understanding the role of floral development in the evolution of angiosperm flowers: clarifications from a historical and physico–dynamic perspective. J Plant Res 131:367–393
Ronse de Craene LP (2022) Floral diagrams: an aid to understanding flower morphology and evolution. Cambridge University Press, Cambridge, UK
Ronse De Craene L, Smets EF (1994) Merosity in flowers: definition, origin, and taxonomic significance. Plant Syst Evol 191:83–104
Simon MF, Grether R, de Queiroz LP, Särkinen TE, Dutra VF, Hughes CE (2011) The evolutionary history of Mimosa (Leguminosae): toward a phylogeny of the sensitive plants. Am J Bot 98:1201–1221
Sinjushin AA (2018) Floral ontogeny in Cordyla pinnata (A. rich.) Milne-Redh. (Leguminosae, Papilionoideae): Away from stability. Flora 241:8–15
Sinjushin AA (2021) Evolutionary history of the leguminous flower. Biol Bull Rev 11:400–413
Sinjushin AA (2023) Unequal stability of different parts in the flag blossom (Leguminosae) With notes on factors affecting variability of pentamerous pentacyclic angiosperm flowers. Plant Syst Evol 309:1
Sinjushin AA, Karasyova TA (2017) Stability of the floral structure in Leguminosae with flag versus non-flag blossom. Wulfenia 24:1–10
Sinjushin AA, Ploshinskaya M (2020) Flower development in Lythrum salicaria L., Cuphea ignea A. DC. And C. hyssopiafolia Kunth (Lythraceae): the making of monosymmetry in hexamerous flowers. Wulfenia 27:303–320
Specht CD, Barlett ME (2009) Flower evolution: the origin and subsequent diversification of the Angiosperm flower. Ann Rev Eco Evol Syst 40:217–43
Spencer V, Kim M (2018) Re“CYC”ling molecular regulators in the evolution and development of flower symmetry. Semin Cell Dev Biol 79:16–26
Teixeira SP, Ranga NT, Tucker SC (2009) Inflorescence and floral development of Dahlstedtia species (Leguminosae: Papilionoideae: Millettieae). Flora 204:769–781
Tucker SC (1984) Unidirectional organ initiation in leguminous flowers. Am J Bot 71:1139–1148
Tucker SC (1987a) Stamen proliferation in Swartzia macrosema, a legume considered transitional between subfamilies Caesalpinioideae and Papilionoideae. Am J Bot 74:627
Tucker SC (1987b) Floral initiation and development in legumes. In: Stirton CH (ed) Advances in legume systematics, part 3. Royal Botanic Gardens, Kew, pp 183–239
Tucker SC (1988) Heteromorphic flower development in Neptunia pubescens, a mimosoid legume. Am J Bot 75:205–224
Tucker SC (1989) Evolutionary implications of floral ontogeny in legumes. Adv Legume Biol Monogr Syst Bot Missouri Bot Gard 29:59–75
Tucker SC (1990) Loss of floral organs in Ateleia (Leguminosae: Papilionoideae: Sophoreae). Am J Bot 77:750–761
Tucker SC (1991) Helical floral organogenesis in Gleditsia, a primitive Caesalpinioid legume. Am J Bot 78(8):1130–1149
Tucker SC (1992a) The role of floral development in studies of legume evolution. Can J Bot 70:692–700
Tucker SC (1992b) The developmental basis for sexual expression in Ceratonia siliqua (Leguminosae: Caesalpinioideae: Cassieae). Am J Bot 79:318–327
Tucker SC (1997) Floral evolution, development, and convergence: the hierarchical-significance hypothesis. Inter J Plant Sci 158(6 suppl):143–161
Tucker SC (1999) Evolutionary lability of symmetry in early floral development. Inter J Plant Sci 160(6 suppl.):25–39
Tucker SC (2000) Floral development in Tribe Detarieae (Leguminosae: Caesalpinioideae): Amherstia Brownea, and Tamarindus. Am J Bot 87:1385–1407
Tucker SC (2003) Floral development in legumes. Plant Physiol 131:911–926
Tucker SC, Douglas AW (1994) Ontogenetic evidence and phylogenetic relationships among basal taxa of legumes. Adv Legume Syst 6:11–32
Wanntorp L, Puglisi C, Penneys D, Ronse De Craene LP (2011) Multiplications of floral organs in flowers: a case study in Conostegia (Melastomataceae, Myrtales). In: Wanntorp L, De Craene LR (eds) Flowers on the tree of life. Cambridge University Press, Cambridge, UK, pp 218–235
Wyatt R, Lipow SR (2021) Reproductive biology of Mimosa microphylla and Albizia julibrissin (Fabaceae: Caesalpinioideae) with a new explanation for the evolution of polyads, restricted stigmas, and polycarpelly in the mimosoid clade. J Torr Bot Soc 148: 97–108
Xu F, Ronse De Craene LP (2010) Floral ontogeny of Annonaceae: evidence for high variability in floral form. Ann Bot 106:591–605
Zimmerman E, Prenner G, Bruneau A (2013) Floral ontogeny in Dialiinae (Caesalpinioideae: Cassieae), a study in organ loss and instability. South Afr J Bot 89:188–209
Acknowledgements
The authors thank Rogério da Costa Figueiredo and Elaine Zózimo de Souza (Instituto de Pesquisas Jardim Botânico do Rio de Janeiro, Brazil), Raquel Pires (Centro Brasileiro de Pesquisas Físicas, Rio de Janeiro, Brazil), Brunno Renato Farias Verçoza Costa (Núcleo Multidisciplinar de Pesquisa/UFRJ, Rio de Janeiro, Brazil) for technical support during electron microscopy work; Lisi Dámaris Pereira Alvarenga for revising the English; Lucas Sá Barreto Jordão for identifying the species. This research was supported by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro – FAPERJ (process numbers: E-26/010.100998/2018; E-26/201.464/2022—BBP), and by CAPES with the scholarship for the first author.
Funding
Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro,E-26/010.100998/2018, Juliana Villela Paulino, Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (BR), E-26/201.464/2022—BBP, Juliana Villela Paulino.
Author information
Authors and Affiliations
Contributions
B.C.F.G., and R.S.M. performed the experiments and analyzed the data. J.V.P. and V.F.M. contributed to the study conception and design. All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gonçalves, B.C.F., Mansano, V.d., de Moraes, R.S. et al. Comparative floral development in Mimosa (Fabaceae: Caesalpinioideae) brings new insights into merism lability in the mimosoid clade. J Plant Res 137, 215–240 (2024). https://doi.org/10.1007/s10265-023-01507-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-023-01507-y