Tropical Homegardens - library.uniteddiversity.coop
Tropical Homegardens - library.uniteddiversity.coop
Tropical Homegardens - library.uniteddiversity.coop
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Tropical</strong> <strong>Homegardens</strong>
Advances in Agroforestry<br />
Volume 3<br />
Series Editor:<br />
P.K.R. Nair<br />
School of Forest Resources and Conservation,<br />
University of Florida, Gainesville, Florida, U.S.A.<br />
Aims and Scope<br />
Agroforestry, the purposeful growing of trees and crops in interacting combinations, began to attain<br />
prominence in the late 1970s, when the international scientific community embraced its potentials in the<br />
tropics and recognized it as a practice in search of science. During the 1990s, the relevance of agroforestry<br />
for solving problems related to deterioration of family farms, increased soil erosion, surface and ground<br />
water pollution, and decreased biodiversity was recognized in the industrialized nations too. Thus,<br />
agroforestry is now receiving increasing attention as a sustainable land-management option the world over<br />
because of its ecological, economic, and social attributes. Consequently, the knowledge-base of<br />
agroforestry is being expanded at a rapid rate as illustrated by the increasing number and quality of<br />
scientific publications of various forms on different aspects of agroforestry.<br />
Making full and efficient use of this upsurge in scientific agroforestry is both a challenge and an<br />
opportunity to the agroforestry scientific community. In order to help prepare themselves better for facing<br />
the challenge and seizing the opportunity, agoroforestry scientists need access to synthesized information<br />
on multi-dimensional aspects of scientific agroforesty.<br />
The aim of this new book-series, Advances in Agroforestry, is to offer state-of-the art synthesis of research<br />
results and evaluations relating to different aspects of agroforestry. Its scope is broad enough to encompass<br />
any and all aspects of agroforestry research and development. Contributions are welcome as well as<br />
solicited from competent authors on any aspect of agroforestry. Volumes in the series will consist of<br />
reference books, subject-specific monographs, peer-reviewed publications out of conferences,<br />
comprehensive evaluations of specific projects, and other book-length compilations of scientific and<br />
professional merit and relevance to the science and practice of agroforestry worldwide.<br />
The titles published in this series are listed at the end of this volume.
<strong>Tropical</strong> <strong>Homegardens</strong><br />
A Time-Tested Example of<br />
Sustainable Agroforestry<br />
Edited by<br />
B.M. Kumar<br />
Kerala Agricultural University, India<br />
and<br />
P.K.R. Nair<br />
University of Florida, Gainesville, FL, U.S.A.
A C.I.P. Catalogue record for this book is available from the Library of Congress.<br />
ISBN-10 1-4020-4947-1 (HB)<br />
ISBN-13 978-1-4020-4947-7 (HB)<br />
ISBN-10 1-4020-4948-X (e-book)<br />
ISBN-13 978-1-4020-4948-4 (e-book)<br />
Published by Springer,<br />
P.O. Box 17, 3300 AA Dordrecht, The Netherlands.<br />
www.springer.com<br />
Printed on acid-free paper<br />
All Rights Reserved<br />
© 2006 Springer<br />
No part of this work may be reproduced, stored in a retrieval system, or transmitted<br />
in any form or by any means, electronic, mechanical, photocopying, microfilming, recording<br />
or otherwise, without written permission from the Publisher, with the exception<br />
of any material supplied specifically for the purpose of being entered<br />
and executed on a computer system, for exclusive use by the purchaser of the work.<br />
Printed in the Netherlands.
List of Contributors<br />
Chapter Reviewers<br />
Preface<br />
Introduction<br />
P.K.R. Nair and B.M. Kumar<br />
CONTENTS<br />
Section 1: Historical and Regional Perspectives<br />
Diversity and change in homegarden cultivation in Indonesia<br />
K.F. Wiersum<br />
Urban and homegarden agroforestry in the Pacific islands: Current status<br />
and future prospects<br />
R.R. Thaman, C.R. Elevitch, and J. Kennedy<br />
Amazonian homegardens: Their ethnohistory and potential contribution<br />
to agroforestry development<br />
R.P. Miller, J.W. Penn, Jr., and J. van Leeuwen<br />
<strong>Homegardens</strong> of Mesoamerica: Biodiversity, food security, and nutrient<br />
management<br />
F. Montagnini<br />
Section 2: Structure, Function, and Dynamics of <strong>Homegardens</strong><br />
Homegarden dynamics in Kerala, India<br />
A. Peyre, A. Guidal, K.F. Wiersum, and F. Bongers<br />
Structure and dynamics of coconut-based agroforestry systems in Melanesia:<br />
A case study from the Vanuatu archipelago<br />
N. Lamanda, E. Malézieux, and P. Martin<br />
Diversity and dynamics in homegardens of southern Ethiopia<br />
Tesfaye Abebe, K.F. Wiersum, F. Bongers, and F. Sterck<br />
Homegarden plant diversity in relation to remoteness from urban centers:<br />
A case study from the Peruvian Amazon region<br />
A. Wezel and J. Ohl<br />
Gender and social dynamics in swidden and homegardens in Latin America<br />
P.L. Howard<br />
vii<br />
xi<br />
xiii<br />
1<br />
13<br />
25<br />
43<br />
61<br />
87<br />
105<br />
123<br />
143<br />
159
vi<br />
Section 3: Some New Thrust Areas<br />
Carbon sequestration potential of tropical homegardens<br />
B.M. Kumar<br />
Medicinal plants in tropical homegardens<br />
M.R. Rao and B.R. Rajeswara Rao<br />
Commercialization of homegardens in an Indonesian village: Vegetation<br />
composition and functional changes<br />
O.S. Abdoellah, H.Y. Hadikusumah, K. Takeuchi, S. Okubo, and Parikesit<br />
Transpiration characteristics of some homegarden tree species in Central<br />
Sri Lanka<br />
W.A.J.M. de Costa, K.S.P. Amaratunga, and R.S. Udumullage<br />
Ecology versus economics in tropical multistrata agroforests<br />
E. Torquebiau and E. Penot<br />
Financial analysis of homegardens: A case study from Kerala state, India<br />
S. Mohan, J.R.R. Alavalapati, and P.K.R. Nair<br />
Section 4: Future of <strong>Homegardens</strong><br />
The role of homegardens in agroforestry development: Lessons<br />
from Tomé-Açu, a Japanese-Brazilian settlement in the Amazon<br />
M. Yamada and H.M.L. Osaqui<br />
Urban homegardens and allotment gardens for sustainable livelihoods:<br />
Management strategies and institutional environments<br />
A.W. Drescher, R.J. Holmer, and D.L. Iaquinta<br />
Are tropical homegardens sustainable? Some evidence from Central Sulawesi,<br />
Indonesia<br />
K. Kehlenbeck and B.L. Maass<br />
Whither <strong>Homegardens</strong>?<br />
P.K.R. Nair<br />
Subject Index<br />
CONTENTS<br />
185<br />
205<br />
233<br />
251<br />
269<br />
283<br />
299<br />
317<br />
339<br />
355<br />
371
LIST OF CONTRIBUTORS<br />
Abdoellah O.S.<br />
Institute of Ecology and Department of Anthropology, Padjadjaran University,<br />
Bandung, Indonesia; E-mail or <br />
Alavalapati J.R.R.<br />
School of Forest Resources and Conservation, Institute of Food and Agricultural<br />
Sciences, University of Florida, Gainesville, FL 32611, USA; E-mail <br />
Amaratunga K.S.P.<br />
Department of Crop Science, Faculty of Agriculture, University of Peradeniya,<br />
Peredeniya 20400, Sri Lanka; E-mail <br />
Bongers F.<br />
Forest Ecology and Management group, Wageningen University, The Netherlands;<br />
E-mail <br />
De Costa W.A.J.M.<br />
Department of Crop Science, Faculty of Agriculture, University of Peradeniya,<br />
Peredeniya 20400, Sri Lanka; E-mail <br />
Drescher A.W.<br />
Albert-Ludwigs-Universität, Freiburg, Germany; E-mail <br />
Elevitch C.R.<br />
Agroforestry Net Inc., Holualoa, Hawai‘i 96725, USA; E-mail <br />
Guidal A.<br />
Forest and Nature Conservation Policy group, Wageningen University, The<br />
Netherlands (present address: GERES-CFSP #45 St.606, Toulkok, PO Box 2528,<br />
Phnom Penh-3, Cambodia);E-mail <br />
Hadikusumah H.Y.<br />
Institute of Ecology and Department of Biology, Padjadjaran University, Bandung,<br />
Indonesia; E-mail <br />
Holmer R.J.<br />
Xavier University College of Agriculture, Cagayan de Oro, The Philippines; E-mail<br />
<br />
Howard P.L.<br />
Department of Social Sciences, Wageningen University, Hollandseweg 1, 6706 KN<br />
Wageningen, the Netherlands; E-mail
viii<br />
LIST L OF CONTRIBUTORS<br />
Iaquinta D.L.,<br />
Nebraska Wesleyan University, Lincoln, Nebraska, USA; E-mail <br />
Kehlenbeck K.<br />
Institute for Crop and Animal Production in the Tropics, Georg-August-University,<br />
Grisebachstr. 6, D-37077 Göttingen, Germany; E-mail <br />
Kennedy J.<br />
Research School of Pacific and Asian Studies, Australian National University,<br />
Canberra, Australia; E-mail <br />
Kumar B.M.<br />
College of Forestry, Kerala Agricultural University, Thrissur 680656, Kerala, India;<br />
E-mail <br />
Lamanda N.<br />
CIRAD UMR SYSTEM, TA 80/ 01, Avenue Agropolis, 34 398 Montpellier Cedex<br />
5, France; E-mail or <br />
Maass B.L.<br />
Institute for Crop and Animal Production in the Tropics, Georg-August-University,<br />
Göttingen, Grisebachstr. 6, D-37077 Göttingen, Germany; E-mail <br />
Malézieux E.<br />
CIRAD UMR SYSTEM, TA 80/ 01, Avenue Agropolis, 34 398 Montpellier Cedex<br />
5, France; E-mail <br />
Martin P.<br />
INA P-G département AGER, bâtiment EGER BP 01 78850 Thiverval-Grignon,<br />
France; E-mail <br />
Miller R.P.<br />
Instituto Olhar Etnográfico, SHIN CA 5 Conj. J Bl. B, Sala 105, Brasília-DF 71505,<br />
Brazil; E-mail <br />
Mohan S.<br />
CREST-RESSACA, Texas A&M University, MSC 213, 700 University Blvd,<br />
Kingsville, TX 78363, USA; E-mail <br />
Montagnini F.<br />
Yale University, School of Forestry and Environmental Studies, 370 Prospect St.,<br />
New Haven, CT 06511, USA; E-mail
LIST L OF CONTRIBUTORS ix<br />
Nair P.K.R.<br />
School of Forest Resources and Conservation, Institute of Food and Agricultural<br />
Sciences, University of Florida, Gainesville, FL 32611, USA; E-mail <br />
Ohl J.<br />
School of Biological Sciences, University of East Anglia, Norwich NR4 7TJ, United<br />
Kingdom; E-mail <br />
Okubo S.<br />
Department of Ecosystem Studies, Graduate School of Agricultural and Life<br />
Sciences, University of Tokyo, Japan; E-mail <br />
Osaqui H.M.L.<br />
Division of International Environmental and Agricultural Science, Graduate School<br />
of Agriculture, Tokyo University of Agriculture and Technology, 3-5-8 Saiwaich,<br />
Fuch-shi, Tky 183-8509 Japan; E-mail <br />
Parikesit<br />
Institute of Ecology and Department of Biology, Padjadjaran University, Bandung,<br />
Indonesia; E-mail <br />
Penn J. W. Jr.<br />
Grand Valley State University, 1155 Au Sable Hall, Allendale, MI, 49401, USA;<br />
e-mail <br />
Penot E.<br />
CIRAD TERA, TA 60/ 15 – 34398 Montpellier CX5 – France; E-mail <br />
Peyre A.<br />
Forest and Nature Conservation Policy group, Wageningen University, The<br />
Netherlands (present address: 50 Avenue Henri GINOUX 92 120 Mont Rouge,<br />
France); E-mail or r <br />
Rajeswara Rao B.R.<br />
Central Institute of Medicinal and Aromatic Plants (CIMAP) Resource Centre,<br />
Boduppal, Uppal P.O. Hyderabad 500 039, India; E-mail .<br />
Rao M.R.<br />
Plot No. 11, ICRISAT Colony (Phase-I), Brig. Syed Road, Manovikasnagar (P.O.),<br />
Secunderabad–500 009, India; E-mail <br />
Sterck F.<br />
Forest Ecology and Management group, Wageningen University, The Netherlands;<br />
x<br />
LIST L OF CONTRIBUTORS<br />
Takeuchi K.<br />
Department of Ecosystem Studies, Graduate School of Agricultural and Life<br />
Sciences, University of Tokyo, Japan<br />
Tesfaye Abebe<br />
Debub University, Awassa College of Agriculture, Ethiopia; E-mail <br />
Thaman R.R.<br />
The University of the South Pacific, Suva, Fiji i Islands; 1487; E-mail <br />
Torquebiau E.,<br />
CIRAD TERA, TA 60/15 – 34398 Montpellier CX5, France; E-mail or <br />
Udumullage R.S.<br />
Department of Crop Science, Faculty of Agriculture, University of Peradeniya,<br />
Peredeniya 20400, Sri Lanka<br />
van Leeuwen J.<br />
Instituto Nacional de Pesquisas da Amazônia – INPA, Manaus, Amazonas, Brazil;<br />
E-mail <br />
Wezel A.<br />
Institute of Landscape and Plant Ecology (320), University of Hohenheim, 70593<br />
Stuttgart, Germany; E-mail <br />
Wiersum K.F.<br />
Forest and Nature Conservation Policy group, Wageningen University, The<br />
Netherlands; E-mail <br />
Yamada M.<br />
Division of International Environmental and Agricultural Science, Graduate School<br />
of Agriculture, Tokyo University of Agriculture and Technology, 3-5-8 Saiwaich,<br />
Fuch-shi, Tky 183-8509 Japan; E-mail
CHAPTER REVIEWERS<br />
Abdoellah, O.S, Padjadjaran University, Bandung, Indonesia<br />
Allen, S.C., University of Florida, USA<br />
Becker, Brian, University of Florida, USA<br />
Bellow, John G., COAPS-Florida State University, USA<br />
Bourdeix, Roland, CIRAD, France<br />
Campilan, Dindo, CIP-Users’ Perspectives with Agricultural Research and Development,<br />
The Philippines<br />
Clement, C.R., National Research Institute for the Amazon (INPA), Brazil<br />
De Costa, W.A.J.M., University of Peradeniya, Sri Lanka<br />
De Zoysa, Mangala, University of Ruhuna, Kamburupitiya, Sri Lanka<br />
Depommier, Denis, CIRAD-Forêt, Montpellier, France<br />
Doolittle, A.A., Yale University, USA<br />
Fleischman, Forrest, Forest Service Employees for Environmental Ethics, Eugene,<br />
USA<br />
Geethakutty, P.S., Kerala Agricultural University, Thrissur, India<br />
Jose, S., University of Florida, USA<br />
Kallarackal, J., Kerala Forest Research Institute, India<br />
Maass, B.L., Georg-August-University, Goettingen, Germany<br />
Miller, R.P., Instituto Olhar Etnográfico, Brazil<br />
Mohan, S., Texas A&M University, USA<br />
Montagnini, Florencia, Yale University, USA<br />
Muraleedharan, P.K., Kerala Forest Research Institute, India<br />
Nair, V.D., University of Florida, USA<br />
Palada, Manuel C., Asian Vegetable Research and Development Centre, Taiwan<br />
Penot, Eric, CIRAD TERA, Montpellier, France<br />
Puri, S., Indira Gandhi Agricultural University, Raipur, India<br />
Rao, J.M., University of Florida, USA<br />
Rao, M.R., Secunderabad, India<br />
Russell, A.E., Iowa State University, USA<br />
Schroth, G., Conservation International, Washington, USA<br />
Torquebiau, E., CIRAD TERA, Montpellier, France<br />
Wiersum, K.F., Wageningen University, The Netherlands<br />
Yamada, Masaaki, Tokyo University of Agriculture and Technology, Japan
PREFACE<br />
<strong>Tropical</strong> homegardens are a topic of discussion in most agroforestry conferences<br />
especially those covering humid tropical lowlands, but publications on this topic are<br />
scattered in the literature; comprehensive books and reports focused on it are rare.<br />
The motivation for this book was the desire to address that deficiency, following a<br />
session on <strong>Tropical</strong> <strong>Homegardens</strong> at the 1st World Congress of Agroforestry,<br />
Orlando, Florida, USA in June – July 2004 (http://conference.ifas.ufl.edu/wca). The<br />
initial idea was to bring out a publication based on the presentations at the Congress<br />
session; but consequent to enthusiastic responses from the professional community,<br />
the scope of the book was broadened to make it more comprehensive than a<br />
conference publication.<br />
As it turned out, only five chapters out of the total 20 in the book are based on<br />
presentations at the above Congress session. Three chapters are adaptations from<br />
papers that have recently been published (or have been accepted for publication) in<br />
Agroforestry Systems journal on issues that are important from the point of<br />
comprehensiveness of the book. Seven of these eight chapters are research articles and<br />
are presented in the conventional research-publication format (Introduction, Materials<br />
and Methods, Results, and Discussion); they present a glimpse of the nature of current<br />
research in homegardens. All other chapters are review and synthesis of current state<br />
of knowledge on homegarden issues from all three developing continents (Africa,<br />
Asia, and Latin America & the Caribbean). The chapters are organized into five<br />
sections (Historical and Regional Perspectives; Structure, Function, and Dynamics;<br />
Some New Thrust Areas; and Future of <strong>Homegardens</strong>); each section contains a mix of<br />
research and review articles. We believe that these 20 chapters represent the state-ofthe-art<br />
of tropical homegardens today.<br />
The expeditious publication of the book would not have been possible without the<br />
<strong>coop</strong>eration and dedication of the authors and reviewers. All chapters were<br />
rigorously peer-reviewed. We thank the reviewers (see the list attached) for their<br />
insightful comments and critical suggestions, which helped to enhance the quality of<br />
the chapters. The authors too have been a very pleasant and professional group to<br />
work with; we greatly appreciate their <strong>coop</strong>eration and understanding in putting up<br />
with our requests for repeated revisions within very short and strict time schedules.<br />
Once again, we sincerely thank all the authors and reviewers for their splendid<br />
<strong>coop</strong>eration. Special thanks go to Dr. Michael Bannister, who did an excellent job of<br />
reading through the manuscripts and scrutinizing the literature citations.<br />
B. Mohan Kumar, Thrissur, Kerala, India<br />
February 2006 P. K. R. Nair, Gainesville, Florida, USA
CHAPTER 1<br />
INTRODUCTION<br />
P.K.R. NAIR 1 AND B.M. KUMAR 2<br />
1 School of Forest Resources and Conservation, University of Florida, Gainesville,<br />
FL 32611, USA; E-mail: . 2 College of Forestry, Kerala<br />
Agricultural University, Thrissur 680656, Kerala, India;<br />
E-mail: <br />
1. THE CONCEPT OF HOMEGARDEN<br />
It is rather customary that any writing on homegardens starts with a “definition” of<br />
the term. The first drafts of several chapters in this book were no exception. This<br />
indicates that there is no universally accepted “definition” of the term and therefore<br />
the authors feel compelled to make their perception clear. An examination of the<br />
various “definitions” used or suggested by various authors (of chapters of this book<br />
as well as other recent homegarden literature) shows that they all revolve around the<br />
basic concept that has been around for at least the past 20 years, i.e., since the “early<br />
literature” on the subject (Wiersum, 1982; Brownrigg, 1985; Fernandes and Nair,<br />
1986; Soemarwoto, 1987): homegardens represent intimate, multistory combinations<br />
of various trees and crops, sometimes in association with domestic animals, around<br />
the homestead. This concept has been developed around the rural settings and<br />
subsistence economy under which most homegardens exist(ed). But, as some<br />
chapters in this book describe, the practice of homegardening is now being extended<br />
to urban settings (Drescher et al., 2006; Thaman et al., 2006) as well as with a<br />
commercial orientation (Abdoellah et al., 2006; Yamada and Osaqui, 2006).<br />
Even before the advent of such new trends as urban and commercial homegardens,<br />
the lack of clear-cut distinctions between various stages in the continuum<br />
from shifting cultivation to high-intensity multistrata systems and the various terms<br />
used in different parts of the world to denote the different systems has often<br />
created confusion in the use of the term homegarden and its underlying concept.<br />
The confusion is compounded by the fact that in many parts of the world, especially<br />
1<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 1-10.<br />
© 2006 Springer. Printed in the Netherlands.
2 P.K.R. NNAIR AND B.M. KUMAR<br />
in the New World, swidden farming such as the milpa of Mesoamerica evolve over a<br />
period of time into full-fledged homegardens consisting of mature fruit trees and<br />
various other types of woody perennials and the typical multistrata canopy<br />
configurations. In such situations, it is unclear where the swidden ends and<br />
homegarden begins – and often they co-exist. Yet another cause of confusion is the<br />
term itself: homegarden. Even for most agricultural professionals who are either not<br />
familiar with or are not appreciative of agroforestry practices, what we write as one<br />
word ‘homegarden’ sounds as two words ‘home’ and ‘garden’ sending the signal<br />
that the reference is to ornamental gardening around homes. While ornamentals are<br />
very much a part of homegardens in many societies, homegardens, in our concept,<br />
are not just home gardens of strictly ornamental nature.<br />
As we explained in our recent paper (Kumar and Nair, 2004), we use the term<br />
homegardens (and homegardening) to refer to farming systems variously described<br />
in English language as agroforestry homegardens, household or homestead farms,<br />
compound farms, backyard gardens, village forest gardens, dooryard gardens and<br />
house gardens. Some local names such as Talun-Kebun and Pekarangan that are<br />
used for various types of homegarden systems of Java (Indonesia), Shamba and<br />
Chagga in East Africa, and Huertos Familiares of Central America, have also<br />
attained international popularity because of the excellent examples of the systems<br />
they represent (Nair, 1993). In spite of the emergence of homegardening as a<br />
practice outside their “traditional” habitat into urban and commercial settings, the<br />
underlying concept of homegardens remains the same as before “intimate,<br />
multistory combinations of various trees and crops, sometimes in association with<br />
domestic animals, around homesteads.” Intimate plant associations of trees and<br />
crops and consequent multistory canopy configuration are essential to this concept.<br />
Equally important in this concept is the home around which most homegardens are<br />
maintained; but in some situations, multistory tree gardens (such as the Talun or<br />
Kebun of Indonesia: Wiersum, 1982) that are not in physical proximity to homes but<br />
receive the same level of constant attention from the owners’ household and have<br />
similar structural and functional attributes as other homegarden units located near<br />
homes are also considered as homegardens.<br />
2. GENESIS AND GLOBAL DISTRIBUTION OF HOMEGARDENS<br />
Tracing the history of homegardening, Kumar and Nair (2004) describe it as the oldest<br />
land use activity next only to shifting cultivation that has evolved through generations of<br />
gradual intensification of cropping in response to increasing human pressure and the<br />
corresponding shortage of arable lands. The Javanese homegardens of Indonesia and the<br />
Kerala homegardens of India – the two oft-cited examples – have reportedly evolved<br />
over centuries of cultural and biological transformations and they represent the<br />
accrued wisdom and insights of farmers who have interacted with environment,<br />
without access to exogenous inputs, capital, or scientific skills. Wiersum (2006)<br />
mentions that the origin of homegardening in Southeast Asia has been associated<br />
with fishing communities living in the moist tropical regions ca 13 000 to 9000 B.C.<br />
Implying the predominance of homegardens in ancient India, Vatsyayana in his<br />
great book of Hindu aesthetics – Kamasutra, written ca 300 to 400 AD, describe
INTRODUCTION<br />
I<br />
house gardens as a source of green vegetables, fig trees (Ficus spp.), mustard<br />
(Brassica spp.) and many other vegetables (c.f. Randhawa, 1980). Ibn Battuta in his<br />
travelogue (1325 – 1354) also wrote that the densely populated and intensively<br />
cultivated landscape with coconut (Cocos nucifera), black pepper (Piper nigrum),<br />
ginger (Zingiber officinale), sugarcane (Saccharum officinarum), pulses (grain<br />
legumes) and the like surrounding the houses formed a distinctive feature of the<br />
Malabar coast of Kerala (Randhawa, 1980). In both Java and Kerala, homegardening<br />
has been a way of life for centuries and is still critical to the local subsistence<br />
economy and food security (Kumar and Nair, 2004). This is true of several other Old<br />
World homegardens as well (e.g., the Chagga of Mt. Kilimanjaro in East Africa:<br />
Fernandes et al., 1984; Soini, 2005).<br />
In spite, or perhaps because, of the pre-historic origin of the practice, accurate<br />
data on the extent of area under homegardens are not available. Estimating the area<br />
of homegardens is beset with several problems (Kumar, 2006). A major one is<br />
the lack of distinct boundaries or demarcation between homegardens and other<br />
cultivated agricultural fields. As Tesfaye Abebe et al. (2006) point out; most<br />
homegardens studies are focused on gardens that constitute a component of a<br />
farming system consisting of cultivated fields away from homes complemented by<br />
the homegardens surrounding residential houses. In those situations, it is difficult to<br />
determine where homegardens end and other cultivated fields begin. Added to this<br />
problem is the “commodity-centric” approach to recording land use statistics:<br />
statistics are prepared and presented for specific (single) crops and commodities. In<br />
most cases, the area is listed under the most conspicuous or visible crop (e.g., fruit<br />
trees, coconut palms, and other trees that occupy the upper stratum of multistoried<br />
homegarden system) and the lower-story crops are seldom reported – and, often the<br />
reporting forms do not allow entries to be made of such mixed stands. Thus,<br />
homegardens are a “non-entity” for agricultural statistics and land revenue records.<br />
In spite of these difficulties, some efforts have been made in compiling statistics<br />
on the spread of homegardens. Such estimates include 5.13 million ha of land under<br />
pekarangans in Indonesia, 0.54 million ha under homesteads in Bangladesh, 1.05<br />
million ha in Sri Lanka, and 1.44 million ha in Kerala, India (Kumar, 2006).<br />
Christanty (1990) reported that more than 70% of all households in the Philippines<br />
maintained homegardens; but the extent of area occupied by them was not reported.<br />
Area statistics of homegardens are also not available from a number of other parts of<br />
the world although the prevalence of the practice – indeed predominance in many<br />
situations – has been reported from various parts of the tropics as several chapters in<br />
this volume also attest to. In an attempt to present a global distribution of<br />
homegardens, we selected 135 entries from the CABI Abstracts for the period from<br />
1990 to 2003 for which geographical locations are either mentioned or can be<br />
deduced; these included: Africa 21, Europe (Catalonia, Austria, etc.) 10, Central and<br />
South America 23, South Asia 45, Southeast Asia 30, other parts of Asia 2, Pacific<br />
islands 4. Based on these reports, supplemented with available statistics from other<br />
sources (e.g., reports on agricultural censuses) as well as personal experiences and<br />
observations of the authors, we have attempted a “Homegarden Map of the World”<br />
as presented in Fig. 1. The presentation only means that homegardens are present in<br />
3
4<br />
P.K.R. NNAIR AND B.M. KUMAR<br />
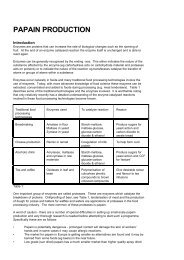
Explanation of Figure 1.<br />
The global distribution of homegardens. This attempt is based on the geographical<br />
distribution of 135 selected studies (the specific geographical locations of which are reported<br />
or can be deduced) from the CABI abstracts for the period from 1990 to 2003, including<br />
Africa (21 studies), Europe: Catalonia, Austria, and others (10), Central and South America<br />
(23), South Asia (45), Southeast Asia (30), other parts of Asia (2), and Pacific Islands (4),<br />
supplemented with available statistics from other sources (e.g., reports on agricultural<br />
censuses) and authors’ experiences/observations. Differing shade intensities in the figure<br />
represent high, moderate, and low frequency of occurrence of homegardens. We have used<br />
‘High’ for areas where the frequency of occurrence in the CABI abstracts is more than 20<br />
and/or if other databases (Statistical Yearbook 2000, Bangladesh Bureau of Statistics;<br />
Statistical Yearbook of Indonesia 2000, Badan Pusat Statistik; Census of Agriculture – Sri<br />
Lanka 2002. Agricultural holdings, extent under major crops and livestock statistics by<br />
district and DS/AGA division—based on operator’s residence: small holding sector,<br />
Colombo; Land Resources of Kerala State 1995, Kerala State Land Use Board; see Kumar,<br />
2006 for full citations) report that more than 50% of all households maintain homegardens,<br />
‘Medium’ for 10 to 20 mentions in CABI abstracts or 25 to 50% of the households maintain<br />
homegardens according to the other reports listed above, and ‘Low’ for all those cases where<br />
presence of homegardens has been reported in one or more ways but at levels below the above<br />
limits. “Apparently present” is the term used to denote regions where homegardens are said to<br />
be abundant based on the authors’ personal observations and/or communications from other<br />
sources, but on which published (accessible) information, especially on their area statistics, is<br />
limited or absent; such regions include tropical and subtropical parts of China, and some such<br />
other regions in Asia and Africa. The presentation only means that homegardens are present<br />
in the regions as indicated; it does not imply that homegardens are the only or the major land<br />
use system in any of these regions.
Figure 1. The global distribution of homegardens (see description on the left hand side, p. 4).<br />
INTRODUCTION<br />
II<br />
5
6<br />
P.K.R. NNAIR AND B.M. KUMAR<br />
the regions as indicated; it does not imply that homegardens are the only or major<br />
land use system in any of these regions.<br />
Based on the above, it is reasonable to assume that homegardens are most<br />
popular in the tropics, but can also be found between 40 o N and 30 o S latitudes.<br />
South- and Southeast Asia, the Pacific islands, East- and West Africa, and<br />
Mesoamerica are the regions where largest concentrations of homegardens can be<br />
found. <strong>Homegardens</strong> are also reportedly very popular in tropical and subtropical<br />
parts of China; however, other than general descriptions of the systems (e.g.,<br />
Zhaohua et al., 1991; Wenhua, 2001), practically no information could be gathered<br />
on their area statistics. The Mediterranean region of Catalonia (Agelet et al., 2000)<br />
and southern Africa (High and Shackleton, 2000) also are reported to have<br />
homegardens. In terms of ecological distribution, the highest concentrations of<br />
homegardens are in the humid and subhumid tropics, but they are also common in<br />
other ecological regions, especially the tropical highlands of Asia, Africa, and<br />
Mesoamerica (Nair, 1989). Clearly, our understanding about the spread of homegardens<br />
is incomplete; more efforts are needed to compile these statistics at local,<br />
regional, national, and global levels.<br />
Although homegardens are known as a predominantly tropical ‘phenomenon’,<br />
homegardening – or, conceptually similar practices – exist outside the tropical zone<br />
as well. For instance, Gold and Hannover (1987) and Herzog (1998) describe fruittree<br />
based agroforestry systems in North America and Europe, respectively. Vogl<br />
and Vogl-Lukasser (2003) reported that homegardens were typical elements of the<br />
mosaic of agroecosystems in the mountainous Alpine region of Austria. Streuobst<br />
(fruit trees grown on agricultural lands with crops or pasture as understorey), a<br />
traditional practice in Europe that has been on the decline since around 1930s, is<br />
now receiving increasing attention and acceptance among the general public and<br />
promoted by nongovernmental and conservation agencies. Although the fruit-tree<br />
based agroforestry systems are strictly not homegardening, such systems occasionally<br />
involve homegardening, and their socio-cultural, ecological, and aesthetic values often<br />
exceed their economic values. Based on an extensive survey and interview with<br />
practitioners of African-American gardening traditions in the rural southern United<br />
States, Westmacott (1992) traced the principal functions and features of African-<br />
American yards and gardens. During slavery, the gardens were used primarily to<br />
grow life-sustaining crops and vegetables, and the yard of a crowded cabin was<br />
often the only place where the slave family could assert some measure of<br />
independence and perhaps find some degree of spiritual refreshment. Since slavery,<br />
working the garden for the survival of the family has become less urgent, but there<br />
seems to be a revival of appreciation of their recreational, social, and other uses.<br />
For example, the gardeners are now finding pleasure in growing flowers and<br />
produce and deriving satisfaction from agrarian life-style, self-reliance, and private<br />
ownership. Through historical research, field observations, and oral interviews,<br />
Westmacott (1992) traces the West African roots of this gardening tradition and<br />
elucidates how the African-American community manipulated the garden space to<br />
their best advantage – something very similar to the motivations of subsistence<br />
gardeners in well-established homegardens in other parts of the world (Fig. 1).
INTRODUCTION<br />
I<br />
Related to the above-mentioned “African-American Yards and Gardens” of the<br />
southern United States is the increasing interest in hobby farming and weekend<br />
gardening that is getting popular in many urban and rapidly urbanizing societies in<br />
both industrialized and developing nations. Drescher et al. (2006) describe the urban<br />
homegardens and some of the operational and institutional issues related to them<br />
from a number of locations around the world. In a survey of agroforestry practices<br />
and opportunities in southeastern United States, Workman et al. (2003) identified<br />
several “special applications” of agroforestry such as use of fruit trees combined<br />
with gardens, ponds, and as bee forage and so-called patio gardens as an increasingly<br />
popular activity especially among immigrant Latin American communities. Thus,<br />
although homegardening as a major land use practice is most widespread in thickly<br />
populated tropical regions, the concept is being adopted in other geographical<br />
regions as well to a limited extent.<br />
3. COMPLEXITY OF HOMEGARDENS<br />
Species diversity is one factor that is common to all homegardens, and this point has<br />
been well brought out in homegarden literature time and again. Indeed, authors tend<br />
to get nostalgic about describing how diverse the plant communities in homegardens<br />
are and rather adamant about including elaborate species lists in their papers on<br />
homegardens to the extent that many seem to consider that a paper on any aspect of<br />
homegarden is incomplete without a species list! Interestingly, most of the plants<br />
that are listed in most such publications are the same irrespective of the geographical<br />
regions from where they are reported (see Nair, 2006). As various analyses and<br />
summary reports have repeatedly indicated (e.g., Kumar and Nair, 2004), food<br />
plants (food crops and fruit trees) are the most common species in most homegardens<br />
throughout the world. This underscores the fact that food- and nutritional<br />
security is the primary role of homegardens – again, a point well recognized in<br />
homegarden literature right from the “early” years (e.g., Brownrigg, 1985; Fernandes<br />
and Nair, 1986). Next in importance to food crops are cash crops, and with<br />
increasing trend toward commercialization, the interest in such crops is likely to<br />
only increase.<br />
We recognize that complexity by itself may not be a desirable attribute in land<br />
use systems that are (also) expected to fulfill production objectives. Being located<br />
on the “prime land” around homesteads and receiving utmost managerial attention<br />
of the homeowners all the time, farmers have high expectations of productivity from<br />
homegardens. After all, farmers decide on the species to be planted and retained in<br />
the homegardens based on the utilitarian value of the species. Species complexity in<br />
homegardens is therefore not a natural phenomenon, but a result of deliberate<br />
attempts and meticulous selection and management by farmers to provide the<br />
products they consider are important for their subsistence and livelihood. Species<br />
complexity in homegardens is thus a manmade feature, unlike in natural systems.<br />
This distinction is seldom recognized in comparisons involving ecological indices of<br />
species diversity of homegardens, several of which have lately been reported (see<br />
Nair, 2006).<br />
7
8<br />
P.K.R. NNAIR AND B.M. KUMAR<br />
Furthermore, it is likely that the extreme structural complexity and diversity may<br />
be a “bane” of the homegardens in a sense. Each homegarden is a unique land use<br />
entity in terms of component arrangement, organization, and management, and it<br />
reflects the personal preferences of its owner. This frustrates the development<br />
community that seeks out “replicable models”; this is presumably the main reason<br />
why homegardens have not received adequate attention in the development paradigms<br />
around the world.<br />
4. HOMEGARDENS IN THE CONTEXT OF CONTEMPORARY LAND USE<br />
ISSUES<br />
Today land use systems are challenged as never before with mounting concerns of<br />
environment and ethics on the one hand and pressures of economic development on<br />
the other. Production and economic issues that reigned supreme as ultimate goals in<br />
agricultural and forestry development activities during the past few decades are<br />
slowly yielding to environmental, societal, and social issues. Sustainability –<br />
meeting today’s needs without compromising the ability of future generations to<br />
satisfy their needs – is a key issue in all land use activities today. Central to this<br />
concept is the urge to achieve a balance between ecological preservation, economic<br />
vitality, and social justice. Land use systems today are thus evaluated based not only<br />
on their ability to fulfill any single objective such as production of a preferred<br />
commodity, but also on how best they fulfill the sustainability criteria. Contemporary<br />
issues that dominate the discussions in this context include natural-resource use in<br />
perpetuity, biodiversity conservation, gender equity, social justice, environmental<br />
integrity, appreciation of indigenous knowledge, preservation of cultural heritage,<br />
and so on.<br />
While systematic studies on the role of homegardens in many of these<br />
contemporary issues have not been done, there is a long-held belief and intuition that<br />
homegardens score very high on most – perhaps all – of these so-called “intangible”<br />
benefits. Logic, circumstantial evidences, and limited empirical results that are<br />
available support these conjectures; but certainly more convincing evidence based<br />
on rigorous research is needed. Several chapters in this book point in this direction<br />
and provide the framework for formulating future research plans.<br />
REFERENCES<br />
Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo S. and Parikesit. 2006.<br />
Commercialization of homegardens in an Indonesian village: vegetation composition<br />
and functional changes. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens:<br />
A time-tested example of sustainable agroforestry, pp 233 – 250. Springer Science,<br />
Dordrecht.<br />
Agelet A., Angels B.M. and Valles J. 2000. <strong>Homegardens</strong> and their role as a main source of<br />
medicinal plants in mountain regions of Catalonia (Iberian Peninsula). Econ Bot 54:<br />
295 – 309.<br />
Brownrigg L. 1985. Home Gardening in International Development: What the literature<br />
shows. The League for International Food Education, Washington, DC, 330p.
INTRODUCTION<br />
I<br />
Christanty L. 1990. <strong>Homegardens</strong> in tropical Asia with special reference to Indonesia.<br />
In: Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 9 – 20. United Nations<br />
University Press, Tokyo.<br />
Drescher A.W., Holmer R.J. and Iaquinta D.L. 2006. Urban homegardens and allotment<br />
gardens for sustainable livelihoods: management strategies and institutional environments.<br />
In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 317 – 338. Springer Science, Dordrecht.<br />
Fernandes E.C.M. and Nair P.K.R. 1986. An evaluation of the structure and function of<br />
tropical homegardens. Agric Syst 21: 279 – 310.<br />
Fernandes E.C.M., O’Kting’ati A. and Maghembe J. 1984. Chagga homegardens: a multistory<br />
agroforestry cropping system on Mt. Kilimanjaro, northern Tanzania. Agroforest Syst 2:<br />
73 – 86.<br />
Gold M.A. and Hanover J.W. 1987. Agroforestry systems of the temperate zone. Agroforest<br />
Syst 5: 109 – 21.<br />
Herzog F. 1998. Streuobst: a traditional agroforestry system as a model for agroforestry<br />
development in temperate Europe. Agroforest Syst 42: 61 – 80.<br />
High C. and Shackleton C.M. 2000. The comparative value of wild and domestic plants<br />
in homegardens of a South African rural village. Agroforest Syst 48: 141 – 156.<br />
Kumar B.M. 2006. Carbon sequestration potential of tropical homegardens. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 185 – 204. Springer Science, Dordrecht.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Nair P.K.R. (ed.). 1989. Agroforestry systems in the tropics. Kluwer, Dordrecht, 664p.<br />
Nair P.K.R. 1993. An introduction to agroforestry. Kluwer, Dordrecht, 499p.<br />
Nair P.K.R. 2006. Wither homegardens? In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 355 – 370. Springer<br />
Science, Dordrecht.<br />
Randhawa M.S. 1980. The history of Indian agriculture, vol. 2, pp 67 – 68 and 414 – 415.<br />
Indian Council of Agricultural Research, New Delhi.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: a traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development,<br />
pp 157 – 170. ICRAF, Nairobi.<br />
Soini E. 2005. Changing livelihoods on the slopes of Mt. Kilimanjaro, Tanzania: challenges<br />
and opportunities in the Chagga homegarden system. Agroforest Syst 64: 157 – 167.<br />
Tesfaye Abebe, Wiersum, K.F., Bongers, F. and Sterck, F. 2006. Diversity and dynamics<br />
in homegardens of southern Ethiopia. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 123 – 142. Springer<br />
Science, Dordrecht.<br />
Thaman R.R., Elevitch C.R. and Kennedy J. 2006. Urban and homegarden agroforestry in<br />
the Pacific islands: current status and future prospects. In: Kumar B.M. and Nair P.K.R.<br />
(eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp 25 –<br />
41. Springer Science, Dordrecht.<br />
Vogl C.R. and Vogl-Lukasser B. 2003. Tradition, dynamics and sustainability of plant species<br />
composition and management in homegardens on organic and non-organic small scale<br />
farms in Alpine Eastern Tyrol, Austria. Biol Agric Hortic 21: 349 – 366.<br />
Wenhua L. (ed.). 2001. Integrated farming systems at different scales. In: Agro-ecological<br />
farming systems in China, Chapter 12, pp 201 – 252. UNESCO Man and Biosphere Series<br />
26, Partheon Publishing, New York.<br />
Westmacott R.N. 1992. African-American gardens and yards in the rural south. University<br />
of Tennessee Press, Knoxville, TN, 198p.<br />
9
10<br />
P.K.R. NNAIR AND B.M. KUMAR<br />
Wiersum K.F. 1982. Tree gardening and taungya in Java: Examples of agroforestry<br />
techniques in the humid tropics. Agroforest Syst 1: 53 – 70.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.<br />
Workman S.W., Bannister M.E. and Nair P.K.R. 2003. Agroforestry potential in the southeastern<br />
United States: Perceptions of landowners and extension professionals. Agroforest<br />
Syst 59: 73 – 83.<br />
Yamada M. and Osaqui H.M.L. 2006. The role of homegardens for agroforestry development:<br />
Lessons from Tomé-Açu, a Japanese-Brazilian settlement in the Amazon. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 299 – 316. Springer Science, Dordrecht.<br />
Zhaohua Z., Mantang C., Shiji W. and Youxu J. (eds). 1991. Agroforestry systems in China.<br />
Chinese Academy of Forestry, Beijing, and International Development Research Centre,<br />
Singapore, 216p.
SECTION 1<br />
HISTORICAL AND REGIONAL<br />
PERSPECTIVES
CHAPTER 2<br />
DIVERSITY AND CHANGE<br />
IN HOMEGARDEN CULTIVATION<br />
IN INDONESIA<br />
K.F. WIERSUM<br />
Forest and Nature Conservation Policy group, Department of Environmental<br />
Sciences, Wageningen University, The Netherlands;<br />
E-mail: <br />
Keywords: Homegarden dynamics, Rural transformations, Social sustainability.<br />
Abstract. <strong>Homegardens</strong> have been described as traditional agroforestry systems that are<br />
ecologically and socially sustainable. The concept of social sustainability has two dimensions:<br />
positive role to present livelihood conditions and ability to respond to socioeconomic<br />
changes. The dynamics of homegardens and its repercussions on social sustainability have<br />
received relatively little research attention. On the basis of results of extensive studies in Java<br />
and other parts of Indonesia, this article summarizes the historic and recent developments in<br />
the homegardening context. The structure and composition of homegardens depend both on<br />
their position in the overall farming system and on livelihood strategies of the managers.<br />
Rural transformations result in changes in livelihoods and farming systems, and have impacts<br />
on homegarden function and composition. The opinions of various authors on homegarden<br />
dynamics range from positive to negative; the former consider that changes in homegarden<br />
features are associated with socio-professional changes of villagers and the rural-urban<br />
interface, while the latter view these changes as indicative of the demise of a traditional<br />
system and argue for its revitalization. These different opinions represent different norms in<br />
assessing social sustainability of homegardens and differences in value judgments on the ideal<br />
structure of homegardens.<br />
1. INTRODUCTION<br />
Homegardening has been hypothesized as being the oldest form of agriculture in<br />
Southeast Asia. Its origin has been associated with fishing communities living in the<br />
13<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 13–24.<br />
© 2006 Springer. Printed in the Netherlands.
14 K.F. WWIERSUM<br />
moist tropical region of Southeast Asia during 13 000 to 9000 B.C. In these regions<br />
an assured supply of fish and shells allowed fixed settlements and a relatively high<br />
population density, while the fertile soils along rivers and coasts favored cultivation<br />
(Sauer, 1969). As happened also in other regions (Miller et al., 2006), homegardening<br />
probably started as a spontaneous growth of plants from leftovers of<br />
products brought to the camps of the hunter/gatherers. Gradually, the accidental<br />
propagation became more deliberate with valuable species being planted to facilitate<br />
their use. At first such cultivation probably involved vegetative propagation techniques<br />
and only later seeding was introduced (Sauer, 1969). The earliest evidence of garden<br />
cultivation dates back to at least 3000 B.C. (Soemarwoto, 1987).<br />
From these pre-historic and probably scattered origins, homegardens has<br />
gradually spread to many humid regions in South- and Southeast Asia including<br />
Java (Indonesia), the Philippines, Thailand, Sri Lanka, India and Bangladesh. For<br />
instance, according to Randhawa (1980), travelers already described homegardens<br />
with coconut (Cocos nucifera), black pepper (Piper nigrum), ginger (Zingiber<br />
officinale), sugarcane (Saccharum officinarum) and pulses (grain legumes) in<br />
Kerala, India, in the early 14th century, while Michon (1983) mentions that tree<br />
gardening systems were already common on the Indonesian island of Java in the<br />
tenth century AD. In all these regions, homegardening is almost always practiced in<br />
combination with other types of land use. The original association with gathering<br />
and fishing was gradually extended to shifting cultivation and permanent cropping.<br />
In the most widely studied homegarden systems in South- and Southeast Asia such<br />
as in Java (Soemarwoto, 1987), Kerala (Nair and Sreedharan, 1986; Kumar et al.,<br />
1994), and Sri Lanka (Jacob and Alles, 1987; McConnell, 1992), gardening is<br />
combined with permanent field cultivation often in the form of wetland rice (Oryza<br />
sativa) production. These regions with good farming conditions and relatively high<br />
population densities contributed to optimal development of the complementary<br />
system of staple food cultivation in open fields and supplementary diversified<br />
homegarden production for self-sufficiency and trade.<br />
Since the recognition of agroforestry as a type of land use worthy of research and<br />
development, homegardens have been considered as an excellent example of a<br />
traditionally developed agroforestry system with good promise for the future<br />
(Soemarwoto, 1984; Hochegger, 1998; Gajaseni and Gajaseni, 1999). Much attention<br />
has been given to analyzing the structure and function of tropical homegardens and<br />
describing their features in respect to both ecological and socioeconomic sustainability<br />
(Torquebiau, 1992; Kumar and Nair, 2004). Regarding socioeconomic<br />
sustainability, these studies focused specifically on the roles of homegardens within<br />
the livelihood systems of rural producers. A commonly perceived indicator of<br />
homegardens’ socioeconomic sustainability is the fact that homegardens typically<br />
contribute towards nutritional security, energy needs and income generation even<br />
under conditions of high population densities (Kumar and Nair, 2004). Recently it<br />
has been remarked, however, that the concept of socioeconomic sustainability<br />
should not only be related to the homegardens’ function in the present livelihood<br />
conditions, but also to their ability to adjust to socioeconomic changes (Peyre et al.,<br />
2006). At present, many rural areas are undergoing major transformations involving<br />
diversification of rural livelihood strategies (Ellis, 1998; Ashley and Maxwell,
DIVERSITY AND CHANGE C IN HOMEGARDEN CULTIVATION IN INDONESIA<br />
I<br />
2001). Due to commercialization, cultivation systems are becoming more specialized<br />
on the one hand, and rural people are increasingly employed in non-primary production<br />
activities on the other. As a result, in many rural areas, farming systems in<br />
general, and homegardens in particular, are changing. Kumar and Nair (2004) have<br />
even posed the question as to whether homegardens are becoming extinct. This<br />
illustrates that the notion of socioeconomic sustainability of homegardens should be<br />
interpreted as referring not only to their ability to contribute towards the livelihood<br />
needs of traditional rural dwellers, but also to their ability to adjust to the process of<br />
rural change.<br />
In contrast to studies on homegarden diversity, relatively little attention has been<br />
given to assessing the dynamics of homegardens. It seems that, since many studies<br />
in the past have been focused on ascertaining factors that explain the ecological<br />
stability of homegardens (Kumar and Nair, 2004), the concept of sustainability has<br />
mainly been attributed as referring to stability in an ecological sense, and that the<br />
concept of socioeconomic sustainability was by association interpreted as referring<br />
to livelihood stability. Only recently have the dynamics of homegardens been<br />
receiving some attention. In some studies, the traditional homegarden structure and<br />
composition is taken as ideal, and changes such as loss in some of the traditional<br />
species and structure are discussed in terms of homegardens becoming extinct<br />
(Kumar and Nair, 2004) and needing revitalization (Parikesit et al., 2004), while<br />
some other studies have tried to relate the various types of dynamics in homegarden<br />
structure and composition to the process of rural transformations (Michon and Mary,<br />
1994; Peyre et al., 2006).<br />
This review will assess the dynamics of homegarden development in Indonesia,<br />
focusing specifically on Java. First, it will describe the historic developments of<br />
homegardens on Java. Next, using data from both Java and Sulawesi, it will<br />
summarize the factors that impact on the structure and composition of homegardens<br />
and describe how under the influence of these factors different types of homegardens<br />
have evolved. On the basis of these data, the main trends in changing homegarden<br />
structure and composition will be summarized.<br />
2. THE DEVELOPMENT OF HOMEGARDENS IN JAVA<br />
The first studies on tropical homegardens in Southeast Asia that were started in the<br />
late 1940s in Java, Indonesia (Terra, 1953a; 1953b) remained relatively unnoticed<br />
for several years. For example, even in the 1970s it was noted that, in contrast to the<br />
open-field land use systems, homegardens had hardly yet been subject to detailed<br />
study (Stoler, 1978). This situation changed in the late 1970s when a series of new<br />
homegarden studies were initiated in Java (Soemarwoto, 1987; Soemarwoto and<br />
Conway, 1991). The Javanese experiences formed an important source of information<br />
when in the 1980s the potential of homegardens to contribute towards<br />
increasing food production and reducing malnutrition in tropical countries received<br />
greater international interest (Niñez, 1984; Brownrigg, 1985). This international<br />
interest in homegardens was further stimulated by the recognition of homegardens<br />
as a typical example of a multistoried agroforestry system (Nair and Sreedharan,<br />
1986; Jacob and Alles, 1987). The first international conference on tropical<br />
15
16<br />
K.F. WIERSUM W<br />
homegardens organized in Java in 1985 (Landauer and Brazil, 1990) is a testament<br />
to the leading role of the homegarden research in Java during that period.<br />
The extensive research on Javanese homegardens has contributed significantly to<br />
the present understanding of the structure and function of tropical homegardens. The<br />
Javanese homegardens demonstrate the typical functions of homegardens as<br />
summarized by Kumar and Nair (2004): they yield products with high nutritional<br />
value (proteins, vitamins, and minerals), medicinal plants and spices, firewood, and<br />
sometimes a1so forage crops and construction wood; all these products are used to<br />
supplement the staple food crops that are usually produced in open-field cultivation<br />
systems. Normally, the homegarden products provide a small, continuous flow of<br />
these supplementary products for subsistence and a possible small surplus for sale<br />
through local markets. In times of sudden necessities (unfavorable climatic conditions or<br />
social necessities like marriage), higher production and marketing levels may be<br />
attained (Wiersum, 1982).<br />
In many homegarden studies (Kumar and Nair, 2004), these gardens have been<br />
described as a distinct agroforestry system with a set of generic features. Relatively<br />
little attention has been given to studying the diversity within homegardens as well<br />
as their relation to the surrounding land use systems. Moreover, in addition to home-<br />
gardens, other types of tree gardening systems consisting of a mixture of several<br />
cultivated fruit- and other trees and crops exist (Wiersum, 2004), and the distinction<br />
between homegardens and other types of tree gardening systems is not straightforward.<br />
In Java, Terra (1953a; 1953b) originally differentiated three different types<br />
(see also Wiersum, 1982; Soemarwoto, 1984; Christanty et al., 1986):<br />
• The homegarden (pekarangan): fenced-in gardens, surrounding individual<br />
houses, planted with fruit- and other trees, vegetable herbs and annual crops.<br />
Historically they are associated with wetland rice fields and more recently also<br />
with dry fields. They occurred in regions with individual land-ownership.<br />
Typically these homegardens occur in Central Java and are inhabited by the<br />
Javanese people.<br />
• The tree garden (kebun or talun): mixed tree plantations on communal lands<br />
surrounding villages with dense clusters of houses, sometimes also at some<br />
distance from the villages. These plots are not inhabited and they are historically<br />
associated with shifting cultivation. They occur in regions with communally<br />
owned land. Mostly they are found in West Java and are inhabited by the<br />
Sundanese people. These tree gardens are much less tended than homegardens<br />
and often include more wild trees than present in the homegardens.<br />
• Clumps of fruit- or other trees planted on abandoned shifting cultivation sites.<br />
Such plantings could denote a right of priority of these lands for the people who<br />
planted the trees in an area of otherwise communal land ownership.<br />
As demonstrated by the characterizations, the tree gardening systems in Java<br />
normally forms a sub-set of an integrated farming system (Terra, 1958), which also<br />
comprises annually cultivated fields used for the production of staple, high calorific<br />
foods such as rice, maize (Zea mays) and cassava (Manihot esculenta). Consequently,<br />
the structure and function of homegardens significantly depends on the nature of the<br />
overall farming system.
DIVERSITY AND CHANGE C IN HOMEGARDEN CULTIVATION IN INDONESIA<br />
I<br />
Over the ages, gradual changes have taken place in these systems (Soemarwoto,<br />
1984). The most important was perhaps the extension of the Javanese culture and<br />
subsequent spread of homegardens. For instance, in the eighteenth century, the<br />
pekarangan system was already practiced in West Java, where it partly replaced the<br />
talun system of the Sundanese (Michon, 1983). Also, gradually communal lands<br />
were divided among individual landowners, who by building houses in such individual<br />
tree gardens, converted them to homegardens. In other tree gardens, annual<br />
crops were introduced and management became more intensive. Also shifting<br />
cultivation virtually disappeared and in areas with clumps of planted trees on<br />
fallow lands, a conversion to tree gardens took place. According to Wiersum (1982),<br />
in the early 1980s it was possible to distinguish the following three types of tree<br />
gardening:<br />
• <strong>Homegardens</strong> (pekarangan): a land use form on private lands surrounding individual<br />
houses with a definite fence, in which several tree species are cultivated<br />
together with annual and perennial crops, often including small livestock.<br />
• Mixed gardens (kebun campuran): a land use form on private lands outside the<br />
village, which is dominated by planted perennial crops, mostly trees, under<br />
which annual crops are cultivated.<br />
• Forest gardens (talun, kebun): a land use form on private lands outside the<br />
village in which planted and sometimes spontaneously grown trees and sometimes<br />
additional perennial crops occur.<br />
The pekarangan is often considered as a typical prototype for homegardens. But<br />
as illustrated by the diversity of tree gardening system in Java, the distinction<br />
between homegardens and other types of tree-gardening systems is often diffuse and<br />
may be related more to location than to vegetation structure 1 . Moreover, homegarden<br />
structure may gradually change with time.<br />
3. DIVERSITY IN HOMEGARDEN STRUCTURE AND COMPOSITION<br />
The diversity in tropical homegardens types is not only illustrated by the historic<br />
developments in tree gardening systems, but also by the existing variation in<br />
homegarden structure and composition. Several homegarden studies in Java have<br />
assessed what factors impact on the homegarden structure and composition as well<br />
as function. Karyono (1990) demonstrated that homegarden composition was<br />
affected both by geographic conditions and their role in the farming systems.<br />
Compared to lowlands, homegardens in highland areas have lower plant diversity<br />
and simpler species composition. Also a different pattern of species composition<br />
exists in homegardens associated with irrigated rice production as opposed to those<br />
associated with dry-land agriculture: fruit species are dominant in the former, and<br />
food crops in the latter. Stoler (1978) also emphasized the relation between garden<br />
composition (as well as management intensity) and other components of the farming<br />
system. Households with sufficient croplands to produce rice to cover basic staple<br />
food requirements cultivated more commercial fruit trees than households who<br />
could not meet staple food requirements from croplands and hence had to cultivate<br />
more subsistence crops in the homegardens. Christanty (1990) differentiated urban<br />
17
18<br />
K.F. WIERSUM W<br />
and rural homegardens, and mentioned that these could be further classified<br />
depending on:<br />
• The dominant plant species grown, e.g., fruit, vegetable, or flower species, and<br />
• The main function of the homegarden, e.g., subsistence garden, kitchen garden,<br />
market garden, plant nursery garden, and aesthetic garden.<br />
Soemarwoto (1984) added that in rural areas homegardens have important social<br />
functions through the provision of gifts in the form of fruits, leaves or products for<br />
religious or medicinal purposes. In urban areas this social function diminishes<br />
whereas their aesthetic function increases with ornamentals replacing food crops.<br />
Michon and Mary (1994) and Abdoellah et al. (2006) described that, in addition to<br />
urbanization, the rise of a market economy profoundly influences the homegarden<br />
function resulting in an increase in commercial crops. Abdoellah (1990) reported<br />
that the effect of various cultures (Javanese or Sundanese) was often still reflected in<br />
the structure of homegardens: for example, vegetables and ornamentals were often<br />
more common in Sundanese homegardens.<br />
Also in the Indonesian island of Sulawesi different types of homegardens have<br />
been reported. For example, Kehlenbeck and Maass (2004) described four homegarden<br />
types distinguished by differences in garden age and size, and the level of<br />
diversity:<br />
1. Small, moderately old, species- and tree-poor spice gardens<br />
2. Medium-sized, old, species-rich fruit tree gardens<br />
3. Large, rather young, species- and tree-poor gardens of transmigrant<br />
families<br />
4. Diverse assemblages of rather old, individual gardens with very high crop<br />
diversity.<br />
According to Terra (1958), the typical Javanese landscape with irrigated rice<br />
fields, dry croplands and mixed gardens was already common in this region in the<br />
1950s. The types 2 and 4 mentioned above may reflect this traditional situation. But<br />
as illustrated by type 3, recently the area is becoming further settled by transmigrants<br />
from Java. These transmigrants do not only open up new agricultural lands,<br />
but also establish homegardens around their new settlements. Such homegarden<br />
development takes time. Often, at first essential food crops are grown and only<br />
gradually supplementary crops are introduced. Other factors influencing homegarden<br />
structure are related to differences in access to markets and availability of<br />
garden products in the market. Moreover, the composition is found to be influenced<br />
by official homegarden development programs (Kehlenbeck and Maass, 2006).<br />
In other studies on Asian homegardens too, several geographic and socioeconomic<br />
factors have been found to influence the homegarden structure and<br />
composition (e.g., Kumar et al., 1994; John and Nair, 1999; Peyre et al., 2006).<br />
Table 1 summarizes the various factors that have been reported to impact on<br />
homegarden composition. As illustrated in this table, notably livelihood conditions<br />
are an important factor influencing the structure and composition of homegardens.<br />
Livelihood conditions are reflected in both the farming system and the<br />
socioeconomic status of households. For poor people, homegardens may form the<br />
only land available to them for primary production, and consequently they are likely
to serve partly for production of essential staple foods rather than only for supplementary<br />
crop production. On the other hand, for affluent people living in urbanized<br />
areas and having access to non-farm incomes, homegardens may not any longer<br />
form a part of a farming system, but function only as an ornamental area around the<br />
living quarters. Thus, not only the overall livelihood conditions, but also specific<br />
socioeconomic variables such as access to land or off-farm labor opportunities<br />
impact the homegarden structure and composition. Generally, a decrease in the<br />
availability of land results in intensification of cultivation and the inclusion of more<br />
annual crops. Also, when alternative income opportunities are present, cultivation is<br />
“extensified” (and more ornamentals are included near urban areas). Where better<br />
marketing opportunities exist (near cities), specialization in fruit production may<br />
take place.<br />
Table 1. Factors impacting structure and composition of homegardens with special reference<br />
to Indonesian homegardens.<br />
Factors Conditions Examples and remarks<br />
Geographic<br />
location<br />
Environmental<br />
conditions<br />
Role in farming<br />
systems<br />
Socioeconomic<br />
conditions of the<br />
household<br />
DIVERSITY AND CHANGE C IN HOMEGARDEN CULTIVATION IN INDONESIA<br />
I<br />
Urban versus rural Urban homegardens often smaller and more<br />
location<br />
aesthetic oriented<br />
Climate conditions Variation in annual crops cultivated only in<br />
favorable climatic seasons is mostly less pronounnced<br />
than in permanent crops that have to be<br />
adapted to variable climatic conditions over much<br />
larger periods<br />
Soil conditions With decreasing soil fertility crop diversity tends<br />
to decrease and the effect of competition by trees<br />
on understorey becomes more pronounced. Dense<br />
tree gardens occur mostly on volcanic soils, while<br />
on tertiary soils tree gardens are more open<br />
Degree of complementarity<br />
to open field<br />
cultivation systems<br />
Established versus<br />
incipient farming system<br />
19<br />
If homegardens are the only land asset more<br />
inclusion of staple food crops<br />
Incipient gardens first dominated by annual crops,<br />
with time increased incorporation tree crops<br />
Wealth status With increased wealth increased importance of<br />
commercial and aesthetic plants<br />
Access to markets Commercial crops stimulated by good market<br />
access<br />
Access to off-farm In case of access to financially lucrative employ-<br />
employment<br />
ment decreased importance commercial crops<br />
Gender-related issues Gardens of female-headed households often more<br />
household use oriented<br />
Cultural factors Food preferences Cultural preferences in respect to consumption of<br />
vegetables and spices
20<br />
Up to a certain level, the cultivation of homegardens can respond well to changes<br />
in socioeconomic conditions by means of intensification of cultivation, shifting the<br />
ratio of perennials to annuals and sometimes domestic animals, and a certain degree<br />
of specialization in crops. But major differences in socioeconomic status are<br />
reflected in homegardens having a clearly different composition. It is possible to<br />
differentiate various types of homegardens in respect to their role in the household<br />
economics (Table 2).<br />
Table 2. Different types of homegardens in relation of household economics.<br />
Homegarden type Characteristics<br />
Survival gardens Gardens form single component farming system of<br />
otherwise landless rural people<br />
Combined production of staple food crops and comple-<br />
mentary crops<br />
Subsistence gardens Part of multi-component farming system in conjuncttion<br />
with permanent or shifting field production<br />
Complementary system to open-field staple food cultivation<br />
systems<br />
Provision of daily supply of vegetables, herbs, spices<br />
and fruits for household needs and occasional sale<br />
Market gardens Specialized farming system or part of multi-component<br />
farming system<br />
Cultivation of cash crops with possible complementary<br />
production of household products<br />
Budget gardens Gardens of households with economic bases in rural or<br />
urban employment; family needs are mostly purchased<br />
from the market<br />
Cultivation of ‘hobby’ products for household consumption<br />
and ornamentals<br />
Source: Adapted from Niñez (1984).<br />
K.F. WIERSUM W<br />
4. HOMEGARDEN DYNAMICS<br />
Many of the factors that impinge on homegarden structure and composition change<br />
with time, and it is therefore logical to infer that the homegarden structure and<br />
composition change whenever socioeconomic factors change (e.g., Peyre et al.,<br />
2006; Abdoellah et al., 2006). Such changes often reflect the general processes of<br />
rural changes and may involve several aspects of rural transformations. Areas that<br />
used to be remote are increasingly being incorporated into the national economy<br />
with traditional land use systems such as shifting cultivation gradually becoming<br />
transferred to more permanent cropping systems. Remote areas may also be actively<br />
opened up for migrants. In Indonesia the transmigration from the densely populated
DIVERSITY AND CHANGE C IN HOMEGARDEN CULTIVATION IN INDONESIA<br />
I<br />
island of Java to other islands is actively stimulated, and as a result the typical<br />
Javanese homegarden is being introduced in new regions. Moreover, in many rural<br />
areas the (semi)subsistence household economies of former times are increasingly<br />
becoming more commercially oriented. In others, urban life-styles are developing<br />
and the household dependence on primary production is changing to include<br />
activities in the manufacturing or service and trade sectors. In some places, these<br />
dynamics are intensive; in others they take place more gradually. Depending on the<br />
nature and intensity of rural changes, the developments in structure and composition<br />
as well as functions of homegardens may show different trends (Table 3).<br />
Table 3. Main trends in homegarden development.<br />
Main trends Consequences<br />
Extension in area<br />
Extension of housing due to population<br />
growth<br />
Extension to new areas due to change in<br />
farming systems, e.g., from shifting cultivation<br />
to permanent cultivation<br />
Extension to new areas due to migration<br />
Changing structure and composition<br />
Adaptation of gardens to new food habits<br />
and changing household needs or new<br />
agronomic practices<br />
Increasing commercialization<br />
• Decreasing importance subsistence<br />
production<br />
• Increasing commercial production<br />
Increasing role of aesthetic function garden<br />
Prevalence of (bi)annual food crops in<br />
newly established gardens<br />
Young homegardens have not yet<br />
reached full diversity<br />
Extension of homegarden to new areas<br />
with adaptation to different land use<br />
systems than in area of origin<br />
Gradual change in structure and composition<br />
including incorporation of new<br />
species<br />
Decreasing importance of supplementary<br />
production<br />
Increasing specialization on either<br />
vegetable, spices or fruit tree production<br />
Increase in ornamental plants<br />
5. CONCLUSIONS<br />
<strong>Homegardens</strong> have often been described as a sustainable agroforestry system with<br />
positive ecological and socioeconomic features. While several studies have<br />
explicitly highlighted homegardens as traditional systems, relatively little attention<br />
has been given to studies on the dynamics of homegardens under influence of rural<br />
transformations. Nonetheless, several studies have demonstrated that homegarden<br />
function and composition depends greatly on socioeconomic conditions as well as<br />
household livelihood strategies. In this context, not only different types of<br />
21
22<br />
K.F. WIERSUM W<br />
homegarden systems can be recognized, but also different pathways of homegarden<br />
development can be identified. The changes in homegarden function and<br />
composition have been interpreted differently by various authors. Some argue that<br />
although the traditional homegardens have gradually lost their original ecological<br />
and economic features, they still are a major asset for the modernization of village<br />
economy and society. The changes in homegarden features are associated with<br />
socio-professional changes of villagers and reflect a search for a new balance in the<br />
relationship between cities and villages. Other authors take a more negative point of<br />
view of the dynamics in tree gardening systems; they view the changes under<br />
influences of rural dynamics as the disappearance of a traditional system and<br />
propose measures to revitalize such traditional tree gardening systems. These<br />
different and somewhat opposing views on the trends in homegarden function<br />
and composition represent different norms in assessing social sustainability<br />
of homegardens and differences in value judgments on the ideal structure of<br />
homegardens.<br />
ENDNOTE<br />
1. The international literature on tropical homegardens is often ambivalent on<br />
whether homegardens are characterized by structure or location.<br />
REFERENCES<br />
Abdoellah O.S. 1990. <strong>Homegardens</strong> in West Java and their future development. In: Landauer<br />
K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 69 – 79. United Nations University<br />
Press, Tokyo.<br />
Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo S. and Parikesit 2006.<br />
Commercialization of homegardens in an Indonesian village: vegetation composition and<br />
functional changes. Agroforest Syst (in press).<br />
Ashley C. and Maxwell S. 2001. Rethinking rural development. Dev Policy Rev 19:<br />
395 – 425.<br />
Brownrigg L. 1985. Home gardening in international development: What literature shows?<br />
The League for International Food Education, Washington, DC, 330p.<br />
Christanty L. 1990. <strong>Homegardens</strong> in tropical Asia with special reference to Indonesia. In:<br />
Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 9 – 20. United Nations<br />
University Press, Tokyo.<br />
Christanty L., Abdoellah O.S., Marten G.G. and Iskander J. 1986. Traditional agroforestry in<br />
West Java: the pekarangan (homegarden) and kebun-talun (annual-perennial rotation)<br />
cropping system. In: Marten G.G. (ed.), Traditional agriculture in South East Asia, pp<br />
132 – 158. Westview Press, Boulder, CO.<br />
Ellis F. 1998. Household strategies and rural livelihood diversification. J Dev Stud 35: 1 – 38.<br />
Gajaseni J. and Gajaseni N. 1999. Ecological rationalities of the traditional homegarden<br />
system in the Chao Phraya Basin, Thailand. Agroforest Syst 46: 3 – 23.<br />
Hochegger K. 1998. Farming like the forest: traditional homegarden systems in Sri Lanka.<br />
<strong>Tropical</strong> agroecology. Margraf Verlag, Weikersheim, 203p.<br />
Jacob V.J. and Alles W.S. 1987. The Kandyan gardens of Sri Lanka. Agroforest Syst 5:<br />
123 – 137.
DIVERSITY AND CHANGE C IN HOMEGARDEN CULTIVATION IN INDONESIA<br />
I<br />
John J. and Nair M.A. 1999. Socio-economic characteristics of homestead farming in south<br />
Kerala. J. Trop Agric 37: 107 – 109.<br />
Karyono 1990. <strong>Homegardens</strong> in Java: their structure and function. In: Landauer K. and Brazil<br />
M. (eds), <strong>Tropical</strong> home gardens, pp 138-146. United Nations University Press, Tokyo.<br />
Kehlenbeck K. and Maass B.L. 2004. Crop diversity and classification of homegardens in<br />
Central Sulawesi, Indonesia. Agroforest Syst 63: 53 – 62.<br />
Kehlenbeck K. and Maass B.L. 2006. Are tropical homegardens sustainable? Some evidence<br />
from Central Sulawesi, Indonesia. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 339 – 354. Springer<br />
Science, Dordrecht.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Kumar B.M., George S.J. and Chinnamani S. 1994. Diversity, structure and standing stock of<br />
wood in homegardens of Kerala in peninsular India. Agroforest Syst 25: 243 – 262.<br />
Landauer K. and Brazil M. (eds) 1990. <strong>Tropical</strong> home gardens. United Nations University<br />
Press, Tokyo, 257p.<br />
McConnell D.J. 1992. The forest-garden farms of Kandy, Sri Lanka. Farm systems management<br />
Series No. 3, FAO, Rome, 117p.<br />
Michon G. 1983. Village-forest-gardens in West Java. In: Huxley P.A. (ed.), Plant research<br />
and agroforestry, pp 13 – 24. ICRAF, Nairobi.<br />
Michon G. and Mary F. 1994. Conversion of traditional village gardens and new economic<br />
strategies of rural households in the area of Bogor, Indonesia. Agroforest Syst<br />
25: 31 – 58.<br />
Miller R.P., Penn, Jr. J.W. and Leeuwen J. van. 2006. Amazonian homegardens: their<br />
ethnohistory and potential contribution to agroforestry development In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 43 – 60. Springer Science, Dordrecht.<br />
Nair M.A. and Sreedharan C. 1986. Agroforestry farming systems in the homesteads of<br />
Kerala, southern India. Agroforest Syst 4: 339 – 363.<br />
Niñez V.K. 1984. Household gardens: theoretical considerations on an old survival strategy.<br />
Potatoes in Food Systems Research Series Report No. 1. International Potato Center,<br />
Lima, Peru, 41p.<br />
Parikesit, Takeuchi K., Tsunekawa A. and Abdoellah O.S. 2004. Kebon tatangkalan: a<br />
disappearing agroforest in the Upper Citarum watershed, West Java, Indonesia.<br />
Agroforest Syst 63: 171 – 182.<br />
Peyre A., Guidal A., Wiersum K.F. and Bongers F. 2006. Dynamics of homegarden structure<br />
and function in Kerala, India. Agroforest Syst 66: 101 – 115.<br />
Randhawa M.S. 1980. The history of Indian agriculture. Vol. 2, pp 414 – 415. Indian Council<br />
of Agricultural Research, New Delhi.<br />
Sauer C.O. 1969. Agricultural origins and dispersals (2 nd ed.). M.I.T. Press, New York. 175p.<br />
Soemarwoto O. 1984. The Javanese rural ecosystem. In: Rambo A.T. and Sajise P.E. (eds),<br />
An introduction to human ecology research on agricultural systems in Southeast Asia,<br />
pp 254 – 287. University of Philippines, College, Los Banos.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: a traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development,<br />
pp 157 – 170. ICRAF, Nairobi.<br />
Soemarowoto O. and Conway G.R. 1991. The Javanese homegarden. J Farming Syst Res<br />
Extn 2: 95 – 118.<br />
Stoler A. 1978. Garden use and household economy in rural Java. Bull Indonesian Econ Stud<br />
14: 85 – 101.<br />
23
24<br />
K.F. WIERSUM W<br />
Terra G.J.A. 1953a. Mixed-garden horticulture in Java. Malaysian J Trop Geogr 1: 33 – 43.<br />
Terra G.J.A. 1953b. The distribution of mixed gardening on Java. Landbouw (Indonesia)<br />
25: 163 – 203.<br />
Terra G.J.A. 1958. Farm systems in South-east Asia. Neth J Agric Sci 6: 157 – 182.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Wiersum K.F. 1982. Tree gardening and taungya on Java: examples of agroforestry<br />
techniques in the humid tropics. Agroforest Syst 1: 53 – 70.<br />
Wiersum K.F. 2004. Forest gardens as an ‘intermediate’ land use system in the nature-culture<br />
continuum: characteristics and future potential. Agroforest Syst 61: 123 – 134.
CHAPTER 3<br />
URBAN AND HOMEGARDEN<br />
AGROFORESTRY IN THE PACIFIC<br />
ISLANDS: CURRENT STATUS AND<br />
FUTURE PROSPECTS<br />
R.R. THAMAN 1 , C.R. ELEVITCH 2 , AND J. KENNEDY 3<br />
1<br />
The University of the South Pacific, Suva, Fiji Islands; 148; E-mail:<br />
. 2 3<br />
Agroforestry.net, Holualoa, Hawai’i. Research School of<br />
Pacific and Asian Studies, Australian National University, Canberra, Australia<br />
Keywords: Agrodeforestation, Food security, Nutrition, Sustainability, Traditional agroforestry.<br />
Abstract. Pacific islanders traditionally had abundant, predominantly rural, agroforestry<br />
systems that provided a wide array of products for meeting the necessities of life, and<br />
conducive environments for the rich Pacific island cultures. In recent years, however,<br />
increasing urbanization and accompanying removal of trees and perennial agroforests<br />
(“agrodeforestation”) have resulted in the breakdown of these traditional agroforestry<br />
systems, accompanied by increasing economic, cultural, nutritional, and environmental<br />
problems, particularly in the urban areas. A critical analysis of the nature and future prospects<br />
of the urban and homegarden agroforestry systems in these rapidly urbanizing islands<br />
suggests that intensification and enrichment of these systems could serve as an important<br />
foundation for sustainable development. In addition to addressing the nutrition-related health<br />
problems, food security, poverty alleviation, and trade deficits, these systems also help protect<br />
and enrich the cultural traditions of Pacific peoples who are increasingly out-migrating from<br />
rural areas and embracing urban living.<br />
1. INTRODUCTION<br />
Pacific island countries have historically been resource self-reliant because of their<br />
relative geographic isolation. Their traditional land- and sea-based economies,<br />
25<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 25–41.<br />
© 2006 Springer. Printed in the Netherlands.
26 R.R. THAMAN T<br />
ET AL.<br />
cultures, and isolation from major markets forced island communities to develop<br />
sustainable land use systems such as agroforestry and freshwater and marine fishery<br />
production systems. Today, however, the small island states of the Pacific Ocean are<br />
rapidly urbanizing and increasingly large populations no longer have access to<br />
traditional agricultural and wildland holdings. Moreover, trees are disappearing from<br />
both rural and urban areas, a process referred to as “agrodeforestation” (Thaman,<br />
1992). Studies in the Ha’ppai Islands of Tonga, for example, showed that about 100<br />
traditionally important trees or shrubs, all integral components of the traditional<br />
Tongan bush-fallow agroforestry, were reported to be endangered or in short supply.<br />
These included 28 large wild trees, 32 large cultivated trees, 19 wild small trees or<br />
shrubs, and 24 planted small trees and shrubs (Thaman et al., 2001). Species most<br />
commonly mentioned were multipurpose trees valued for medicine, fruit, firewood,<br />
construction purposes, and fragrant plants used for body ornamentation and<br />
perfume. Foremost among the reasons for the loss of tree cover was the failure to<br />
plant or replant trees in general, as well as changing land-management practices that<br />
included indiscriminate felling, clearing and burning, and the increasing use of the<br />
plow. These practices have caused a gradual shift away from the traditional mixed<br />
agroforestry systems in which fruit trees and other culturally useful trees, such as<br />
coconut (Cocos nucifera), breadfruit (Artocarpus ( altilis),<br />
traditional banana and<br />
plantain clones (Musa spp.), citrus (Citruss spp.), Malay apple ( Syzygium malaccense)<br />
and Polynesian vi-apple (Spondias dulcis) were dominant, to monocultural<br />
production of commodities. Spread of tall grass species such as guinea grass<br />
(Panicum maximum) and the impact of grazing animals and other domesticated<br />
livestock made regeneration of trees particularly difficult (Thaman et al., 2001).<br />
Today, most Pacific island countries are increasingly dependent on imported<br />
food, fossil fuels, medicines, and other industrial products to satisfy the basic needs,<br />
with the result that they experience negative balance of payment situations. Food<br />
security has become a major concern in all of the independent island states 1 .<br />
Disappearance of forests and agroforests, which traditionally provided these<br />
products and services, may partially explain this increasing dependence on imported<br />
foods and fuels (Thaman, 1988). The people of the Pacific islands also have high<br />
rates of nutrition-related, non-communicable diseases, such as diabetes, cardiovascular<br />
disease, hypertension, hyperuricemia and gout, obesity, iron deficiency<br />
anemia, and dental diseases (Parkinson, 1982; Thaman, 1982; Coyne, 2000)—with<br />
rates of obesity as high as 75% reported in Nauru, Samoa, the Cook Islands, Tonga,<br />
and French Polynesia. The main factor for most of these diseases has been the shift<br />
in the food habits from traditional “healthy” foods to processed, imported “unhealthy”<br />
foods (Curtis, 2004).<br />
Experience from the region as well as elsewhere suggests that the promotion of<br />
urban and homegarden agroforestry may provide culturally appropriate and costeffective<br />
means of addressing both urban agrodeforestation, and many of the economic,<br />
cultural, nutritional and environmental problems arising out of urbanization and<br />
globalization (Thaman and Clarke, 1993; Thaman, 2002; Lamanda et al., 2006). It<br />
can also help to address indirectly many emerging environmental problems, such as<br />
climate change and associated sea-level rise, coastal erosion, pollution, and loss of
native trees and biodiversity. In this scenario, this chapter assesses the status and<br />
future prospects of urban and homegarden agroforestry in the Pacific islands.<br />
This chapter is based on the authors’ extensive experience in the region as well<br />
as inventories of urban and homegardens in Papua New Guinea (PNG), Fiji, Tonga,<br />
Kiribati, and Nauru during the 1970s and 1980s (Thaman, 1983; 1987), together<br />
with subsequent studies in these countries, and in New Caledonia, Solomon Islands,<br />
Vanuatu, Samoa, Niue, the Cook Islands, Tuvalu, French Polynesia, Hawai’i, the<br />
Marshall Islands, and Palau (Thaman, 1995).<br />
1.1. <strong>Homegardens</strong><br />
<strong>Homegardens</strong> are a ubiquitous feature of the Pacific island landscapes, from the very<br />
densely populated urban areas in atoll microstates, such as South Tarawa, Kiribati,<br />
Fogafale Islet on Funafuti Atoll, Tuvalu, and RETA in northeast Majuro Atoll in the<br />
Marshall Islands to rural villages and plantations in areas of low population density<br />
in Fiji, Vanuatu, and Papua New Guinea. Even in areas not known for agricultural<br />
diversity, such as Kiribati, Tuvalu, the Marshall Islands, and Nauru, homegardens<br />
contain a wide range of food trees, non-tree staple and supplementary food plants,<br />
medicinal plants, and other non-food trees and plants of cultural and economic<br />
importance (Table 1). Homegarden surveys in these localities have indicated the<br />
cultivation of 33 to 114 species or distinct types of food plants in these localities<br />
(Thaman, 1995). In Palau, where women are responsible for most gardening, homegardens<br />
with a diversity of food trees and extensive multispecies, taro-dominated<br />
agroforests are found throughout the main towns and in areas surrounding villages.<br />
Table 1. Number of species and distinct varieties of food plants found in surveys of<br />
homegardens systems in different Pacific islands.<br />
Crop types<br />
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
Number of species/varieties in different Pacific islands<br />
PNG Fiji Tonga Kiribati Nauru Location 1<br />
Non-tree staples 7 10 8 6 5 8<br />
Non-tree supplementary 48 65 44 35 14 41<br />
Food trees 2 30 39 27 20 14 16<br />
Total 85 114 79 61 33 65<br />
Source: Thaman (1995); 1 Location, a contract worker settlement in Nauru; 2 The totals for<br />
Papua New Guinea and Nauru, where banana clones and Citrus spp., respectively, were not<br />
differentiated, would have been slightly higher for tree crops if these differentiations had been<br />
made).<br />
In addition to the food plants, many useful non-food plants were also found in<br />
the homegardens. Examples include important “handicraft plants” such as Pandanus,<br />
the leaves of which are processed to make mats, thatching, baskets, hats and a wide<br />
range of other plaited ware; paper mulberry (Broussonetia papyrifera), the treated<br />
27
28<br />
bast fiber of which is used for bark cloth (tapa); and sources of dyes such as annatto<br />
(Bixa orellana) and Java cedar (Bischofia javanica). There may also be<br />
multipurpose trees such as Leucaena leucocephala, a wide range of medicinal<br />
plants, plus countless other plants of considerable technological, economic, social,<br />
ecological, and ornamental values (Table 2; Fig. 1).<br />
Table 2. Significant plant species of urban and rural Pacific island homegardens and<br />
underdeveloped urban and periurban open areas.<br />
a. <strong>Homegardens</strong><br />
Category Species<br />
Staple root<br />
crops<br />
Supplementary<br />
food crops<br />
R.R. THAMAN T ET AL.<br />
Alocasia macrorrhiza (giant taro)<br />
Colocasia esculenta (taro)<br />
Cyrtosperma chamissonis (giant swamp taro)<br />
Dioscorea alata (greater yam)<br />
Dioscorea esculenta (sweet yam)<br />
Ipomoea batatas (sweet potato)<br />
Manihot esculenta (cassava)<br />
Xanthosoma spp. (tannia or cocoyam)<br />
Abelmoschus esculentus (okra)<br />
Abelmoschus manihot t (hibiscus spinach)<br />
Allium spp. (bunching onions)<br />
Amaranthus spp. (amaranth spinach)<br />
Ananas comosus (pineapple)<br />
Arachis hypogaea (peanuts)<br />
Brassica chinensis, B. juncea and B. oleracea vars. (cabbages,<br />
including Chinese, mustard and English or head cabbages)<br />
Cajanus cajan (pigeon pea)<br />
Citrullus lanatus (watermelon)<br />
Coccinea grandis (ivy gourd)<br />
Colocasia esculenta (taro leaf spinach)<br />
Cucumis melo var. cantalupensis (cantaloupe or rock melon)<br />
Cucumis sativus (cucumber)<br />
Cucurbita pepo (pumpkin)<br />
Luffa acutangula (angled loofah or ridgegourd)<br />
Luffa cylindrica (sponge gourd)<br />
Momordica charantia (bitter gourd)<br />
Passiflora edulis (passionfruit)<br />
Phaseolus, Psophocarpus and Vigna spp. (beans and other<br />
legumes)<br />
Saccharum officinarum (sugarcane)<br />
Solanum lycopersicon (tomato)<br />
Solanum melongena (eggplant)<br />
Xanthosoma spp. (taro leaf spinach)<br />
Zea mays (corn)
Fruit and nut<br />
yielding trees<br />
Spice plants<br />
and social<br />
beverage and<br />
stimulant plants<br />
Non-food<br />
plants<br />
b. Undeveloped open areas<br />
Staple root<br />
crops<br />
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
Annona spp. (soursop and sweetsop)<br />
Artocarpus altilis and A. camansi (breadfruit and breadnut)<br />
Carica papaya (papaya)<br />
Citrus aurantifolia (limes), C. aurantium (sour orange), C. limon<br />
and C. medica x limon (lemon), C. maxima (pummelo),<br />
C. mitis (calamondin), C. reticulata (tangerine and mandarin<br />
orange), and C. sinensis (orange)<br />
Cocos nucifera (coconut)<br />
Ficus spp. (fig trees)<br />
Inocarpus fagifer r (Tahitian chestnut)<br />
Mangifera indica (mango)<br />
Musa cultivars (banana and plantains)<br />
Pandanus spp. (edible pandanus)<br />
Persea americana (avocado)<br />
Pometia pinnata (oceanic litchi)<br />
Psidium guajava (guava)<br />
Spondias dulcis (vi apple)<br />
Syzygium aqueum (water apple)<br />
Syzygium malaccense (Malay apple)<br />
Terminalia catappa (beach almond)<br />
Areca catechu (betel nut palms)<br />
Capsicum frutescens and C. annum cvs (chili)<br />
Coriandrum sativum (coriander)<br />
Cymbopogon citratus (lemon grass)<br />
Mentha spp. (mint)<br />
Piper betle (betel vine)<br />
Piper methysticum (kava)<br />
Zingiber officinale (ginger)<br />
Cananga odorata (ylang-ylang)<br />
Fagraea berteroana (pua ( )<br />
Gardenia taitensis (Tahitian gardenia)<br />
Guettarda speciosa (guettarda)<br />
Pandanus spp. (pandanus cultivars)<br />
Pimenta racemosa (bay rum)<br />
Plumeria obtusa and P. rubra ( frangipani)<br />
Colocasia esculenta (taro)<br />
Dioscorea esculenta (sweet yam)<br />
Ipomoea batatas (sweet potato)<br />
Manihot esculenta (cassava)<br />
Xanthosoma spp. (tannia or cocoyam)<br />
29<br />
Table 2 (cont.)
30<br />
Category Species<br />
R.R. THAMAN T ET AL.<br />
Fruit and nut Artocarpus altilis (breadfruit)<br />
yielding trees<br />
Carica papaya (papaya)<br />
Citrus spp. (citrus)<br />
Cocos nucifera (coconut)<br />
Mangifera indica (mango)<br />
Musa cultivars (banana and plantains)<br />
Pometia pinnata (oceanic litchi)<br />
Psidium guajava (guava)<br />
Syzygium spp.<br />
Terminalia catappa (beach almond)<br />
Non-food plants Bischofia javanica (koka)<br />
Cassia and Senna spp. (shower trees)<br />
Casuarina spp.<br />
Delonix regia (flamboyant)<br />
Erythrina variegata (coral tree)<br />
Eucalyptus spp.<br />
Ficus spp. (banyans)<br />
Gliricidia sepium (madre de cacao)<br />
Hibiscus rosa-sinensis<br />
Hibiscus tiliaceus (beach hibiscus)<br />
Lagerstroemia speciosa (pride of India)<br />
Leucaena leucocephala<br />
Macaranga spp.<br />
Morinda citrifolia (noni)<br />
Plumeria obtuse and P. rubra ( frangipani)<br />
Polyscias spp. (hedge panax)<br />
Samanea saman (rain tree)<br />
Spathodea campanulata (African tulip tree)<br />
Source: Based on Thaman (1983, 1987, 1995, 2002); Levett (1992, 1996); Levett and Uvano<br />
(1992). Categories such as ‘supplementary food crops’ and ‘spice plants and social beverage<br />
and stimulant plants’ were clearly absent in the undeveloped open areas.<br />
<strong>Homegardens</strong> also contain a great diversity of cultivars of important food and<br />
handicraft plants. As stressed by Soemarwoto et al. (1985) in their study of Javanese<br />
homegardens, true plant diversity is far greater than indicated by the numbers of<br />
species, since many species are represented by numerous cultivars. In Tonga, for<br />
example, there are numerous distinct breadfruit cultivars, the most common of<br />
which include ma’ofala, maopo, puou, loutoko, kea and ’aveloloa. There is similarly<br />
great cultivar diversity among other tree crops such as coconuts, banana, mango<br />
(Mangifera indica), pandanus (Pandanus spp.), papaya (Carica papaya), and<br />
especially among the traditional staple root crops such as yams (Dioscorea spp.),<br />
taros (Colocasia esculenta, Alocasia macrorrhiza and Xanthosoma spp.), and sweet<br />
potato (Ipomoea batatas), all of which add economic, ecological, and nutritional
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
stability to the urban gardening systems. “Tree gardens” in the settlements in Yap, in<br />
the Federated States of Micronesia, for example, had 21 coconut cultivars, 28<br />
breadfruit cultivars, and 37 banana cultivars (Falanruw, 1995). Similar cultivar<br />
diversity is found in the taro (Colocasia esculenta)-dominated agroforestry gardens<br />
in and around the main town of Koror and villages in Palau. Throughout Papua New<br />
Guinea, tree crops continue to provide a crucial component of the diverse<br />
subsistence agricultural systems of the rural population, with high cultivar diversity<br />
of many species such as bananas, breadfruit, pandanus, and many indigenous fruit<br />
and nut trees. This diversity is retained despite the addition of newly introduced<br />
high-yielding cultivars 2 .<br />
Figure 1. Homegarden in downtown Apia, Samoa includes numerous useful tree species<br />
including the fast-growing timber tree poumuli ( Flueggea flexuosa), fruit trees such as<br />
coconut (Cocos nucifera) and breadfruit (Artocarpus altilis), as well as many ornamentals<br />
(Photo: R. R. Thaman).<br />
Countless species, commonly overlooked as “weeds,” are important components<br />
of homegardens (Soemarwoto et al., 1985). Homegardeners have many uses for<br />
spontaneously propagating plants as medicines, fuel, fodder, mulch, roofing, fish<br />
poisons, toothbrushes, and food. “Weeds” such as Amaranthus spp., black<br />
nightshade (Solanum americanum), purslane (Portulaca oleracea), water spinach<br />
(Ipomoea aquatica), and fetid sea holly (Eryngium foetidum), for example, are<br />
important potherbs in Fiji and are often sold in the municipal market of Suva<br />
31
32<br />
R.R. THAMAN T<br />
ET AL.<br />
(Thaman, 1976/77), and almost all grass species are used for fodder if domestic<br />
animals are kept.<br />
1.2. Urban agroforestry gardens apart from homegardens<br />
Cultivation outside homegardens on undeveloped land (i.e., land without residences,<br />
buildings, or for other uses such as playing fields, parks, etc.) is very widespread in<br />
the Pacific island urban areas. These urban and periurban gardens also develop into<br />
agroforestry systems, and are important sources of food (including leaves, fruits, and<br />
nuts) and other products such as timber, fence posts, fuelwood, handicraft and light<br />
construction materials, medicines, and flowers (Table 2). Such areas include road<br />
frontages, empty lots, riverbanks and valleys, rights-of-way for proposed or existing<br />
paths and roads, and open land in general including hillsides and swamplands. Both<br />
subsistence and limited commercial production are attempted in these urban and<br />
periurban agroforestry gardens (Fig. 2).<br />
Figure 2. Periurban mixed planting with fruits, timber, medicinal, and staple crops on ‘Upolu<br />
island’, Samoa. Species include coconut (Cocos nucifera), breadfruit (Artocarpus altilis),<br />
poumuli ( Flueggea flexuosa), bananas and plantains ( Musa a spp.), and noni ( Morinda<br />
citrifolia). Note yam vine ( Dioscorea sp.) trellised onto breadfruit f tree on left (Photo:<br />
C. Elevitch).<br />
In the suburbs of Port Moresby, PNG, sampled in the 1970s, more than one-third<br />
of all households had “gardens” on other lands in addition to their homegardens. The
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
distinction here is in the location of these gardens with respect to homes: while<br />
homegardens are located surrounding homes, these “other gardens” are not<br />
physically close to the homes. Kilakila villagers, who were then largely original<br />
inhabitants of the area, had particularly large tracts of urban savanna lands, and all<br />
households had, in addition to their homegardens, up to four “bush” gardens<br />
averaging 1135 m 2 located on urban lands within 3 km of the center of Kilakila.<br />
With the expansion of the Port Moresby population from 124 000 in 1980 to over<br />
250 000 in 2000 (National Statistical Office of Papua New Guinea, 2000; Allen<br />
et al., 2002), such large urban gardens can no longer exist, although no detailed<br />
follow-up study has been undertaken.<br />
Open hillsides within Port Moresby support a distinctive system of agriculture<br />
based on wet season plantings dominated by sweet potato, along with cassava<br />
(Manihot esculenta), banana, taro (Colocasia and Xanthosoma spp.), hibiscus<br />
spinach ( (Abelmoschus manihot),<br />
Chinese cabbage (Brassica chinensis), corn (Zea<br />
mays), cucumber (Cucumis sativus), passionfruit (Passiflora spp.), peanut ( (Arachis<br />
hypogaea), pineapple ( (Ananas comosus),<br />
pumpkin (Cucurbita pepo), snake or long<br />
bean (Vigna unguiculata subsp. sesquipedalis), bunching onion ( (Allium spp.),<br />
sugarcane (Saccharum officinarum), tomato (Solanum lycopersicon) and watermelon<br />
(Citrullus lanatus). Tree crops include breadfruit, coconut, mandarin and<br />
sweet oranges (Citrus reticulata and C. sinensis), mango and papaya. Practiced by<br />
urban migrants who rent the gardened land from local traditional landowners, this<br />
system differs from the surrounding agriculture of rural people in that the grassland<br />
fallow period is shorter, and drains are dug across the slope, with soil heaped behind<br />
them into long beds. These gardens are even less well studied than Port Moresby’s<br />
homegardens (Allen et al., 2002), and they periodically attract public criticism in<br />
local newspapers, as a cause of erosion and smoke pollution.<br />
In Suva, Fiji, about 20% of all households surveyed in the late 1970s cultivated<br />
“unused” open lands. It has been estimated that in the 30 km 2 Suva Peninsula,<br />
approximately 5 km 2 which represents more than 70% of the area not under swamp<br />
or mangrove – is still under this type of cultivation (Thaman, 1995). Planting is done<br />
along road frontage in about 20% of all households despite the City Council<br />
regulations forbidding such practices, and the practice seems to have intensified<br />
recently in parts of Suva.<br />
In Tonga, Kiribati, and Nauru, there is little undeveloped “urban” land. However,<br />
in a number of cases, the Tongans planted entire adjacent unoccupied “town allotments”<br />
with sweet potato, taro, tannia (Xanthosoma<br />
( spp.), and a mixture of trees, or with<br />
traditional mixed yam gardens, where yams, giant taro (Cyrtosperma chamissonis),<br />
plantains (Musa spp.), and taro are intercropped, usually among or under coconuts<br />
and other trees (Thaman, 1978). There is virtually no open land in urban Kiribati,<br />
but in Nauru, some Chinese, Tuvaluan and I-Kiribati (nationals of Kiribati) contract<br />
laborers plant food gardens near the Nauru Phosphate Corporation’s workshops on<br />
the phosphate-rich central plateau and in the swampy areas surrounding landlocked<br />
Buada Lagoon. In Tuvaluan and I-Kiribati gardens, coconuts and banana clones<br />
were dominant.<br />
33
34<br />
1.3. Animal husbandry<br />
R.R. THAMAN T<br />
ET AL.<br />
Small-scale animal husbandry, although playing a minor role compared with plant<br />
food production, is also an important activity in urban and rural homegardens. In<br />
Port Moresby suburbs studied in the 1970s, animal keeping was minimal, with 11 of<br />
79 households keeping pigs, chickens, or ducks and a few households keeping<br />
tethered cows or goats. More recently, there are a few reports of urban household<br />
pigs, and of raising pigs on food wastes at the city dump (Hide, 2003), but there has<br />
been no recent detailed study. Pigs were not kept in Suva, but in Tonga over half of<br />
all sample households kept tethered or penned pigs, and almost two-thirds kept<br />
chickens or ducks. In most cases, poultry were penned or tethered at night and<br />
allowed to roam around during the day, and pigs and other larger animals were<br />
generally tethered or penned at all times. In Kiribati, Tuvalu and Nauru, pigs and<br />
chickens are also kept on home allotments. In Nauru, there was a large communal<br />
pig rearing area along the beach in Denigomodu District. In Betio, the most heavily<br />
populated area of South Tarawa, there was a large communal pig rearing area with<br />
individualized pens, established by the local town council, under coconuts,<br />
breadfruit, and other trees. In Tuvalu, pigs are kept near the main urban village<br />
along the airport runway on the seaside of the main Fogafale Islet, where they are<br />
fed with kitchen wastes and mangrove leaves. In general, homegardens in rural areas<br />
also have animals which are penned, tethered, or sometimes free ranging –<br />
particularly chickens around houses, which also serve to control cockroaches and<br />
other insects.<br />
Apart from kitchen waste, the main feed for pigs and chickens in most areas is<br />
coconut kernel. In Tonga, goats and pigs are commonly fed the leaves of Leucaena<br />
leucocephala, Pisonia grandis, and Erythrina variegata, while “living edible pens” (pens<br />
with edible living fencing) for poultry and pigs are made of these same species, plus<br />
others such as Hibiscus tiliaceus s and Polyscias s spp., all of which are easily pruned or<br />
pollarded to provide fodder. On open lands, horses, cattle, and goats are commonly<br />
tethered to trees, which also give them shade. Small animal pens that are commonly<br />
constructed of coconut logs, bamboo, Leucaena, or other local timber are also<br />
found occasionally. In rural homegardens, pigs, goats, and even cattle in Fiji, are<br />
stall-fed, or rotationally tethered to trees or fence posts where they can graze or<br />
browse.<br />
On the negative side, grazing animals and pigs seem to accelerate agrodeforestation<br />
in urban areas through browsing or trampling effects. Once established,<br />
however, trees and animals co-exist well, except where browsing goats eat the bark<br />
of trees. Cattle, in fact, seem to enhance the establishment and spread of guava<br />
(Psidium guajava), which although an important fruit, medicinal, and fuelwood<br />
source, has become a noxious pasture weed in many areas. Another serious problem<br />
related to pig keeping in urban areas is the effect of high-nutrient waste runoff on<br />
the nearby shore coral reefs. Nutrient-enriched water favors the growth of algae and<br />
phytoplankton over the growth and maintenance of coral reefs, which require clear,<br />
nutrient-poor waters. In the rural outer islands of Ha’ppai in Tonga, free-ranging<br />
pigs were seen as one of the major constraints to expanded homegardening and the<br />
planting of trees in rural villages (Thaman et al., 2001).
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
1.4. Ethnic basis of garden composition<br />
The most common plants of Pacific island homegardens tend to be traditionally<br />
important native plants or pre-European (aboriginal) introductions, except where the<br />
gardeners are from immigrant populations. For example, the Indian population of<br />
Fiji prefers species such as eggplant (Solanum melongena), okra ( (Abelmoschus<br />
esculentus), Amaranthus spp., pulses and cucurbits, and tree crops such as jackfruit<br />
(Artocarpus ( heterophyllus),<br />
tamarind (Tamarindus indicus), mango, Citrus spp.,<br />
curry leaf (Murraya koenigii), Sebesten plum (Cordia dichotoma), horseradish or<br />
drumstick tree (Moringa oleifera), and the spiritually and medicinally important<br />
neem tree ( (Azadirachta indica).<br />
Similarly in a study of 150 urban lots in Hawai’i, where the native population is<br />
small relative to the immigrant population of Japanese, Chinese, Filipino, European<br />
and North American origin, plants introduced after European contact dominated the<br />
homegardens (Ikagawa, 1994). Of the 42 genera present in more than 10% of<br />
Honolulu gardens, only two were introduced by Hawaiians, ti (Cordyline fruticosa)<br />
and Musa spp. A strong preference for ornamental landscapes and the strong moneybased<br />
economy and culture presumably explain the relative lack of edible, culinary,<br />
and medicinal plants in Hawaiian homegardens.<br />
In Port Moresby and most other PNG urban areas and plantation or mining<br />
settlements, where there are high percentages of immigrants from other areas of<br />
PNG, homegardens reflect the great diversity of species, cultivars and cultivation<br />
practices arising from the cultural and ecological diversity for which the country is<br />
famous. This diversity is evident in the gardens of settlers on the oil palm (Elaeis<br />
guineensis) projects of West New Britain (Benjamin, 1985) and Milne Bay<br />
Province. In Port Moresby, urban migrants often have preferences to traditional<br />
crops of their native habitats that may be unsuited to the local soils or climate.<br />
Examples include struggling sago palms (Metroxylon sagu), and the small potherb<br />
Rungia klossii, lovingly nurtured to coax a second crop of leaves from cuttings<br />
brought from the highlands. Similarly, Trobriand islanders, attached to the social<br />
values of their crops, have transferred competitive yam growing to Port Moresby<br />
(Battaglia, 1985).<br />
Despite the dominance of these traditional crops, there is also a great range of<br />
more recently introduced crops, such as temperate vegetables, pineapple, papaya,<br />
avocado (Persea americana), guava, and improved citrus varieties and banana<br />
clones, as well as cassava, which is a ubiquitous staple in most Pacific island towns<br />
(Thaman and Thomas, 1985). In fact, Pacific homegardens seem to have been, and<br />
will probably continue to be, one of the most effective avenues for the introduction<br />
and acceptance of new plant species. The introduction of chaya or tree spinach<br />
(Cnidoscolus chayamansa) into homegardens in urban South Tarawa and elsewhere,<br />
mentioned above, is an excellent example.<br />
1.5. Spatial arrangement of components in the homegardens<br />
There is great diversity in the spatial distribution of food crops and their area.<br />
Whereas some households have only a few scattered fruit trees and vegetables,<br />
35
36<br />
many cultivate food crops on over 50% of the total area of their property. In Port<br />
Moresby, for example, in Morata and Gerehu suburbs, recently settled in the mid-<br />
1970s, an average of approximately 40% of 450 m 2 allotments were then under food<br />
crops. Similarly, in some cases in Nuku‘alofa, up to 75% of 500 to 1000 m 2<br />
allotments were under food cultivation, primarily root crops (such as yam, taro,<br />
tannia, cassava, and sweet potato) and banana among scattered trees (Thaman,<br />
1995). Trees gradually become dominant in long-settled areas as cash incomes increase,<br />
and tree seedlings mature and increasingly shade garden areas. Nevertheless, in suburbs<br />
such as Gerehu, where trees have matured, socioeconomic status has risen. Although<br />
and the contribution to household economies that homegardens provide has<br />
declined, gardening continues to be important (Levett, 1996).<br />
Ornamentals are commonly planted closest to the home, often in front yards, as<br />
well as in containers on front porches. Medicinal plants, sacred or fragrant plants,<br />
and other culturally valuable, common multipurpose plants, are scattered amongst<br />
the food plants and ornamentals. In gardens of the indigenous Nauruans (who as a<br />
result of phosphate mining royalties, have historically had high per capita incomes),<br />
ornamental, aromatic and medicinal plants dominate, along with the ubiquitous<br />
coconut, edible pandanus, some bananas, and breadfruit. At the Location contract<br />
worker settlement in Nauru, where people live in multistory tenements, and where<br />
family gardening is limited to no more than 15 to 30 m 2 , most families have only a<br />
few plants. The gardens of Tuvaluans and I-Kiribati who live as contract workers in<br />
Nauru often consist of juvenile tree seedlings, staple root crops, or a single coconut<br />
palm or stand of bananas. In the gardens of Chinese (mostly recruited from Hong<br />
Kong) and Filipinos, the emphasis is on intensive vegetable gardening, often in<br />
containers, reflecting a more intensive system than that was practiced by most<br />
indigenous Pacific island peoples. In Kiribati and Tonga, however, recent emphasis<br />
has been placed by the government and non-governmental organizations on more<br />
intensive types of gardening: in Kiribati, using hydroponic and deep mulching<br />
techniques because of the highly infertile calcareous and sandy soils there. In<br />
Kiribati, where vitamin A deficiency-induced night blindness and xerophthalmia<br />
have become problems, the planting and consumption of the vitamin-rich leaves of<br />
two native tree species: noni (Morinda citrifolia) and Pisonia grandis, and more<br />
recently chaya have been encouraged in urban areas (Thaman, 1995).<br />
1.6. Trends toward agrodeforestation<br />
R.R. THAMAN T<br />
ET AL.<br />
Despite the current importance of gardening on open urban and periurban land, these<br />
areas have been severely affected by deforestation and agrodeforestation (Thaman,<br />
1992). Increasing population, poverty, and need for firewood, expansion of squatter<br />
settlements, lack of legislation controlling tree removal, increasing dependence on<br />
root crops such as cassava and sweet potatoes, and the loss of knowledge on the<br />
importance of trees in the context of a rapidly urbanizing Pacific have led to the<br />
increasing elimination of trees from urban landscapes throughout the islands<br />
(Thaman, 2002). In rural areas, promotion of a wide range of export cash crops (e.g.,<br />
coconut, banana, cacao [Theobroma cacao], sugarcane, coffee [Coffea spp.], ginger<br />
[Zingiber officinale], and butter pumpkin [Curcurbita maxima]) has led to the
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
clearing of diverse agroforests. This has been particularly serious in Tonga, where<br />
rapid expansion in the export of pumpkins to Japan has led to increasing use of the<br />
plow and clearance of multipurpose trees from agricultural holdings 3 . The Southeast<br />
Asian homegardens also experience a similar situation with varying degrees of<br />
commercialization (Abdoellah et al., 2006). When clearing land for short-term<br />
crops, trees in traditional agroforests used to be severely pruned or pollarded, but not<br />
killed, so they would regenerate after the crops have been harvested. However, in<br />
recent times they are commonly bulldozed, ploughed, deliberately killed with<br />
herbicides, or burned to make way for cash crops or for urban expansion.<br />
1.7. Constraints and limitations to homegardening<br />
Homegardeners face a number of problems in maintaining their traditional agroforests.<br />
These include poor soils, cost, and availability of land and water, insufficient<br />
time and labor, agricultural thefts, lack of planting materials, and lack of government<br />
assistance (Thaman, 1995). For example, drought is a major problem in Port<br />
Moresby, which has a 7-month dry season and has suffered prolonged droughts<br />
during the recent El Niño events. Gardeners must contend with the increasing<br />
unreliability of the overstretched, reticulated, water supply system and the failure of<br />
community faucets, regulations against the use of water for gardening purposes, and<br />
lack of alternative water supplies (Vasey, 1990). Restrictions on the use of water in<br />
gardens are also imposed during periods of extended droughts in Fiji. The atolls are<br />
also periodically affected by prolonged droughts, which commonly lead to the death<br />
of a significant proportion of the breadfruit, citrus, and other trees and food plants<br />
that are only marginally suited to the atoll environment 4 .<br />
Urban gardeners commonly have to contend with infertile, poor soils, such as the<br />
rocky or stony Lithosols of Port Moresby, the shallow soils that overlie a marl<br />
substrate in Suva, hydromorphic soils in low-lying areas, and the notoriously<br />
infertile, calcimorphic atoll soils of Kiribati, Tuvalu, and the Marshall Islands.<br />
Continual cropping on small urban plots also leads to declining fertility and loss of<br />
soil structure, unless ameliorative measures are taken (Thaman, 1995). Both water<br />
shortage and poor soils, however, often make trees a more attractive proposition<br />
than short-term ground crops, which require water and higher soil fertility.<br />
Insufficient land and insecurity of tenure are problems in most areas. More than<br />
half of all households in Suva, Fiji, said land shortage was a problem (Thaman,<br />
1983). Insecurity of tenure, especially in Suva, where a number of people had shortterm<br />
leases or were squatters, seems to be a major problem and a strong disincentive<br />
to homegardens and the protection of trees. City Council regulations, although not<br />
strictly upheld, have also been considered a disincentive; and have discouraged<br />
cultivation of ground crops and trees along road frontages, and the keeping of pigs,<br />
goats, cows, and horses within the city limits. Other problems for gardeners include:<br />
plant diseases, insects, snails, birds, rats, dogs, mongooses, and noxious weeds; theft<br />
of produce, especially of banana bunches and tree fruits; insufficient time; high costs<br />
of poultry feed and fertilizer; predation of firewood and deforestation on<br />
undeveloped urban and periurban lands where most low-income families still<br />
depend on firewood to cook their meals; boundary problems with respect to<br />
37
38<br />
R.R. THAMAN T<br />
ET AL.<br />
ownership of crops; and neighbors’ unfavorable response to gardening or livestock<br />
rearing (Thaman, 1995).<br />
In Port Moresby, hillside gardening has once again become the focus of<br />
criticism, on the grounds that it causes environmental damage, to the point that, in<br />
2005, the Prime Minister promised publicly a legislation to ban it (Quartermain,<br />
2005). Constraints to expanded homegardening are the greatest in Kiribati, Tuvalu,<br />
the Marshall Island, and Nauru, where extremely poor soils, limited water<br />
availability, and very high population densities, especially in South Tarawa and at<br />
Location, Nauru, are serious problems. Among the indigenous Nauruans, who are<br />
considered to be 100% urbanized, extremely high per capita incomes from<br />
phosphate royalties in the past and a resulting overdependence on imported foods<br />
seem to be the major disincentive to urban food gardening. The problem is<br />
complicated in Funafuti, where the soil from over half of the highest quality land on<br />
the main urban islet of Fogafale was excavated during World War II to build a<br />
runway, leaving only soil-less “borrow pits” of no agricultural utility.<br />
1.8. Future prospects of urban and homegarden agroforestry<br />
The importance of urban and homegarden agroforestry and its implications for<br />
planning are not clearly understood by most planners and policymakers in the<br />
Pacific islands because of a lack of quantitative data on its nature, extent, and<br />
cultural and ecological significance. There is little sign of a continuation of the<br />
interest once shown by some city planners and administrators. For example, the Port<br />
Moresby Housing Commission’s survey of urban gardening in the early 1970s and<br />
the studies by the Committee on Food Supplies of the Solomon Islands (1974) of the<br />
production of major staple crops (primarily sweet potato) in Honiara stressed the<br />
need to increase production per capita in both rural and urban areas. Fitzroy (1981)<br />
pointed out the correlation between vitamin deficiencies in “urbanized” people<br />
without garden plots in Honiara. Although further studies stressing the importance<br />
of urban and homegardens have been conducted since the mid-1970s, there is still a<br />
need for more information on the problems faced by gardeners, such as crops that do<br />
best under conditions of increasing pressure on land and deteriorating soils, best<br />
practices in terms of soil conservation and improvement, successful models for<br />
promoting urban and homegardening, and, models for the propagation and<br />
distribution of desirable cultivars of particularly useful plants.<br />
Nevertheless, there are some hopeful signs in favor of urban and homegarden<br />
agroforestry in the region. Among these are the continued efforts supporting the<br />
spread of kitchen gardening (“supsup” gardens) in Solomon Islands (Jansen et al.,<br />
2001), recognition by the National Agricultural Research Institute and other bodies<br />
in Papua New Guinea of the continuing importance of urban gardening and the need<br />
for remediation of erosion problems (Quartermain, 2005), and the international Slow<br />
Food movement 5 , which promotes the appreciation of locally-grown food, and is<br />
gaining ground in Hawai’i.
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
It has been recognized that urban and homegarden garden agroforestry could<br />
help to prevent and alleviate poverty, reduce the alarming incidence of nutritional<br />
disorders and nutrition-related, non-communicable diseases, promote greater food<br />
security, reduce dependence on inferior imported medicines, fuels, ornamentation,<br />
handicrafts and other products and address environmental problems such as coastal<br />
erosion and pollution, loss of biodiversity and urban agrodeforestation (Thaman,<br />
1988). These practices can also stem the loss of traditional ethnobiodiversity (e.g.,<br />
the uses, knowledge, beliefs, management systems, and languages; Thaman, 2004)<br />
of which trees, forests and tree-rich agroforestry systems constitutes a dominant<br />
component. Particular emphasis must be placed on the protection and enhancement<br />
of existing urban and homegarden agroforestry systems. Preserving and improving<br />
existing systems is an appropriate and cost-effective means of fostering the use of<br />
trees within the fabric of a rapidly urbanizing and homegarden-oriented Pacific<br />
island landscape.<br />
ACKNOWLEDGEMENTS<br />
We thank the countless Pacific island urban and homegarden agroforesters and<br />
gardeners, both male and female, who have shared knowledge of their plants and<br />
technologies and allowed us to spend time with them in their agroforests over the<br />
past decades. Many thanks also to Robin Hide for his generous help in writing this<br />
article.<br />
ENDNOTES<br />
1. Bourke R.M., Allen M.G. and Salisbury J.G. 2001. Food security for Papua<br />
New Guinea. Proceedings of the Papua New Guinea Food and Nutrition 2000<br />
Conference, ACIAR Proceedings 99. ACIAR, Canberra, Australia, 892p.<br />
2. Kennedy J. and Clarke W.C. 2004. Cultivated landscapes of the southwest<br />
Pacific. RMAP Working Paper 50. Resource Management in Asia–Pacific<br />
Program, Research School of Pacific and Asian Studies, Australian National<br />
University, 47p.<br />
3. Thaman R.R. 2004. Akau, A poto mo e ofa fonua: Ko e makatu unga<br />
,<br />
tukufakaholo (Trees, arboreal diversity and ethnobiodiversity: An agroforestry<br />
action plan for sustainable development in the Kingdom of Tonga). Technical<br />
Report 2004/04. Institute of Applied Science, University of the South Pacific,<br />
Suva, Fiji, 128p.<br />
4. Thaman R.R. 2004. Cool spots under threat: The conservation status of atoll<br />
biodiversity and ethno-biodiversity in the Pacific Islands. In: Lee K.J. and Tsai<br />
H.-M. (eds), Changing islands–changing worlds: Proceedings of Islands of the<br />
World VIII, pp 60–64. International Small Islands Studies Association (ISISA),<br />
Sydney, Australia.<br />
5. last accessed: January 2006.<br />
,<br />
,<br />
,<br />
39<br />
o e
40<br />
R.R. THAMAN T ET AL.<br />
REFERENCES<br />
Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo and Parikesit, 2006. Commercialization<br />
of homegardens in an Indonesian village: vegetation composition and functional<br />
changes. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 233 – 250. Springer Science, Dordrecht.<br />
Allen B.J., Nen T., Bourke R.M., Hide R.L., Fritsch D., Grau R., Hobsbawn P, and Lyon S.<br />
2002. Central Province: Text Summaries, maps, code lists and village identification.<br />
Agricultural Systems of Papua New Guinea. Working Paper 15. Revised edition.<br />
Department of Human Geography, Research School of Pacific and Asian Studies, The<br />
Australian National University, Canberra, Australia, 152p.<br />
Battaglia D. 1985. Bringing home to Moresby: Urban gardening and ethnic pride among<br />
Trobriand Islanders in the national capital. IASER Special Publication 11. Institute of<br />
Applied Social and Economic Research, Port Moresby, Papua New Guinea, 53p.<br />
Benjamin C. 1985. Some food market influences of a large-scale smallholder development in<br />
the West New Britain area of Papua New Guinea. Papua New Guinea J Agric Forest<br />
Fisher 33(3–4): 133 –141.<br />
Committee on Food Supplies (BSIP). 1974. Food and self-reliance: report of the Committee<br />
on Food Supplies. Government House, Honiara, Solomon Islands, 68p.<br />
Coyne T. 2000. Lifestyle diseases in Pacific communities. Technical Paper 219. Secretariat of<br />
the Pacific Community, Noumea, New Caledonia, 331p.<br />
Curtis M. 2004. The obesity epidemic in the Pacific Islands. J Dev Social Transfor 1: 37 – 42.<br />
Falanruw M.C. 1995. The Yapese agricultural system. PhD thesis. University of the South<br />
Pacific, Suva, Fiji, 386p.<br />
Fitzroy G.J. 1981. Commentary: Influence of development factors on nutritional patterns in<br />
the Solomon Islands. Ecol Food Nutr 10: 87 – 119.<br />
Hide R. 2003. Pig husbandry in New Guinea. A literature review and bibliography. ACIAR<br />
Monograph 108. ACIAR, Canberra, Australia, 291p.<br />
Ikagawa T. 1994. Residential gardens in urban Honolulu, Hawaii: neighborhood, ethnicity<br />
and ornamental plants. PhD thesis. University of Hawai i at Manoa, Honolulu, 208p.<br />
Jansen T., Kotali C. and Pitavavini G. 2001. Improving household food security in Lauru,<br />
Solomon Islands, through grass roots extension, kitchen gardens and nutrition education.<br />
In: Bourke R.M., Allen M.G., and Salisbury J.G. (eds), Food security for Papua New<br />
Guinea. Proceedings of the Papua New Guinea Food and Nutrition 2000 Conference, pp<br />
509–515. ACIAR Proceedings 99, ACIAR, Canberra, Australia.<br />
Lamanda N., Malézieux E. and Martin P. 2006. Structure and dynamics of coconut-based<br />
agroforestry systems in Melanesia: a case study from Vanuatu archipelago. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 105 – 121. Springer Science, Dordrecht.<br />
Levett M.P. 1992. Urban gardening in Port Moresby: A survey of the suburb of Gerehu. Yagl-<br />
Ambu, Papua New Guinea J Soc Sci Hum 16(3): 47 – 68.<br />
Levett M.P. 1996. Fresh food production and marketing: national and Port Moresby<br />
perspectives. In: Gladman J., Mowbray D., and Duguman J. (eds), From Rio to Rai:<br />
Environment and development in Papua New Guinea up to 2000 and beyond: A quarter of<br />
next to nothing. Papers from the 20th Waigani Seminar Vol. 3, University of Papua New<br />
Guinea, Port Moresby, 302p.<br />
Levett M.P. and Uvano M. 1992. Urban gardening in Port Moresby: A survey of the suburbs<br />
of Morata and Waigani. Yagl-Ambu, Papua New Guinea J Soc Sci Hum 16(3): 69 – 91.<br />
National Statistical Office of Papua New Guinea. http://www.nso.gov.pg/2000_Census (last<br />
accessed: January 2006).
URBAN U AND HOMEGARDEN AGROFORESTRY IN THE PACIFIC P ISLANDS I<br />
Parkinson S.V. 1982. Nutrition in the south Pacific—past and present. J Food Nutr 39(3):<br />
121 –125.<br />
Quartermain A. 2005. Food for thought. Nari Nius, Newsletter of the Papua New Guinea<br />
National Agric Res Inst 8(1): 9.<br />
Soemarwoto O.M., Karyono I., Soekar, Tadiredia W.M. and Raman A. 1985. The Javanese<br />
homegarden as an integrated agro-ecosystem. Food Nutr. Bull. 7(3): 44 – 47.<br />
Thaman R.R. 1976/77. Plant resources of the Suva Municipal Market, Fiji. Ethnomedicine IV<br />
1/2 (1976/77): 23 – 62.<br />
Thaman R.R. 1978. Cooperative yam gardens: An adaptation of a traditional agricultural<br />
system to serve the needs of the developing Tongan market economy. In: Fisk E.K. (ed.),<br />
The adaptation of traditional agriculture, pp 116–130. Development Studies Center<br />
Monograph No. 11. Australian National University, Canberra.<br />
Thaman R.R. 1982. Deterioration of traditional food systems, increasing malnutrition and<br />
food dependency in the Pacific Islands. J Food Nutr 39(3): 109 – 121.<br />
Thaman R.R. 1983. Urban gardening in Fiji: A direct means for sustainable national<br />
development. Proc Fiji Soc 13: 1 – 27.<br />
Thaman R.R. 1987. Urban agroforestry: The Pacific islands and beyond. Unasylva 39(55):<br />
2 – 13.<br />
Thaman R.R. 1988. Environmental issues in the Pacific Islands: Constraints to sustainable<br />
island development. Pacific Issues 1, pp 1 – 77. Pacific Circle Consortium, Woden,<br />
Canberra, Australia.<br />
Thaman R.R. 1992. Agrodeforestation as a major threat to sustainable development. In:<br />
Thistlethwaite R. and Votaw G. (eds), Environment and development: A Pacific Island<br />
perspective, pp 194–195. Asian Development Bank, Manila and South Pacific Regional<br />
Environment Programme, Apia, Samoa.<br />
Thaman R.R. 1995. Urban food gardening in the Pacific Islands: A basis for food security in<br />
rapidly urbanising small-island states. Habitat Int 19: 209 – 224.<br />
Thaman R.R. 2002. Trees outside forests as a foundation for sustainable development in the<br />
small island developing states of the Pacific Ocean. Int For Rev 4: 268 – 276.<br />
Thaman R.R. 2004. Sustaining culture and biodiversity in Pacific Islands with local and<br />
indigenous knowledge. Pac Ecologist (7&8): 43 – 48.<br />
Thaman R.R. and Clarke W.C. 1993. Pacific island agroforestry: Functional and utilitarian<br />
diversity. In: Clarke W.C. and Thaman R.R. (eds), Pacific Island agroforestry: Systems<br />
for sustainability, pp 17–33. United Nations University Press, Tokyo.<br />
Thaman R.R. and Thomas P.M. 1985. Cassava and change in Pacific Island food systems. In:<br />
Cattle D.J. and Schwerin K.H. (eds), Food energy in tropical ecosystems, pp 191–228.<br />
Gordon & Breach, New York.<br />
Thaman R.R., Eritaia B. and Faka’ osi S. 2001. Community-based biodiversity surveys and<br />
conservation action plans as tools for nature conservation in the Pacific Islands: Lessons<br />
learned from Fiji, Tonga and Kiribati (Melanesia, Polynesia and Micronesia). In: Miller S.<br />
and Sim J. (eds), Tools for conservation: 6th South Pacific Conference on Nature<br />
Conservation & Protected Areas (29 September – 3 October 1997), Pohnpei, Federated<br />
States of Micronesia. Volume 3: Conference papers, pp 225 – 285. South Pacific Regional<br />
Environment Programme, Apia, Samoa.<br />
Vasey D.E. 1990. On estimating the net social and economic value of urban home gardens.<br />
In: Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 203–213. United Nations<br />
University Press, Tokyo.<br />
41
CHAPTER 4<br />
AMAZONIAN HOMEGARDENS:<br />
THEIR ETHNOHISTORY AND POTENTIAL<br />
CONTRIBUTION TO AGROFORESTRY<br />
DEVELOPMENT<br />
R.P. MILLER 1 , J.W. PENN, JR. 2 , AND J. VAN LEEUWEN 3<br />
1 Instituto Olhar Etnográfico, SHIN CA 5 Conj. J Bl. B, Sala 105, Brasília-DF<br />
71505, Brazil; E-mail: . 2 Grand Valley State<br />
University, 1155 Au Sable Hall, Allendale, MI, 49401, USA. 3 Instituto Nacional de<br />
Pesquisas da Amazônia – INPA, Manaus, Amazonas, Brazil<br />
Keywords: Caboclo, Fruit tree domestication, Indigenous knowledge, Ribereño.<br />
Abstract. This chapter reviews how homegardens and a number of other traditional<br />
agricultural practices survived the aftermath of European conquest of Amazonia. The<br />
historical development of homegardens in Amazonia began with the evolution of agriculture<br />
and domestication of trees in prehistoric times, followed by the development of cultural<br />
complexes along the Amazon River and its main tributaries. These traditional societies,<br />
characterized by rich material culture and well-developed agricultural systems, were<br />
decimated by the combination of epidemics, wars and slavery that accompanied the European<br />
conquest. Yet, the homegardens survived in Amazonia, and today they represent the<br />
reorganization of the original indigenous practices within the context of the upheaval and<br />
changes brought by colonization and market economies, including the incorporation of<br />
introduced Asian fruit trees. Although homegardens near urban centers may provide income,<br />
in rural areas they are important chiefly for household subsistence. They are often the focus of<br />
experimentation with new tree species and cultivation techniques, and thus have the potential<br />
to contribute to the development of other agroforestry systems, and to extension efforts that<br />
seek alternatives for agricultural development in Amazonia.<br />
1. INTRODUCTION<br />
The local and regional diversity of Amazonian homegardens is best understood by<br />
studying their origins and how they have been influenced by the socioeconomic and<br />
43<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 43–60.<br />
© 2006 Springer. Printed in the Netherlands.
44 R.P. MMILLER<br />
ET AL.<br />
cultural forces that have shaped social organization and subsistence practices in the<br />
region, from prehistoric times to the present. This historical development begins<br />
with the evolution of agriculture and the domestication of trees in prehistoric times,<br />
followed by the emergence of complex cultures or chiefdoms along the main rivers,<br />
described by the first European explorers as exhibiting elaborate material culture and<br />
agricultural systems (Carvajal, 1542; Acuña, 1639). Although European conquest subsequently<br />
decimated these societies through a combination of epidemics, wars and<br />
slavery, as this chapter will show, a number of their agricultural practices, including<br />
homegardens, survived.<br />
The traditional l (i.e., prior to any interventions by research/extension agencies)<br />
homegardens of Amazonia represent a dynamic equilibrium of these original<br />
indigenous practices with the new social order and scenario created by the process<br />
of colonization. Included in this process was the incorporation of many Asian fruit<br />
trees introduced by the Europeans. The culture of traditional river-edge inhabitants,<br />
known as caboclos (in Brazil) or ribereños (in Peru) represents the fusion and<br />
synthesis resulting from this historical process, and homegardens today are an<br />
integral part of life throughout Amazonia.<br />
Some of these homegardens and their ethnoecology have been formally<br />
described in many scientific f publications (Denevan and Padoch, 1987; Padoch and<br />
de Jong, 1991; Smith, 1996; 1999; Coomes and Burt, 1997; Lamont et al., 1999;<br />
Denevan, 2002; Coomes and Ban, 2004), including some dissertations (Bahri,<br />
1992), Annals of the Brazilian Agroforestry Congresses, and other such records<br />
(e.g., Miller, 1994; van Leeuwen and Gomes, 1995; Rosa et al., 1998a; 1998b;<br />
1998c). Although a portion of this literature limits its scope to descriptions or lists of<br />
species found in the homegardens, some of these evaluate the factors determining<br />
choice of species, their management, and how proximity of markets influence these<br />
(e.g., Lamont et al., 1999). Based on this body of literature, and the personal<br />
experience of the authors in Amazonia, this chapter will attempt to reach some<br />
general conclusions as to the historical and cultural importance of homegardens, and<br />
how this can be linked to the underlying processes of the relationship between<br />
humans and cultivated trees. An understanding of this relationship is essential for<br />
evaluating the potential contribution of homegardens to extension efforts that seek<br />
alternatives for agricultural development in Amazonia, and some suggestions will be<br />
made along this line.<br />
2. ETHNOHISTORY OF HOMEGARDENS IN AMAZONIA<br />
2.1. Pre-historical development of agriculture and homegardens in Amazonia<br />
Archeological evidence from the lowland neotropics in Colombia, Ecuador, Peru,<br />
and Mesoamerica indicates that between 10 000 and 8600 b.p. (before present)<br />
horticulture emphasizing both native tubers and seed plants was taking place outside<br />
Amazonia (Piperno and Pearsall, 1998; Piperno et al., 2000; Smith, 2001). However,<br />
in a site in Rondônia, in western Brazilian Amazonia, where human occupation by<br />
hunter-gatherers dates back to 9000 b.p., vestiges of agricultural activity, in the form
AMAZONIAN HOMEGARDENS<br />
of processing utensils, only begin to appear around 4500 b.p. (Miller, 1992). Lathrap<br />
(1977) argues that the earliest agriculture in Amazonia was probably adjacent to<br />
dwellings, along or near rivers in forests that did not require frequent clearing. At<br />
some moment, native fruit trees were domesticated and incorporated into these<br />
prehistoric agricultural systems. This process may have occurred initially through<br />
the ‘dump heap’ (sensu Anderson, 1952) or incidental route to domestication, when<br />
seeds of edible fruits collected in the forest were discarded near dwellings. Although<br />
little information is available on the sequence of domestication for neotropical tree<br />
crops, it is likely that this was concurrent with the domestication of root-crops, as<br />
the maintenance of gardens near dwellings would have provided an ideal location<br />
for the discarded seeds of useful tree species to germinate and grow. The recognition<br />
and management of such 'volunteers' would have been the first step along the road to<br />
their domestication.<br />
By 3000 to 2000 b.p., agricultural development made possible the existence of<br />
larger villages of many hectares on the middle and lower Orinoco River in<br />
Venezuela, and by 2000 years ago, large, socially stratified chiefdoms were thriving<br />
along the principal rivers of Amazonia. There is evidence of crop domestication and<br />
diffusion from this period of Amazonian history. For example, Salick (1992) has<br />
found that the domestication and exchange of cocona (Solanum sessiliflorum),<br />
common to Western Amazonian homegardens today, may have begun as long as<br />
2000 years before present. When the first European explorers arrived in Amazonia<br />
in the 16th century, large population complexes, exhibiting an elaborate material<br />
culture and ceremonial art, occupied the margins of the main rivers, with links to<br />
surrounding regions through extensive trade networks (Roosevelt, 1994). From the<br />
description by Jesuit friar Gaspar de Carvajal, in his account of the first European<br />
exploration of Amazon in 1541–`42, we know that part of this cultural development<br />
consisted of agricultural systems based on a great variety of cultivated plants,<br />
including fruit trees, and the storage of various foods such as cassava (Manihot<br />
esculenta), maize (Zea mays), dried fish, and penned river turtles (Carvajal, 1542).<br />
Although the existence of some sort of homegarden is clear in these historical<br />
accounts, little detail is provided on the nature of these indigenous agroforestry<br />
systems. Carvajal, for example, mentions only that “much fruit of all kinds” was<br />
found in one village, and that fruit trees were planted on either sides of the road<br />
leading to another village (Carvajal, 1542). In all, at least 138 species of plants are<br />
thought to have been under cultivation or management at the time of European<br />
arrival in Amazonia, of which 68% were trees or woody perennials (Clement,<br />
1999a). Besides the species mentioned in historical accounts, it is possible that in<br />
pre-Columbian times many more species were also cultivated, or were in a state of<br />
incipient domestication. A number of commonly cultivated Amazonian fruit trees<br />
have the characteristics of long periods of selection and genetic improvement.<br />
Clement (1989; 1999b) suggests the existence of a pre-Columbian center of crop<br />
diversity in Western Amazonia, based on the genetic diversity of fruit tree<br />
domesticates. In terms of their manipulation of plant resources, pre-Columbian<br />
cultures in Amazonia appear to have operated along a gradient of domestication,<br />
with plants fully domesticated and reliant on human care for their dispersal and<br />
survival at one extreme, as is the case of the peach palm (Bactris gasipaes). At the<br />
45
46<br />
R.P. MMILLER<br />
ET AL.<br />
other extreme of this gradient were those wild plants that may be found in greater<br />
than normal concentrations around ancient village sites, as a result of agricultural<br />
clearing and burning, with the possible favoring of their regeneration, but which do<br />
not exhibit any apparent genetic differentiation from their wild counterparts.<br />
Between these extremes are found a number of interesting and useful plants,<br />
suggesting that an active process of genetic selection and domestication was taking<br />
place in pre-Columbian Amazonia. An example of how this process may have<br />
occurred (and continues to occur) is described by Schroth et al. (2004), for the palm<br />
Astrocaryum tucuma in the Manaus region. Nevertheless, for the most part, the<br />
continuing domestication of wild species was truncated by the European conquest.<br />
In less than 200 years after the events described in Carvajal’s report (Carvajal,<br />
1542), the great chiefdoms along the Amazon had succumbed to epidemics of<br />
imported diseases such as smallpox and measles, wars, and enslavement. Their<br />
sophisticated culture and political and trade networks collapsed, and large stretches<br />
of the Amazon River and its tributaries were totally deserted (Daniel, 1776).<br />
Despite the decimation of native Amazonian populations that occurred during<br />
European conquest, with an ensuing loss of agrobiodiversity, many elements of their<br />
agricultural and agroforestry systems survived and can be seen among the modern<br />
tribal groups. The agroforestry practices of some of the tribal peoples in Amazonia,<br />
reviewed in Miller and Nair (2006), range from the cultivation of fruit trees and<br />
other useful plants around dwellings (homegardens), to the incorporation of trees<br />
in agricultural fields and fallows, which may involve practices such as actively<br />
planting or managing useful tree species or sparing seedlings that regenerate<br />
naturally. The homegarden of fruit trees, condiments and medicinal plants may<br />
grade into a belt of fruit trees surrounding a village, fruit trees interspersed with field<br />
crops, orchards of mixed fruit trees, and fallows of forest species enriched with fruit<br />
trees – these last mentioned configurations having been termed “swidden-fallow<br />
agroforestry” (Denevan and Padoch, 1987; Denevan, 2002). Although there are<br />
exceptions, as in the case of tribes with a very rudimentary agriculture, for the most<br />
part, homegardens can be considered as an important component of the subsistence<br />
technologies and cultural knowledge of Amazonian tribes.<br />
Whether the specific cultivation methods employed by contemporary indigenous<br />
groups are the same as those of their pre-colonial ancestors is a difficult question<br />
to answer. Nevertheless, it is probable that the agroforestry systems practiced by<br />
indigenous peoples as well as the caboclos and ribereños are direct descendants<br />
of the systems in existence prior to European arrival, with the addition of a number<br />
of exotic species of fruit trees. This contribution of exotic species introduced by<br />
Europeans is discussed in the context of the ethnohistory of caboclo and ribereño<br />
culture, the subject of the following section.<br />
2.2. Ethnohistory of caboclo and ribereño culture and homegardens in Amazonia<br />
Although the use of the term caboclo has been criticized due to its negative social<br />
connotations (Lima, 1999), it is difficult to substitute, as it encompasses both<br />
colloquial as well as academic meanings in Brazil, and is a broad descriptor of a<br />
regional form of life and natural resource use. While modern-day tribal groups of
AMAZONIAN HOMEGARDENS<br />
Amazonia in most cases represent the fragments of populations and cultures that<br />
escaped to survive and regroup following the colonial holocaust, caboclo society in<br />
Brazil or ribereño society in Peru and their cultures are the result of the fusion of the<br />
remnants of the native populations, decimated during colonization, with European<br />
and African racial and cultural elements (Padoch and Pinedo-Vasquez, 2001;<br />
Ribeiro, 1997). In this process, agricultural, social, economic, and belief systems<br />
were reconfigured and reconstructed upon an existing knowledge base of ecological<br />
systems and subsistence practices, with the addition of new tools and technologies.<br />
Key players in this process were the Catholic missionaries in Amazonia. As allies to<br />
the colonial economic system, they had a major role in providing an ideology for<br />
the domination of the native populations and their transformation into a labor force.<br />
Along with the forts, missions were fundamental elements in guaranteeing the<br />
domination of the region by the Portuguese from 1650 – 1750, and allowing<br />
the functioning of commerce (Alves-Filho et al., 2005).<br />
Despite the superiority of Portuguese armaments, the native peoples did not<br />
submit easily to Portuguese attempts to enslave or otherwise conscript them as<br />
agricultural workers growing subsistence and commercial crops, collectors of forest<br />
products (such as cacao, Theobroma cacao), in the construction of public works, and<br />
other forms of labor, without which the colonial economy in Brazil would have<br />
collapsed (Alves-Filho et al., 2005). In response, they waged war, rebelled in<br />
villages and missions, deserted from royal services, massacred when possible their<br />
enemies, and even made peace treaties when convenient (Santos, 2002). Elsewhere<br />
in Amazonia, natives also put up fierce resistance, lasting well into the republican<br />
period of the former Spanish colonies, especially in Peru and Colombia (San<br />
Ramon, 1994; Stanfield, 1998; Rios, 2001).<br />
The search for cacao using Indian labor, primarily from stands of wild or feral<br />
trees, motivated the Portuguese to range far upriver, leading Portuguese incursions<br />
west into Spanish territory (now Peru) to kidnap Indians on the Marañon River<br />
during 1686 – 1723 (Edmundson, 1922). By 1730, cacao had become the region’s<br />
dominant export, remaining so for more than a century (Alden, 1976; Hemming,<br />
1987). Cacao gathering expeditions had ceased by 1750 and cacao was being<br />
cultivated in plantations along the Amazon. Farmers grew seedlings on raised beds<br />
for a year, and then transplanted them into their cassava fields, where banana plants<br />
(Musa sp.) had been previously planted to provide shade. Native fruit trees, along<br />
with introduced species, such as orange (Citrus sinensis) and avocado (Persea<br />
americana), were also interplanted with cacao, as it was known that cacao produced<br />
better in shade (Daniel, 1776). Cacao appears to have been an important, if not the<br />
principal, economic element of the agroforestry systems of that time. By the mid-<br />
1800s, another exotic species, coffee (Coffea arabica), was one of the main<br />
agricultural exports of the region, along with cotton (Gossypium sp.), cacao, guaraná<br />
(Paulinia cupana), and tobacco (Nicotiana tabacum) (Amazonas, 1852).<br />
By 1875, the rising demand for rubber, an important material for the Industrial<br />
Revolution, led to an economic boom in Amazonia. Rubber, extracted from the<br />
forest tree Hevea brasiliensis, had by 1880 become the third most important export<br />
in Brazil and Peru (Stanfield, 1998; Homma, 2003). The caboclo population,<br />
concentrated on the Amazon and Solimões Rivers, spread out through the entire<br />
47
48<br />
R.P. MMILLER<br />
ET AL.<br />
basin in search of rubber trees. A mixture of caboclo, mestizo, European, and<br />
indigenous (tribal) gatherers tapped the forests of Peru, Colombia, and Bolivia; and<br />
Manaus, Belém, and Iquitos grew into the principal commerce centers along the<br />
Amazon River. The boom attracted many migrants as well as absorbing the local<br />
labor force, with the result that agricultural production in Amazonia dropped sharply<br />
(Ribeiro, 1997; Stanfield, 1998). The rubber boom also brought disastrous<br />
consequences to the remaining forest tribes, as rubber tappers penetrated even the<br />
most distant headwaters. The atrocities committed against the Indians and their<br />
conscription as forced labor were so widespread that they attracted international<br />
attention (Renard-Casevitz, 1992; Stanfield, 1998). With the drop in agricultural<br />
production, food prices soared. Tribal societies involved in the trade could do little<br />
farming, suffered from severe hunger, and often lost their lands to rubber tappers<br />
(Stanfield, 1998). Where they survived, homegardens undoubtedly played a key role<br />
in providing food for rural inhabitants, regardless of their ethnicity.<br />
The crash in rubber prices returned Amazonia to the state of an economic<br />
backwater by the end of the First World War (Homma, 2003). Indigenous knowledge,<br />
so important to the European and mestizo efforts to cultivate and exploit the<br />
most economically lucrative resources of the region, lay dying in the form of<br />
abandoned fields across the wide swaths of Amazon basin. According to Denevan<br />
(2002), homegardens in Amazonia became less important and poorly developed<br />
after the arrival of Europeans, mostly because indigenous villages changed their<br />
locations much more frequently than they did in the past, yet another consequence of<br />
this tragic history.<br />
2.3. Transformation of traditional agriculture during colonial times<br />
Although the Portuguese introduced a number of new crops to Amazonia, such as<br />
sugarcane (Saccharum officinarum), indigo (Indigofera indica), and rice (Oryza<br />
sativa), as well as domestic animals, indigenous agricultural practices remained the<br />
basis for subsistence, and they were also adapted for the production of commercial<br />
crops such as cacao. At the same time that technology guaranteed Portuguese<br />
military superiority, agricultural technology in the form of steel tools resulted in the<br />
transformation of indigenous practices, with stone axes and digging sticks being<br />
substituted by steel axes, machetes, hoes and brush hooks. Where previously large<br />
trees were ringed with stone axes and left to dry slowly, and saplings were<br />
bludgeoned over (Daniel, 1776), steel tools greatly reduced the labor expended in<br />
agricultural clearing, with the result that what is considered today as “slash-andburn”<br />
agriculture probably is quite different from what was practiced in pre-<br />
European Amazonia. Pre-Columbian agriculture most likely had greater affinity<br />
with slash-mulch systems, as fires used to prepare fields would have been much less<br />
intense, and ringed trees would slowly drop a layer of leaves over the field. The<br />
initial difficulty in opening fields out of forest probably led to a longer use of<br />
cleared areas, through complex polycultures and crop sequences, including trees.<br />
A more extended use of fields may have been possible due to the input of organic<br />
matter from the slowly dying original vegetation.
AMAZONIAN HOMEGARDENS<br />
Catholic missions were in part responsible for the introduction of new<br />
technologies and agricultural practices. The Jesuit missions in particular were<br />
generally well-managed enterprises that exported a part of their production.<br />
Persuading natives to leave their villages and move to these missions involved a<br />
number of strategies, besides force, including convincing them that epidemics of<br />
introduced European diseases were caused by the insalubrities of their village sites.<br />
In some cases, life in a mission was the only alternative to being attacked and<br />
enslaved by colonists.<br />
Life in the missions brought together individuals of separate tribes, with different<br />
languages and cultures, for the compulsory adoption of the body of beliefs and<br />
customs of the colonizer. The cultural result was a patchwork of beliefs, the<br />
syncretism of shamanism with a vague observance of Catholic saints and holidays,<br />
the base for a “folk Catholicism,” incorporating various native practices and beliefs<br />
and the colonial influences of the Portuguese, as well as African slaves (Ribeiro,<br />
1997; Maués, 2001). Some of these beliefs are associated with a variety of<br />
magical/medicinal plants (e.g., pião roxo, Jatropha gossypiifolia) often cultivated in<br />
modern homegardens, and which along with ornamentals, are often seen even in<br />
diminutive front yards in cities such as Manaus.<br />
A characteristic of European colonization of Amazonia was the introduction of a<br />
number of exotic fruit trees. In 1662, Mauricio Heriarte (in Huber, 1904) described<br />
Belém as cheerful and full of fruit trees such as oranges, limes (Citrus aurantifolia),<br />
sweet limes (Citrus limetta) and biribás (Rollinia mucosa). The introduction of<br />
mango (Mangifera indica) to Belém in 1780 is credited to the Genovese architect<br />
Antonio Landi, who brought seeds from Bahia, the capital of Brazil until 1763. The<br />
Portuguese Crown officially sponsored a number of plant introductions from its<br />
eastern colonies of Goa (India) and Macau (China) and the establishment of a<br />
botanical garden in Belém (Dean, 1995). In 1808, in retaliation for the invasion of<br />
Portugal by France, the Portuguese invaded French Guiana and were able to take<br />
advantage of the collection of useful plants cultivated in Cayenne’s botanical<br />
garden. By the time Cayenne was returned to the French in 1818, a number of<br />
tropical species had been sent to Belém, along with unspecified European fruit<br />
trees that had been acclimated in Cayenne (Holanda, 1965). Coffee was another<br />
introduced tree crop that soon proved lucrative for Brazil by the 1800s. Coffee<br />
germplasm was introduced to Belém in 1727 by Sargeant-Major Francisco de Mello<br />
Palheta, who transported five coffee seedlings and a handful of seeds from Cayenne.<br />
The first sample of coffee grown in Pará was sent to Lisbon in 1732, and two years<br />
later in 1734, 45 tons were shipped (Homma, 2003).<br />
By the mid-19th century, exotic fruit trees had been fully incorporated into<br />
homegardens along the Amazon. Traveling on the Amazon between Óbidos and<br />
Manaus in 1849, the British naturalist Henry Walter Bates described homegardens<br />
with banana, papaya (Carica papaya), mango, orange, lemon (Citrus sp.), guava<br />
(Psidium guajava), avocado (Persea americana), abiu (Pouteria caimito), genipap<br />
(Genipa americana), and biribá, as well as coffee shrubs growing under the shade of<br />
the fruit trees (Bates, 1863). Ten years later, French traveler Robert Avé-Lallemant<br />
recorded a variety of fruit trees growing near houses on the outskirts of Belém:<br />
banana, mango, jackfruit (Artocarpus ( heterophyllus),<br />
various Annonaceae, orange<br />
49
50<br />
R.P. MMILLER<br />
ET AL.<br />
trees, coffee, as well as the giant granadilla or maracujá-açu (Passiflora quadrangularis).<br />
Surrounding the dwellings of Indians near Cametá, Pará, he found native<br />
calabash trees (Crescentia cujete) and orange trees competing with mango, and the<br />
native açaí (Euterpe oleracea) and bacaba (Oenocarpus bacaba) palms. The<br />
presence of various Annonaceae, the bacuri (Platonia insignis) and brazilnut<br />
(Bertholletia excelsa) trees was also noted. Besides the homegarden, other tree<br />
species were cultivated as commercial crops, and income sources for these households<br />
came from “extensive stands of cacao” and rubber trees. Continuing up the<br />
Amazon to Santarém, he found many cacao and orange groves, as well as<br />
concentrations of the native tucumã palm ( (Astrocaryum vulgare),<br />
highly appreciated<br />
for the edible mesocarp of its fruits (Avé-Lallemant, 1859).<br />
In Peru, coffee, mango and avocado germplasm entered the Amazon Basin from<br />
both the east and west. Avocado entered Peru and the Peruvian Amazon well before<br />
the arrival of the Spaniards, while coffee and mango cultivars in Amazonia were<br />
introduced from either direction. Accounts from early explorers suggest most mango<br />
germplasm came from coastal Peru. Besides Asian species, the Spaniards also<br />
brought plant species from and via Central America and the Caribbean. Thus, we<br />
might expect common crops of the colonial era such as bananas, beans (Phaseolus<br />
vulgaris), citrus, or sugarcane in the Peruvian Amazon to have diverse origins even<br />
soon after their introduction to the region. Explorers such as Eduard Poeppig, who<br />
studied the upper Amazon in 1829-31, have found that much of the cassava<br />
germplasm in Peru came from downriver in Brazil, while banana germplasm as far<br />
downriver as Manaus, Brazil, often came from Peru (Poeppig, 2003).<br />
By no means, however, was the cultivation of trees limited to the traditional<br />
pattern of homegardens or commodity crops. Some homegarden species were<br />
creatively adapted to other uses, as is the case of the yellow mombin (Spondias<br />
mombin; Smith, 1999) and the calabash tree for live fences in the várzea (floodplain)<br />
region. Similarly, other species that were not previously cultivated, such as the<br />
munguba (Pseudobombax munguba), a common tree of the várzea, were enrolled to<br />
mark property boundaries on floodplain ranches. Species such as the rubber tree<br />
were added as economic elements, as a small rubber boom during World War II led<br />
to a renewed interest in this crop, and a low level of tapping continued even after<br />
the war.<br />
2.4. The caboclo and ribereño in the regional economy<br />
While colonization caused the demise and/or slow absorption of the indigenous<br />
tribal populations, a new hybrid society of non-tribal peoples was on the rise. The<br />
caboclos of Brazilian Amazonia are of mixed descent, as well as the remnants of the<br />
acculturated tribes. Similarly, the ribereños in Peru are of mixed European and<br />
Amerindian descent. Despite the persistent use of the term in the literature, these<br />
rural inhabitants do not actually call themselves “ribereños.” They most often refer<br />
to themselves in occupational or class terms such as pescador r (fisherman) or<br />
chacarero, as chacra is the common name for the plots of land they farm (Penn,<br />
2004). Researchers point to the Cocama-Cocamilla tribal origins of ribereños in<br />
Peru, but ribereños have diverse origins, and it is not advisable to generalize about
AMAZONIAN HOMEGARDENS<br />
their ethnicity. The origin and ethnicity of the Cocama-Cocamilla themselves is still<br />
poorly understood (Cabral, 1995).<br />
Although very similar to the original native populations in terms of their<br />
ecological adaptations and subsistence practices, the caboclos in Brazil were very<br />
different socially (Ribeiro, 1997). Historically, they have been embedded in an<br />
agricultural and extractive economy, trading raw materials and products collected<br />
from the forests and rivers, or grown in their fields, for the manufactured items and<br />
tools necessary for their subsistence. For the most part, there was an ample supply<br />
of land for the harvest of extractive products and for fields, under communal tenure<br />
or belonging to absentee owners and defunct rubber estates. In recent decades,<br />
however, this situation has changed as development of a different form has reached<br />
Amazonia, with the construction of roads shifting the economic axes away from rivers<br />
and floodplains to the terra firme, where human occupation has been characterized by a<br />
moving frontier of logging, ranching, and agricultural colonization, that leaves in its<br />
wake a landscape dominated by pasture and to a lesser extent swidden agriculture.<br />
As rights to land have become more disputed, homegardens have taken on another<br />
socioeconomic function, with the presence of cultivated trees used as proof of land<br />
tenure and property rights.<br />
3. HOMEGARDENS IN PRESENT-DAY CABOCLO AND RIBEREÑO<br />
SOCIETIES<br />
<strong>Homegardens</strong> in Amazonia are variously referred to in folk denomination as<br />
“huertos” or “jardíns” (in Peru), and “quintais” (yards) or “sítios” (homesteads)<br />
in Brazil, as well as “pomares caseiros” (home orchards) or “miscelânea” by researchers.<br />
They combine native species with fruit trees introduced from other parts of<br />
the globe during European colonization, as well as more recent introductions. In a<br />
survey of 33 upland homegardens across the Brazilian Amazon, Smith (1996) found<br />
a total of 77 tree species, of which 46% are indigenous to Amazonia, and 27% are<br />
from the Old World. In a study of 51 homegardens in Peru (Lamont et al., 1999) at<br />
least 30 of the 161 species found were exotics, including nine tree species. In the<br />
three villages (two of the Yagua tribe and one ribereño), the two most common<br />
species in all 51 gardens were of Asian origin (i.e., mango and banana).<br />
The importance of homegardens is chiefly the domestic supply of fruits,<br />
condiments, medicines, craft materials, and shade. Near urban centers, however,<br />
they may become part of both subsistence and income-earning strategies through the<br />
production of marketable fruits. How farmers manage the composition of their<br />
homegardens in order to influence production and income generation has been little<br />
studied, but it appears that there is a ubiquitous stock of species valued for domestic<br />
consumption, while others are cultivated specifically as income-earners. <strong>Homegardens</strong><br />
near Iquitos, Peru, may cultivate native palms for use in the handicraft<br />
business (Lamont et al., 1999), or exotic species such as taperibá (Spondias dulcis)<br />
for their prized fruits. In the Colombian Amazon, lulo (Solanum sessiliflorum) is<br />
common in homegardens to supply the markets of Leticia, while the market for fruit<br />
from the ocoró tree (Rheedia spp.) makes it popular in homegardens near Santa<br />
Cruz, Bolivia (J. Penn, pers. obs.).<br />
51
52<br />
R.P. MMILLER<br />
ET AL.<br />
Amazonian homegardens are very diverse in terms of size and number of<br />
species, both on a local level, with properties in the same community exhibiting very<br />
different assemblages, as well as on a regional level. While some of these differences<br />
can be explained, it becomes clear that there is no such thing as a “typical”<br />
homegarden, only trends or patterns. The 21 homegardens studied by Padoch and de<br />
Jong (1991) in the community of Santa Rosa, 150 km upstream from Iquitos,<br />
generally covered between 300 to 700 m 2<br />
m , the size of a usual house lot in that<br />
community. However, the range in size was from 67 to 7322 m 2 . Outlying houses<br />
had larger gardens, but this was not always the case. A typical pattern observed in<br />
many parts of Amazonia is for houses to be located in the central area of the<br />
community, where school, church, meeting hall, soccer field, and television are<br />
normally found. These hamlets can be part of planned “agrovilas” of colonization<br />
projects, or spontaneously formed communities (often based on kin ties) that group<br />
together in order to be attended by municipal services such as schools, health posts,<br />
or power generators. In these cases agricultural fields are located at a distance, and<br />
some sort of homegarden may be found surrounding the shelter used for processing<br />
the cassava crop.<br />
<strong>Homegardens</strong> in Amazonia also must be studied in the context of how dynamism<br />
and change affect the economic, social, and cultural aspects of caboclo and ribereño<br />
societies. A community of 60 households near Iquitos, Peru, whose homegardens<br />
were studied by Coomes and Burt (1997), for example, was originally founded as<br />
an agricultural estate for the production of sugarcane, rum, and fuelwood, and subsequently<br />
was divided up among the former workers in 1971 as an act of agrarian<br />
reform. In the community studied by Padoch and de Jong (1991), also near Iquitos,<br />
life histories of the adults were found to typically include several long economic<br />
migrations and many changes of residence. Lamont et al. (1999) found that the<br />
intermarriage of ribereños within families of the Yagua tribe was associated with<br />
declining use of homegardens in Peru, indicating that researchers need to examine<br />
the resilience of these agricultural systems to social and cultural change.<br />
Further study is needed to determine the extent to which differences in<br />
homegarden size and diversity are random, a product of local processes of sociocultural<br />
development and germplasm accession, or whether they reflect changes in<br />
management choice with regard to cash and energy flows and the perceived<br />
functions of the homegardens. In some cases, traditional homegardens may be<br />
eliminated to make place for more profitable plantations, if agricultural land (space)<br />
increases in value, as has been observed in the region near Manaus. If the farmer has<br />
the means to invest in a profitable crop, the homegarden can be eliminated to plant<br />
papaya (Carica papaya) or passionfruit (Passiflora edulis), or if still closer to<br />
Manaus, to plant horticultural crops (e.g., okra, Abelmoschus esculentus). This<br />
happens especially on better soils, such as anthropogenic black earths or the várzea<br />
alta, the higher part of the floodplain or natural levee that accompanies the Solimões<br />
and Amazonas rivers (J. van Leeuwen, pers. obs.). Penn (2004; 2006) found that<br />
homegardens in Peru were being planted with camu camu trees (Myrciaria dubia)<br />
by ribereños anxious to participate in a regional development program that promoted<br />
the cultivation of this species, extremely rich in vitamin C.
AMAZONIAN HOMEGARDENS<br />
A category of Amazonian homegardens originating from rubber-cacao plantations,<br />
in which an upper stratum of rubber tree canopies is combined with a lower stratum<br />
of cacao, frequently is found on the várzea alta of the rivers Solimões, Amazonas,<br />
and Madeira. The cacao and rubber trees of this two-layer system are always quite<br />
old (J. van Leeuwen, pers. obs.). On the Ilha de Careiro, cacao and rubber were<br />
planted at the beginning of the twentieth century when production of these two<br />
commodities was much more profitable, but planting no longer occurs (Bahri, 1993).<br />
On the Ilha de Careiro and elsewhere many cases can be seen of the gradual<br />
substitution of cacao and rubber by other fruit trees, with the result that the plantation<br />
develops into a multispecies homegarden (Bahri, 1992; 1993). These examples indicate<br />
that homegardens can have a long history, in the sense that present day species<br />
composition does not necessarily closely reflect current economic scenarios. This is the<br />
case in Central Amazonia, where várzea homegardens may contain rubber trees<br />
that have not been tapped for many years. Although the presence of species that<br />
presently have little economic contribution may simply result from low levels of<br />
management, and not a conscious effort of conservation, their maintenance may also<br />
be part of risk-avoidance strategies. Poor farmers will generally refuse to cut a tree if<br />
it is thought that it might be useful at some moment in the future (J. van Leeuwen,<br />
pers. obs., based on work with small farmers in Mozambique and the Amazon).<br />
Differing time horizons and expectations of farmers with regard to local market<br />
demands, land tenure and property size all can influence the configuration of<br />
homegardens and other agroforestry systems. Access to the markets of larger urban<br />
centers represents an important economic factor that comes into play. Studies by<br />
Rosa et al. (1998a; 1998b) near the state capitals Macapá (Amapá), and Belém<br />
(Pará), Brazil, for example, found that small livestock can have considerable<br />
economic importance as components of the homegarden system. In properties<br />
averaging 90 ha near Macapá, although more than 50% of the chickens, ducks, and<br />
pigs raised was consumed by the household, weekly revenue from livestock<br />
averaged R$ 35, a value greater than that obtained from the sale of fruits such<br />
as açaí, bananas, mangos, limes, and cupuaçu (Theobroma grandiflorum), which<br />
averaged R$ 20/week [the real (R$) was approximately equal to the US dollar at that<br />
time and is now exchanged at R$ 2.3 per US$]. Nevertheless, a good portion of the<br />
feed for these animals was said to come from homegarden fruits. In a survey of 20<br />
households near Belém, where property size averaged 1.7 ha, it was found that<br />
families consumed 69% of the fruits, 100% of the medicinals, 85% of the<br />
vegetables, and 85% of the livestock, with the remainder being sold (Rosa et al.,<br />
1998c). Conversely, livestock can destroy homegardens, and make it impossible to<br />
maintain or restart a homegarden. The introduction of water buffalo near Iquitos has<br />
greatly reduced the number of homegardens where these animals are present<br />
(J. Penn, pers. obs.).<br />
4. HOMEGARDEN MANAGEMENT IN AMAZONIA<br />
According to Lathrap (1977), the maintenance of homegardens and clean yards<br />
around the dwellings of indigenous communities creates a domesticated microcosm<br />
out of the surrounding wild forest, otherwise the abode of spirits and other dangers.<br />
53
54<br />
R.P. MMILLER<br />
ET AL.<br />
In Waimiri Atroari villages in Central Amazonia, this zone is used by small<br />
children, who both forage and play at activities such as shooting lizards with toy<br />
bows and arrows (R. Miller, pers. obs.). Although the extent to which Lathrap’s<br />
cosmological interpretation of the significance of homegardens can be applied to<br />
caboclo and ribereño societies may be limited, the maintenance of a terreiro, or<br />
patio (bare-earth yard) often swept daily, is a ubiquitous feature of rural homes in<br />
Amazonia, and serves to reduce hiding places for snakes and insects. The size of this<br />
yard is typically about 500 m 2 (20 x 25 m) and may often be larger. The exact limit<br />
of the terreiro, however, may depend on the time and labor available for weeding.<br />
Beyond the terreiro, the divide between the homegarden of planted trees and<br />
neighboring second growth may not be clearly distinguishable. These fluctuating<br />
boundaries between the bare earth yard, the homegarden, and encroaching second<br />
growth vegetation are important in permitting the establishment and recruitment of<br />
volunteer seedlings of useful trees. Discarded or fallen seeds will germinate in the<br />
shelter of leaf litter and undergrowth, and resulting seedlings may be spared by the<br />
observant farmer during periodic weeding. This process was noted by Huber (1904),<br />
who was probably the first to make specific mention of the ease with which even<br />
introduced species of fruit trees in Amazonia become sub-spontaneous, germinating<br />
from discarded seeds in the more fertile soil around dwellings. This “spontaneous”<br />
aspect of homegardens is in fact an important form of management. Near Iquitos, for<br />
example, Padoch and de Jong (1991) found homegardens to be a “combination of<br />
trees left from pre-existing fallows or forests, deliberately planted vegetation,<br />
spontaneously occurring useful forest plants, species transplanted from the forest,<br />
seeds germinating from the forest,” resulting in mosaics of different-age vegetation.<br />
They also found that 14% of the plants identified as “non-cultivated” were useful<br />
and had been selected for in previous weeding operations. This process, also<br />
important for outlying fields, fits into what Wiersum (1996) described as the<br />
“second stage of domestication,” and is suggestive of how trees may have been<br />
incorporated into agricultural systems in Amazonia during the past millennia. Some<br />
species will simply regenerate more easily than others in these environments. This is<br />
a major reason why Rheedia, Genipa, and Inga species are so common in homegardens<br />
along the Peruvian Amazon (Penn, 2006).<br />
Areas beyond the yard that are not kept “clean” provide a dumping ground for<br />
assorted household and garden wastes, which besides being important as sources of<br />
seeds and forage for domestic fowl, can also represent significant nutrient additions.<br />
Over millennial time scales in Amazonia, humans have generated patches of higher<br />
fertility around their dwellings by concentrating nutrients obtained from surrounding<br />
terrestrial and aquatic ecosystems, resulting in anthropogenic “black earths”<br />
(Lehmann et al., 2004). Data from a hunting study with the Waimiri Atroari tribe in<br />
Central Amazonia (Mazurek, 2001) indicates that an average-size village of 50<br />
people discards approximately 1.5 Mg of bones of game animals every year. Bones<br />
represent a significant contribution of calcium and phosphorus, which complement<br />
the other nutrient elements found in other forms of household wastes. Although<br />
redirecting nutrients can be a conscious practice, such as when farmers place<br />
cassava peelings at the foot of selected fruit trees as fertilizer, for the most part, the<br />
nutrient peak around dwellings that greatly benefits homegardens is an unconscious
AMAZONIAN HOMEGARDENS<br />
practice. Nevertheless, in the case of Waimiri Atroari villages, the zone of greater<br />
fertility is explored for the initial establishment of a belt of fruit trees around the<br />
communal dwelling, which then expands outward concentrically (Miller, 1994).<br />
5. IMPORTANCE OF HOMEGARDENS FOR AGROFORESTRY<br />
DEVELOPMENT IN AMAZONIA<br />
Throughout history, Amazonian farmers were subjected to exploitation as forces of<br />
colonization and trade penetrated the region. They have suffered immensely and<br />
have often been dispossessed of their traditional lands, but have shown a remarkable<br />
ability to adapt to new environments and socioeconomic scenarios. During this<br />
period, their homegardens have changed in many ways. Asian species soon became<br />
common in homegardens after the Conquests, and are an increasingly common part<br />
of these cultural landscapes. Among the various configurations of agroforestry<br />
systems, such as tree/crop combinations in fields, orchards of mixed fruit trees, and<br />
enriched fallows, homegardens represent the most widespread agroforestry practice<br />
employed by farmers in Amazonia today.<br />
Although farmers near urban centers sell homegarden products (Lamont et al.,<br />
1999) as well as livestock (principally fowl) raised in and around homegardens, their<br />
overall contribution for domestic consumption is probably more important. In this<br />
regard, homegardens represent a robust and time-tested technology, employed by the<br />
traditional inhabitants of Amazonia, whether indigenous tribes or caboclos and<br />
ribereños, and from the point of view of food security, they may be of great value on<br />
agricultural colonization frontiers, where farmers face a difficult struggle to<br />
establish themselves and their families.<br />
Originally managed for subsistence according to ethnic practices, homegardens<br />
are now increasingly important for farmer experimentation with commercial crops.<br />
As the locus of experimentation with new tree species and cultivation techniques,<br />
homegardens have the potential to contribute to the development of other agroforestry<br />
systems, and may expand into more commercial groves, as discussed by<br />
Penn (2004) on the new camu camu industry in Peru, and Yamada and Osaqui<br />
(2006) concerning the farmers of Japanese descent in Tomé-açu, Pará, Brazil.<br />
The homegarden can function as a “staging area” for testing new species and<br />
storing, safeguarding, and multiplying germplasm for transfer to and between fields<br />
(Coomes and Ban, 2004). In this manner, the homegarden can be an integral<br />
component of the larger agricultural system of the property as well as a key node in<br />
the local network of agrobiodiversity, if one considers the exchange of plant genetic<br />
resources between households in a community.<br />
The historical study of the course of development of homegardens as a basic unit<br />
of interaction between humans and trees holds lessons relevant to the present-day<br />
scenario of advancing deforestation, in which agroforestry is ascribed a potential<br />
role in developing more sustainable land use. While the technologies or practices<br />
involved in expanding agroforestry systems out to fields are not necessarily those<br />
employed in homegardens, they entail similar concepts such as tree culture, nutrient<br />
cycling, and permanent soil cover, among others, and in this respect, homegardens<br />
55
56<br />
R.P. MMILLER<br />
ET AL.<br />
could be considered as a conceptual core for agroforestry development. The basic<br />
units of information that farmers need to develop new models of agroforestry<br />
systems are in essence the knowledge of tree species, as to their behavior and<br />
interaction with other species. <strong>Homegardens</strong>, where trees can more easily be cared<br />
for and observed, offer optimal locations for the introduction and evaluation of new<br />
species.<br />
Nevertheless, in any given community, members will exhibit different levels of<br />
perception and relationship with plants, varying from the “green thumbs” to those<br />
whose interest in plants goes little beyond their daily needs. In the past, such plant<br />
lovers were most likely responsible for the domestication of useful species, and<br />
today, they are the experimenters and innovators who generate new technologies by<br />
acute observation and the ability to create heuristic models of the behavior, growth,<br />
and interactions of the various components of their agroforestry systems. This is a<br />
very personal and human process of plant management, which mixes personality<br />
traits and life histories, and cannot simply be replicated or substituted by research<br />
agencies! The complexity of this social/agronomic interface may explain why<br />
homegardens appear to elude science, as Nair (2001) remarked.<br />
Making the leap from growing fruit trees around houses for domestic<br />
consumption to planting trees in fields for production of fruit, timber, and other<br />
products, nonetheless, requires dealing with an entirely different set of constraints.<br />
The main constraints to further developing homegardens or expanding them out to<br />
fields for greater productivity and income generation are the lack of adequate<br />
germplasm, risk of accidental fires, survival of seedlings in the dry season and soil<br />
fertility (Smith et al., 1995; Smith et al., 1996; Smith et al., 1998; Miller, 2001).<br />
There may also be a need to modify the configuration of species and management<br />
practices observed in traditional systems to meet increased nutrient exports and<br />
labor requirements, as well as market demands. At present, commercial products<br />
obtained from early stages of agroforestry systems are mostly fruits, and marketing<br />
such products, especially processed pulps, requires facilities most farmers cannot<br />
afford to have by themselves, while farmers’ associations lack the entrepreneurial<br />
and managerial expertise to run such installations. This factor has led many<br />
innovative agroforestry projects dependent on pulp processing facilities down the<br />
path to failure (Penn, 2004).<br />
Despite the official interest in agroforestry, due to the immensity of the Amazon<br />
region, extension services have been unable to meet the growing demands for<br />
technical assistance. This scenario implies that if agroforestry is to fulfill its promise<br />
of providing an alternative and more sustainable form of land use in Amazonia,<br />
extension efforts need to break out of traditional paradigms and the mold of<br />
commodity-based systems to interact with farmers on a different level of knowledge.<br />
The traditional socio-cultural practices involved in acquiring and testing new<br />
germplasm, as seen in homegardens, must be included in rural development projects,<br />
and stimulated by creative new approaches, with farmers viewed as partners and<br />
experimenters in the development and domestication of new generations of tree<br />
crops. In this partnership, a major role for extension should be to help provide the<br />
necessary germplasm and information.
AMAZONIAN HOMEGARDENS<br />
Surrogate homegardens, based at rural schools, where interesting germplasm can<br />
be tested and multiplied for access by frontier farmers, while at the same time<br />
improving nutrition for their children, are one suggestion to increase the spread and<br />
efficiency of extension services. With homegardens as a conceptual core, this form<br />
of agroforestry extension should be accompanied by other initiatives and small-scale<br />
experiments to improve the productivity of subsistence crops, through the use of<br />
green manures, polycultures, and management of organic matter, among other practices.<br />
Although this proposal appears to be simple, existing experiences in a similar vein<br />
must be identified and studied to know if it can work and how to make it work.<br />
ACKNOWLEDGEMENTS<br />
Portions of this paper were adapted from Miller and Nair (2006). Charles Clement<br />
provided valuable comments on an early version of this chapter. We also thank the<br />
comments and suggestions of three anonymous reviewers.<br />
REFERENCES<br />
Acuña C. 1639. Novo descobrimento do Grande Rio das Amazonas. Agir, Rio de Janeiro<br />
[reprinted 1994], 179p.<br />
Alden D. 1976. The significance of cacao production in the Amazon Region during the late<br />
Colonial period: an essay in comparative economic history. Proc Am Philos Soc 120:<br />
103 – 135.<br />
Alves Filho A., Souza Jr. J.A. and Bezerra Neto J. M. 2005. Pontos de história da Amazônia<br />
Vol. 1 Paka-Tatu, Belém, 208p.<br />
Amazonas L.S.A. 1852. Dicionário Topográfico, Historico, Descritivo do Alto Amazonas.<br />
Facsimile ed., Grafima, Manaus [reprinted 1984], 208p.<br />
Anderson E. 1952. Plants, man and life. Little, Brown, Boston, 245p.<br />
Avé-Lallemant R. 1859. No Rio Amazonas. EDUSP, São Paulo [reprinted 1980], 288p.<br />
Bahri S. 1992. L’Agroforesterie, une alternative pour le développement de la plaine alluviale<br />
de l’Amazone – L’exemple de l’île de Careiro. PhD dissertation, Université de<br />
Montpellier II. Montpellier, 277p.<br />
Bahri S. 1993. Les systèmes agroforestiers de l’île de Careiro. Amazoniana 12: 551 – 563.<br />
Bates H.W. 1863. The naturalist on the river Amazons. University of California Press,<br />
Berkeley [reprinted 1988], 465p.<br />
Cabral A.S. 1995. Contact-induced language change in western Amazonia: The non-genetic<br />
origin of the Kokama language. PhD dissertation. University of Pittsburg, Pittsburg, 415p.<br />
Carvajal G. 1542. The Discovery of the Amazon. AMS Press, New York [reprinted 1970],<br />
467p.<br />
Clement C.R. 1989. A center of crop genetic diversity in western Amazonia: a new<br />
hypothesis of indigenous fruit-crop distribution. BioScience 39: 624 – 631.<br />
Clement C.R. 1999a. 1492 and the loss of Amazonian crop genetic resources. I. The relation<br />
between domestication and human population decline. Econ Bot 53: 188 – 202.<br />
Clement C.R. 1999b. 1492 and the loss of Amazonian crop genetic resources. II. Crop<br />
biogeography at contact. Econ Bot 53: 203 – 216.<br />
Coomes O.T. and Ban N. 2004. Cultivated plant species diversity in a home garden of an<br />
Amazonian peasant village in northeastern Peru. Econ Bot 58: 420 – 434.<br />
Coomes O.T. and Burt, G.J. 1997. Indigenous market-oriented agroforestry: dissecting local<br />
diversity in western Amazonia. Agroforest Syst 37: 27 – 44.<br />
57
58<br />
R.P. MILLER M<br />
ET AL.<br />
Daniel J. 1776. Tesouro descoberto no Rio Amazonas. Contraponto, Rio de Janeiro (2 vol.)<br />
[reprinted 2004], 1219p.<br />
Dean W. 1995. With broadax and firebrand: the destruction of the Brazilian Atlantic forest.<br />
University of California, Berkeley, 482p.<br />
Denevan W.M. 2002. Cultivated landscapes of native Amazonia and the Andes. Oxford<br />
University Press, Oxford, 395p.<br />
Denevan W.M. and Padoch C. 1987. Swidden-fallow agroforestry in the Peruvian Amazon.<br />
Advances in Economic Botany 5, New York Botanical Garden, Bronx, 112p.<br />
Edmundson G. 1922. Journal of the travels and labors of Father Samuel Fritz in the River<br />
Amazons between 1686 and 1723. Hakluyt Society, London, Second Series, No. 51,<br />
164p.<br />
Hemming J. 1987. Amazon frontier: The defeat of the Brazilian Indians. Harvard University<br />
Press, Cambridge, MA, 647p.<br />
Holanda S.B. 1965. Historia Geral da Civilização Brasileira. Tomo 2 – O Brasil Monárquico.<br />
DIFEL, São Paulo, 415p.<br />
Homma A.K.O. 2003. História da agricultura na Amazônia: da era pré-colombiana ao terceiro<br />
milênio. Embrapa Informação Tecnológica, Brasília, 274p.<br />
Huber J. 1904. Notas sobre a patria e distribuição geographica das arvores fructiferas do Pará,<br />
pp 375 – 406. Boletim Museu Goeldi Historia Natural e Ethnographia. Tomo IV.<br />
Lamont S.R., Eshbaugh W.H. and Greenberg A.M. 1999. Species composition, diversity, and<br />
use of homegardens among three Amazonian villages. Econ Bot 53: 312 – 326.<br />
Lathrap D.W. 1977. Our father the cayman, our mother the gourd: Spinden revisited, or a<br />
unitary model for the emergence of agriculture in the New World. In: Reed C.A. (ed.),<br />
Origins of agriculture, pp. 713 – 751. Mouton, The Hague.<br />
Lehmann J., Kern D.C., Glaser B. and Woods W.I. 2004. Amazonian dark earths: origin,<br />
properties and management. Kluwer, Dordrecht, 523p.<br />
Lima D.M. 1999. A construção histórica do termo caboclo. Novos Cadernos NAEA 2: 5 – 32.<br />
Maués R.H. 2001. Um aspecto da diversidade cultural do caboclo amazônico: a religião. In:<br />
Vieira I.C.G., Silva J.M.C., Oren D.C. and D’Incao M.A. (eds), Diversidade biológica e<br />
cultural da Amazônia, pp. 253-272. Museu Paraense Emílio Goeldi, Belém.<br />
Mazurek R.R.S. 2001. Kinja txi taka nukwa myrykwase: fishing and hunting among the<br />
Waimiri Atroari Indians from Central Amazonia. PhD dissertation, University of Illinois,<br />
Chicago, 115p.<br />
Miller E.T. 1992. Arqueologia nos empreendimentos hidrelétricos da Eletronorte; resultados<br />
preliminares. Eletronorte, Brasília, 93p.<br />
Miller R.P. 1994. Estudo da fruticultura tradicional dos índios Waimiri-Atroari: Base para a<br />
extensão agroflorestal. Anais I Congresso Brasileiro de Sistemas Agroflorestais 2:<br />
449 – 462, Embrapa-CNPF, Colombo.<br />
Miller R.P. 2001. Extractive forest products and agroforestry on an agricultural frontier: A<br />
case study with the Parakanã ã tribe of the trans-Amazon region, Pará, Brazil. PhD<br />
dissertation. University of Florida, Gainesville, 227p.<br />
Miller R.P. and Nair P.K.R. 2006. Indigenous agroforestry systems in Amazonia: from<br />
prehistory to today. Agroforest Syst 66: 151 – 164.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Padoch C. and de Jong W. 1991. The house gardens of Santa Rosa: diversity and variability in<br />
an Amazonian agricultural system. Econ Bot 45: 166 – 175.<br />
Padoch C. and Pinedo-Vasquez M. 2001. Resource management in Amazonia: Caboclo and<br />
ribereño traditions. In: Maffi L. (ed.), On biocultural diversity: Linking language,<br />
knowledge, and the environment, pp. 364-378. Smithsonian Institution press, Washington,<br />
DC, 578p.
AMAZONIAN HOMEGARDENS<br />
Penn J.W. Jr. 2004. Another boom for Amazonia? Socioeconomic and environmental<br />
implications of the new camu camu industry in Peru. PhD dissertation, University of<br />
Florida, Gainesville, 298p.<br />
Penn J.W. Jr. 2006. The cultivation of camu camu (Myrciaria dubia): A tree planting<br />
programme in the Peruvian Amazon. Forests Trees Livelihoods 16: 85 – 101.<br />
Piperno D.R. and Pearsall D.M. 1998. The origins of agriculture in the lowland Neotropics.<br />
Academic Press, San Diego, 400p.<br />
Piperno D.R., Ranere A.J., Holst I. and Hansell P. 2000. Starch grains reveal early root crop<br />
horticulture in the Panamanian tropical forest. Nature 407: 894 – 897.<br />
Poeppig E. 2003. Viaje al Perú y al Río Amazonas, 1827-1832. CETA, Iquitos, 435p.<br />
Renard-Casevitz F.-M. 1992. História Kampa, memória Ashaninca. In: Cunha M.C. (ed.),<br />
História dos índios no Brasil. Companhia das Letras, pp 197 – 212. Sec. Municipal de<br />
Cultura, São Paulo.<br />
Ribeiro D. 1997. O povo brasileiro: a formação e o sentido do Brasil. Companhia das Letras,<br />
São Paulo, 476p.<br />
Rios M.C. 2001. Historia de la Amazonía Peruana. Periodos: Independencia y república.<br />
Editora Selva, Iquitos, 322p.<br />
Roosevelt A.C. 1994. Amazonian anthropology: strategy for a new synthesis. In: Roosevelt<br />
A.C. (ed.), Amazonian Indians from prehistory to present: Anthropological perspectives,<br />
pp 1 – 29. University of Arizona Press, Tucson.<br />
Rosa L.S., Cruz H.d.S., Tourinho M.M. and Ramos C.A.P. 1998a. Aspectos estruturais e<br />
funcionais dos quintais agroflorestais localizados nas várzeas da costa amapaense. II<br />
Congresso Brasileiro em Sistemas Agroflorestais – Resumos Expandidos, pp 164 – 166.<br />
Embrapa, Belém.<br />
Rosa L.S., Cruz H.d.S., Tourinho M.M. and Ramos C.A.P. 1998b. Caracterização dos<br />
quintais agroflorestais localizados nas várzeas do estuário amazônico. II Congresso<br />
Brasileiro em Sistemas Agroflorestais – Resumos Expandidos, pp 161 – 163. Embrapa,<br />
Belém.<br />
Rosa L.S., Da Silva L.C.B., Melo A.C.G. and Cabral W.d.S. 1998c. Avaliação e<br />
diversificação de quintais agroflorestais na comunidade de Murinim – Benfica, Município<br />
de Benevides - Pará. II Congresso Brasileiro em Sistemas Agroflorestais – Resumos<br />
Expandidos, pp 167 – 169. Embrapa, Belém.<br />
Salick J. 1992. Crop domestication and the evolutionary ecology of cocona (Solanum<br />
sessiliflorum Dunal). Evol Biol 26: 247 – 285.<br />
San Ramon J.S. 1994. Perfiles históricos de la Amazonia Peruana. CETA-CAAAP-IIAP,<br />
Iquitos, Perú. 274p.<br />
Santos F.J. 2002. Além da conquista: guerras e rebeliões na Amazônia pombalina. Editora da<br />
Universidade do Amazonas, Manaus, 239p.<br />
Schroth G., Mota M.S.S., Lopes R. and Freitas A.F. 2004. Extractive use, management, and in<br />
situ domestication of a weedy palm, Astrocaryum tucuma, in the Central Amazon. For<br />
Ecol Manag 202: 161 – 179.<br />
Smith B.D. 2001. Documenting plant domestication: the consilience of biological and<br />
archeological approaches. Proc Nat Acad Sci 98: 1324 – 1326.<br />
Smith N.J.H. 1996. Home gardens as a springboard for agroforestry development in<br />
Amazonia. Int Tree Crops J 9:11 – 30.<br />
Smith N.J.H. 1999. The Amazon river forest: a natural history of plants, animals, and people.<br />
Oxford University Press, New York, 208p.<br />
Smith N.J.H., Dubois J., Current D.L.E. and Clement C. 1998. Agroforestry experiences in<br />
the Brazilian Amazon: constraints and opportunities. Pilot program to conserve the<br />
Brazilian rainforest. World Bank, Brasília, 80p.<br />
59
60<br />
R.P. MILLER M<br />
ET AL.<br />
Smith N.J.H., Falesi I.C., Alvim P.d.T. and Serrão E.A.S. 1996. Agroforestry trajectories<br />
among smallholders in the Brazilian Amazon: innovation and resiliency in pioneer and<br />
older settled areas. Ecol Econ 18: 15 – 27.<br />
Smith N.J.H., Fik T.J., Alvim P.d.T., Falesi I.C. and Serrão E.A.S. 1995. Agroforestry<br />
developments and potential in the Brazilian Amazon. Land Degrad Rehabil 6: 251 – 263.<br />
Stanfield M.E. 1998. Red rubber, bleeding trees: Violence, slavery, and empire in northwest<br />
Amazonia, 1850-1933. University of New Mexico Press, Albuquerque, 270p.<br />
Van Leeuwen J. and Gomes J.B. 1995. O pomar caseiro na região de Manaus, Amazonas, um<br />
importante sistema agroflorestal tradicional. Actas II Encontro da Sociedade Brasileira de<br />
Sistemas de Produção, Londrina, PR, 21-23/nov/95, pp 180 – 189. IAPAR, Londrina<br />
(http://www.inpa.gov.br/cpca/johannes/joha-pomar.html; last accessed: February 5, 2006).<br />
Wiersum K.F. 1996. Domestication of valuable tree species in agroforestry systems: evolutionary<br />
stages from gathering to breeding. In: Leakey R.B., Temu A.B., Melnyk M., and<br />
Vantomme P. (eds), Domestication and commercialization of non-timber forest products,<br />
pp 147 – 158. FAO, Rome.<br />
Yamada M. and Osaqui H.M.L. 2006. The role of homegardens in agroforestry development:<br />
lessons from Tomé-Açu, a Japanese-Brazilian settlement in the Amazon. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 299 – 316. Springer Science, Dordrecht.
CHAPTER 5<br />
HOMEGARDENS OF MESOAMERICA:<br />
BIODIVERSITY, FOOD SECURITY,<br />
AND NUTRIENT MANAGEMENT<br />
F. MONTAGNINI<br />
Yale University, School of Forestry and Environmental Studies, 370 Prospect St.,<br />
New Haven, CT 06511, USA; E-mail: <br />
Keywords: Mayan culture, Nutritional value, Rural development, Structural complexity.<br />
Abstract. The region of Mesoamerica is densely populated and it suffers from poverty and<br />
malnutrition both in urban and rural areas. It is home to the Mayan civilization that practiced<br />
sustainable agricultural systems, involving many native crops and soil conservation strategies,<br />
for centuries. The homegardens, which provide the household with a basic food source as well<br />
as high value products to generate cash income are important in Mesoamerica, and are often<br />
used as tools in development projects that promote food security, especially in the poorest<br />
areas of Mesoamerica. The Mesoamerican homegardens are quite diverse in vertical and<br />
horizontal structure and species composition. Both exotic and native plants are used, with<br />
emphasis on fruit trees. Domestic animals, especially chickens and pigs, add protein to a diet<br />
that is generally protein-deficient. Many indigenous communities (descendants of the ancient<br />
Maya) still manage these homegardens using techniques that include residue management and<br />
ash deposition, thus enhancing nutrient recycling and conservation. Carbon sequestration may<br />
be important due to the efficient capture of solar radiation in the multi-layered homegardens,<br />
although its global or regional importance is minimal due to the relatively small area under<br />
the homegarden system. Management strategies that promote nutrient recycling and maintain<br />
high species diversity should be encouraged to ensure sustainability of homegardens in the<br />
region.<br />
1. INTRODUCTION<br />
A vast area of what is known today as Mesoamerica was the home of the Mayan<br />
civilization. The remnants of Mayan culture are concentrated in southern Mexico,<br />
61<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 61–84.<br />
© 2006 Springer. Printed in the Netherlands.
62 F. MONTAGNINI<br />
Guatemala, and Belize. The Mayan people are known to have practiced sustainable<br />
agricultural systems for centuries, cultivating a wide variety of native crops and<br />
applying indigenous knowledge on nutrient cycling and soil conservation (De Clerck<br />
and Negreros-Castillo, 2000; Benjamin et al., 2001). In regions such as the<br />
Tehuacán-Cuicatlán Valley in Central Mexico, human cultures have a history of<br />
nearly 10 000 years and at present several indigenous ethnic groups continue to<br />
follow cultural traditions in plant gathering and cultivation (González-Soberanis and<br />
Casas, 2004). The long history of interactions between human cultures and plant<br />
diversity has created a substantial body of traditional knowledge on the myriad uses<br />
of plants. The existence of nearly 1200 plant species utilized by local peoples for<br />
different purposes, most of them native wild plants, has been documented; many of<br />
these species are obtained through gathering, but several species are also under<br />
silvicultural management (Casas et al., 2001; González-Soberanis and Casas, 2004).<br />
This rich tradition of sustainable agricultural practices in Mesoamerica justifies<br />
an extensive study of homegardens in the region. Several types of homegardens are<br />
practiced in the region by the descendants of Maya in present-day Mexico,<br />
Guatemala, Belize, and Honduras, by other indigenous groups, and by people of<br />
Hispanic descent in Nicaragua, El Salvador, Costa Rica, and Panama. Traditional<br />
agroecosystems, which include ‘forest gardens’ or ‘homegardens,’ contain combinations<br />
of trees with an understorey of annual and perennial crops and sometimes<br />
livestock. Villagers live within or adjacent to their gardens and maintain them over<br />
many generations. In present-day Mayan towns in the Yucatán Peninsula of Mexico,<br />
this type of forest gardens covers about 10% of the region’s forested area (Noble and<br />
Dirzo, 1997). Small, scattered forest or agroforests can provide local or regional<br />
environmental services such as conservation of biodiversity (Guindon, 1996; Harvey<br />
and Haber, 1999). Thus, the practice of homegardens can meet forest conservation<br />
needs in regions where deforestation and population growth are constant threats, as<br />
is the case in much of the Mesoamerican region.<br />
As in other regions of the neotropics, such as Amazonia (Miller et al., 2006),<br />
present-day homegardens of Mesoamerica represent the reorganization of original<br />
indigenous practices as a result of the changes brought by colonization, among<br />
which the most outstanding feature was the incorporation of non-native fruit trees<br />
and crops. Today, homegardens are of vital importance to the local subsistence<br />
economy and food security in the region, especially in regions that still carry the<br />
influence of Mayan culture (De Clerck and Negreros-Castillo, 2000; Méndez et al.,<br />
2001; Zaldívar et al., 2002; Wezel and Bender, 2003; Blanckaert et al., 2004;<br />
González-Soberanis and Casas, 2004).<br />
This chapter describes the characteristics of homegardens in Mesoamerica, with<br />
emphasis on biodiversity, their importance in sustaining food security in rural areas,<br />
and their role in nutrient cycling. Information is presented on indigenous systems<br />
that have been practiced by descendants of the ancient Maya for many centuries in<br />
regions of Mexico, Belize, and Guatemala, as well as on systems currently being<br />
practiced in regions beyond the Mayan influence such as in Nicaragua, El Salvador,<br />
Honduras, Costa Rica, and Panama.
2. GENERAL ECOLOGICAL AND SOCIOECONOMIC CHARACTERISTICS<br />
OF MESOAMERICA<br />
Culturally Mesoamerica joins the present-day middle and south Mexico, Belize,<br />
Guatemala, parts of Honduras, and El Salvador (Fig. 1). Geographically, the other<br />
three countries of Central America (Nicaragua, Costa Rica, and Panama) are also<br />
included in the region. Most geographers consider Central America to be part of the<br />
North American continent; however, they do not consider Mexico to be a part of<br />
Central America. The Caribbean islands are often considered separately from<br />
Mesoamerica because they are culturally very diverse. For the purposes of this<br />
chapter an example is drawn from Cuba, a country with a Hispanic tradition as rich<br />
as many of the countries of Mesoamerica, and with similar ecological and economic<br />
conditions.<br />
Figure 1. Map of Mesoamerica (source: www.biodiversityhotspots/mesoamerica/; last<br />
accessed: January 2006).<br />
2.1. Ecological setting<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
The climate, vegetation, and soils of Mesoamerica are very heterogeneous given the<br />
latitudinal expanse of the region and its montane relief. This results in high<br />
ecological heterogeneity that gives room to a whole variety of agricultural systems.<br />
The climate ranges from mild temperate-subtropical in north-central Mexico and<br />
Guatemala, to tropical in the rest of Central America (Richards, 1996). There is a<br />
general pattern of a more humid climate (annual precipitation 3000 to 5000 mm)<br />
along the eastern or Caribbean side due to the influence of the humidity brought by<br />
the trade winds. Winds reach the central volcanic mountain range in the region,<br />
63
64<br />
cause rains on the Atlantic side, and quickly lose much of their humidity.<br />
Consequently, most of the Pacific watersheds are drier with annual precipitation<br />
ranging from 300 to 2000 mm yr –1 and a marked dry season (November through<br />
April). The vegetation follows the climatic pattern ranging from subtropical and<br />
tropical rainforest in the Caribbean lowlands to dry forests and savannas in the<br />
Pacific watersheds (Richards, 1996).<br />
The soil types cover a whole array from older, less fertile Oxisols and Ultisols<br />
(US Soil Taxonomy System), to younger Andosols and Inceptisols, especially in<br />
areas affected by past or present volcanic activity and in alluvial zones. Several<br />
other types of soils can be found due to the varied climatic and topographic<br />
conditions of the region (Sanchez, 1976; De Las Salas, 1987). In general, due to the<br />
recent volcanic influence, the soils of the region are relatively younger and more<br />
fertile than many soils of other regions of Latin America, such as the Amazon Basin.<br />
The Mesoamerican region comprises an area with diverse plant and animal life.<br />
The varied topography, geology, vegetation, and drainage patterns within the region<br />
result in a rich array of vegetation types and animal communities. More than 24 000<br />
plant species, 521 mammal species, 1193 bird species, 685 reptile species, and 460<br />
amphibian species have been identified within Mesoamerica, many of which are<br />
endemic to the region 1 . Moreover, Mesoamerica is the third most biologically<br />
diverse region in the world; Myers et al. (2000) identified it as one of the world’s 25<br />
biological hotspots. Covering an estimated 0.5% of the world’s terrestrial surface, it<br />
is home to roughly 7 to 10% of the world’s plant and animal species (Harvey et al.,<br />
2005). Several trees that are currently planted worldwide in agroforestry<br />
combinations, such as Leucaena leucocephala, Gliricidia sepium, and some species<br />
of Acacia and Mimosa, have their centers of origin and diversity in Mesoamerica<br />
(NAS, 1979; 1980; Dommergues, 1987). Mesoamerican homegardens, in addition to<br />
meeting the immediate alimentary and economic needs of the people, also act as<br />
repositories of local biodiversity as they include a dynamic mixture of native and<br />
useful species.<br />
2.2. Cultural setting<br />
F. MONTAGNINI<br />
The region of Mesoamerica is culturally and socioeconomically diverse, sharing<br />
certain characteristics such as a strong Spanish colonial influence (with the<br />
exception of Belize) and a strong dependence on agriculture and natural resources<br />
(Harvey et al., 2005). In parts of Mesoamerica (southern Mexico, northern<br />
Guatemala, and Belize) homegardens and other types of agriculture carry the<br />
influence of ancient traditions from the indigenous Mayan groups that lived in this<br />
region prior to the arrival of European conquerors.<br />
Several studies have reported and discussed sustainable land use practices that<br />
were used by the Maya, including terracing, using soil algae or wetland soil to<br />
enrich upland garden plots, and other soil conservation strategies (Barrera et al.,<br />
1977; Turner and Harrison, 1981; Beach and Dunning, 1995; Fedick and Morrison,<br />
2004). Archaeological evidence of the use of homegardens by the ancient Maya<br />
include the location of residential sites within prime agricultural land, strategic<br />
placement of households to allow for gardening space, the addition of soil
amendments as indicated by nutrient enrichment within house lots, and the<br />
distribution of tools in the vicinity of residences (Fedick and Morrison, 2004).<br />
The decline of the Maya civilization (~700 BC to 800 AD) has been attributed in<br />
part to complex economic, political, and social changes that led people to change<br />
their traditional sustainable agricultural practices to less diverse agricultural systems<br />
(Barrera et al., 1977; Turner and Harrison, 1981; Atran, 1993; Atran et al., 1999).<br />
A set of political and ecological factors apparently led some Maya groups such as<br />
the lowland Maya of Petén, Guatemala, to reject a diverse swidden-fallow<br />
management strategy for a more simple ‘milpa’ or shifting agricultural system that<br />
provided fewer forest products. This led to a less diverse agricultural landscape and<br />
a less diverse biological landscape (Atran, t 1993; Steinberg, 1998; Atran et al.,<br />
1999). The milpa system consisted of 2 to 5 ha plots that were cut and burnt, and<br />
cultivated mainly with maize (Zea mays). In the traditional system, after a few<br />
harvests the plots were left to regenerate with a long fallow cycle, leaving tree<br />
species time to mature and bear fruits (15 to 40 years).<br />
At present, many Maya groups such as the Mopan of Belize have shortened the<br />
fallow periods to about 5 years. With such a short fallow cycle, the vegetation<br />
regenerating in the milpas is much less diverse than in the traditional Mayan<br />
systems, with only a few useful species of shrubs and palms (Steinberg, 1998).<br />
However, some authors argue that only the most sophisticated and intensive type of<br />
Maya agriculture collapsed, while the oldest, simplest, and most ecologically stable<br />
type is still being practiced (Atran, 1993). The milpa system – as practiced today –<br />
with dispersed fruit trees and vegetable crops and livestock has the attributes of a<br />
productive homegarden.<br />
2.3. Socioeconomic conditions<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
With a total land area of almost 2.5 million km 2 and a total population size of almost<br />
140 million people, Mesoamerica is one of the most densely populated regions of<br />
not only Latin America but also the entire world (Harvey et al., 2005). The current<br />
population of Central America is approximately 38 million people, of which about<br />
20% are indigenous (Harvey et al., 2005). With a yearly growth rate of about 2.6%,<br />
the population is expected to double within the next 25 years. The overall population<br />
density of the region is 56 people per km 2 , with a range from 296 people per km 2 in<br />
El Salvador to just 11 in Belize (Table 1).<br />
Despite its recent economic growth, Central America remains one of the world’s<br />
poorest regions. About 50% of the population is poor (i.e., unable to cover basic<br />
needs such as nutrition and housing) and 23% is extremely poor (i.e., not able to<br />
cover even daily basic nutrition; Harvey et al., 2005). Particularly striking are the<br />
cases of Honduras and Guatemala with poverty levels of 74.5% and 78.5%,<br />
respectively (Harvey et al., 2005). The region’s poverty has led to the massive<br />
exploitation of its natural resources. Large areas of forest have been cut down and<br />
burnt for firewood, used in the production of paper, and cleared for agricultural uses.<br />
Despite an equal distribution of population between rural and urban areas, rural<br />
populations are considerably poorer than their urban counterparts (Harvey et al.,<br />
2005).<br />
65
66<br />
Mesoamerica has diverse ecological, cultural, and socioeconomic conditions that<br />
have given origin to varied agricultural systems. The prevalent conditions of rural<br />
poverty and associated malnutrition call for the need of agricultural systems that can<br />
help fulfill urgent household needs. <strong>Homegardens</strong>, whose main function is to provide the<br />
household with a basic food source and marketable products, are extremely important—<br />
given the socioeconomic conditions prevalent today in Mesoamerica.<br />
Country<br />
F. MONTAGNINI<br />
Table 1. Area,population size and population density of Mesoamerica.<br />
Area<br />
(km²)<br />
Population<br />
size<br />
Population<br />
density<br />
(no./km²)<br />
Mexico 1,964,375 101,879,000 52<br />
Nicaragua 131,847 4,918,000 37<br />
Honduras 112,520 6,406,000 57<br />
Guatemala 108,917 12,974,000 119<br />
Panama 75,536 2,846,000 38<br />
Costa Rica 51,113 3,773,000 74<br />
Belize 22,965 256,000 11<br />
El Salvador 21,046 6,238,000 296<br />
Total 2,488,319 139,290,000 56<br />
Source: Data update 2001 estimates, http://www.globalgeografia.com/north_america/nam_<br />
sup.htm (last accessed: January 2006).<br />
3. COMPOSITION AND STRUCTURE OF HOMEGARDENS IN<br />
MESOAMERICA<br />
As in other regions worldwide, the structure and composition of homegardens in<br />
Mesoamerica are quite complex. A full spectrum of homegarden practices can be<br />
found in different locations of Mesoamerica, ranging from near complete<br />
domination of woody perennials to homegardens where trees may account for less<br />
than 20% of the annual productivity. Plant composition in homegardens of<br />
Mesoamerica is influenced by access to water, owners’ economic activities, labor<br />
availability, traditional social organization, modernization processes, and economic<br />
development (Blanckaert et al., 2004). In general, plant species composition within<br />
the homegardens is the result of continuous selection in which the family usually<br />
favors the planting of fruit trees with high productivity (Caballero, 1992).<br />
Most homegardens of Mesoamerica consist of several vertical and horizontal<br />
strata in which plants are arranged according to their adaptability to the existing<br />
light conditions and nutrient resources (Fig. 2). The number of individual plants per<br />
stratum, however, varies among homegardens; older, more mature homegardens<br />
display more developed tree strata. Some homegardens resemble agricultural fields<br />
with an emphasis on herbaceous and low shrub strata, with a greater focus on<br />
agricultural crop production. Others have more trees, with architecture similar to
that of the native forests of the region (Gillespie et al., 1993; De Clerck and<br />
Negreros-Castillo, 2000; Méndez et al., 2001; Zaldívar et al., 2002; Blanckaert et al.,<br />
2004).<br />
Figure 2. Most homegardens of Mesoamerica consist of several vertical and horizontal<br />
strata, with plants arranged according to their adaptability to light and nutrient resources. A<br />
homegarden in Siquirres, in the Caribbean lowlands of Costa Rica showing vertical<br />
stratification with peach palm ( Bactris gasipaes) in the top layer (right), coconut palms<br />
(Cocos nucifera) and plantains or bananas ( Musa a spp.) in the lower tree strata (left),<br />
sugarcane (Saccharum officinarum), and other herbaceous crops in the herb layer (Photo: R.<br />
González).<br />
3.1. Vertical stratification<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
The vertically stratified homegardens are potentially more productive on an area<br />
basis since they can capture more resources and exhibit tighter nutrient cycling, than<br />
those without a stratified arrangement. For example, in a study of four homegardens<br />
in the Petén, Guatemala, Gillespie et al. (1993) reported high structural complexity,<br />
with full canopy closure in the layers within the canopy. The garden architecture<br />
made efficient use of light and space, with intensive management for food and fuel<br />
production. The development of homegardens in the area utilized existing trees,<br />
leaving the most useful as residuals after thinning, and inserting other desirable trees<br />
and shrubs in the understorey and open space. This strategy seemed to maximize<br />
67
68<br />
light use, according to results of measurements of incident radiation at different<br />
canopy levels reported by Gillespie et al. (1993).<br />
In most Mesoamerican homegardens, each stratum contains plant species that<br />
belong to a characteristic life form, much like in a native forest of the same region.<br />
In homegardens of the Zona Maya of Quintana Roo, Yucatán Península, Mexico,<br />
there were six strata: low herbs, low shrubs, tall shrubs, fruit trees, timber trees, and<br />
a stratum with vines (De Clerck and Negreros-Castillo, 2000). These authors studied<br />
the species composition of each stratum, and concluded that in these systems the<br />
efficient use of space and resources maximized the production of food, timber,<br />
medicinal plants, and non-timber products to cover the farmers’ needs. They<br />
suggested that these systems (or analogs of these in terms of structure and<br />
composition) could be managed in a manner that protects the natural resource base<br />
of the region.<br />
3.2. Plant species composition<br />
F. MONTAGNINI<br />
The species composition of the homegardens in Quintana Roo analyzed by De<br />
Clerck and Negreros-Castillo (2000) was much like others in Mesoamerica and in<br />
other regions of Latin America as well, with a mixture of native and exotic species<br />
in each stratum fulfilling the farmers’ needs. The herbaceous stratum (0 to 0.5 m<br />
tall) was comprised of herbs and creepers such as basil (Ocimum basilicum), squash<br />
(Cucurbita spp.), and sweet potatoes (Ipomoea batatas), containing an average of<br />
14% of all species in the homegarden. The low shrub stratum (0.5 to 1.5 m tall)<br />
contained annual and perennial herbaceous plants such as tomatoes (Lycopersicum<br />
esculentum), maize or corn (Zea mays), ruda (Ruta chalapensis), and included<br />
several shade-tolerant species such as cassava (Manihot esculenta), ginger (Zingiber<br />
officinale), pineapple ( (Ananas comosus),<br />
and taro (Colocasia esculenta). The low<br />
shrub stratum contained 12% and the tall shrub stratum contained 15% of the total<br />
number of species of the homegardens. The low tree stratum was dominated by fruit<br />
trees, most frequently by Citrus spp., and contained 41% of the total number of<br />
species; this stratum was often dominant in the absence of the fifth stratum (tall<br />
trees). The presence of the tall tree stratum, with 15% of the species, was an<br />
indicator of the maturity of homegardens. It was composed of several species of<br />
palms, tall fruit trees such as mango (Mangifera indica) and avocado (Persea<br />
americana), and timber trees. The vine stratum started at ground level and rose up to<br />
the top of the canopy, with 4% of the total number of species, composed mainly of<br />
tuber-forming vines such as sweet potatoes and several species of yams (Dioscorea<br />
spp.). Many epiphytic species were found on trees and shrubs (De Clerck and<br />
Negreros-Castillo, 2000).<br />
This complex horizontal and vertical structure allows for a variety of agricultural<br />
crops and tree products that are consumed in the household and sold in the local<br />
markets. Multistrata agroforests combining agricultural crops with high-value timber<br />
species, as described in the above example, can provide farmers with long- and<br />
short-term revenue with harvest distributed throughout the year.<br />
In a study of homegardens located in eastern Cuba, Wezel and Bender (2003)<br />
found that species composition and structure were similar to “typical” homegardens
of other regions in Mesoamerica. The top layer (3 to 10 m) consisted mostly of trees<br />
such as avocado, coconut (Cocos nucifera), mango, and breadfruit ( (Artocarpus<br />
communis). In the middle layer (1 to 3 m), smaller trees like guava (Psidium<br />
guajava), soursop ( (Annona muricata),<br />
orange (Citrus sinensis), or papaya (Carica<br />
papaya) were found together with bananas and plantains (Musa spp.), sugarcane<br />
(Saccharum officinarum), pigeon pea (Cajanus cajan), and climber yam (Dioscorea<br />
spp.). In the ground layer (0 to 1 m), different vegetables, spices, and medicinal<br />
plants were cultivated while others grew spontaneously.<br />
3.3. Horizontal structure<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
The horizontal structure of homegardens shows interesting patterns, governed by the<br />
uses/functions of the different plant species. For example, ornamental plants are often<br />
found in linear patterns around the house. They are also found along the roadside of the<br />
garden, reflecting their aesthetic purpose as well as their use for the delineation of<br />
property or sections thereof (Blanckaert et al., 2004). In general, edible plants are found a<br />
little farther away from the house, mostly in small groups to facilitate management such<br />
as weeding or pruning. In semiarid environments such as south-central Mexico<br />
(Blanckaert et al., 2004), central Nicaragua (Méndez et al., 2001), and in the Baitiriqui<br />
region of Cuba (Wezel and Bender, 2003), irrigation is frequently used. In these cases,<br />
edible plants are located downhill from the house and in close proximity to it so that<br />
they can be watered using the wastewater recycled from domestic uses. Medicinal<br />
plants are often found still farther away than ornamental or edible plants (Blanckaert<br />
et al., 2004). <strong>Homegardens</strong> are also important for providing additional living and<br />
working space to supplement small household structures (Lok, 1998).<br />
In an effort to organize and systematize the study of this very complex type of<br />
agroecosystem, many authors have used statistical procedures to group descriptive<br />
characteristics of homegardens. For example, cluster analysis, correspondence<br />
analysis, and diversity indices have been used by several authors to explain the<br />
patterns of variations in floristic composition of the homegardens (Méndez et al.,<br />
2001; Zaldívar et al., 2002; Blanckaert ett al., 2004). These procedures help in the<br />
description of the characteristics of the specific homegardens under study such as<br />
explaining differences in species diversity among homegardens of different<br />
settlements or localities in a region.<br />
4. PLANT SPECIES DIVERSITY IN HOMEGARDENS<br />
OF MESOAMERICA<br />
Results of several studies indicate that homegardens of Mesoamerica are rich in<br />
biodiversity, and need to be considered for in situ conservation and development<br />
programs. Table 2 shows a summary of studies on plant biodiversity in homegardens<br />
of different geographic regions of Mesoamerica. Several of the studies shown in<br />
Table 2 emphasize tree and shrub species and their uses and relevance for forest<br />
conservation, while others focus on the variety of plant species of all life forms and<br />
69
Location Climate Number of Number of plant species Source<br />
homegardens<br />
studied<br />
Tehuacán-Cuicatlán Valley, semiarid to arid 30 233 (66% ornamental, 30% Blanckaert et al., 2004<br />
Puebla, south-central Mexico<br />
Tixpeual and Tixcacaltuyub,<br />
Yucatán, Mexico<br />
<strong>Tropical</strong> forests of nine states,<br />
south-southeast Mexico<br />
Totonac community in<br />
Coxquihui, Veracruz, Mexico<br />
Zona Maya of Quintana Roo,<br />
Yucatán Peninsula, Mexico<br />
Maya community of San Jose,<br />
Toledo district, Belize<br />
El Camalote, Copán, SW<br />
Honduras near the border with<br />
Guatemala<br />
Table 2. Examples of studies on plant diversity of homegardens in Mesoamerica.<br />
tropical humid<br />
lowland<br />
edible, 9% medicinal)<br />
not available 301 trees and shrubs (70% Rico-Gray et al., 1991<br />
medicinal, 40% apiculture, 30%<br />
edible, 17% fuel, 19% building,<br />
12% timber)<br />
not available 278 Toledo et al., 1995<br />
tropical humid<br />
lowland<br />
warm, subhumid, 40 223 Del Angel-Perez and<br />
low elevation<br />
Mendoza, 2004<br />
tropical humid 78 80 De Clerck and Negreros<br />
lowland<br />
Castillo, 2000<br />
tropical humid 18 164 Levasseur and Oliver,<br />
lowland<br />
2000<br />
montane wet 10 253 (91 trees, 42 shrubs 90 House and Ochoa, 1998<br />
herbs, 24 lianas, 2 palms, 2<br />
mushrooms)<br />
70<br />
F. MONTAGNINI
Nicoya, SW Costa Rica tropical seasonal 12 289 (63 varieties) Lok et al., 1998<br />
lowland wet<br />
Five life zones (sensu Holdridge, tropical subhumid 225 236 (excluding ornamentals) Price, 1989<br />
1987) of Costa Rica<br />
to humid<br />
Eastern Costa Rica wet tropical 45 133 Price, 1989<br />
Talamanca, S. Costa Rica wet tropical 83 46 cultivated species Zaldivar et al., 2002<br />
Coto Brus, S. Costa Rica wet tropical 55 27 cultivated species Zaldivar et al., 2002<br />
Masaya, Nicaragua semiarid to arid 20 334 Mendez et al., 2001<br />
Eastern Cuba semiarid 31 101 Wezel and Bender, 2003<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
71
72<br />
their role in sustaining local livelihood needs. In a region with such broad<br />
geographic diversity as Mesoamerica, diversity of plants found in homegardens is<br />
expected to vary according to latitude, elevation, and rainfall. These trends are not<br />
evident from the data shown in Table 2, as similar numbers of species are reported<br />
for wet and for semiarid to arid locations. The number of species reported by the<br />
authors depends on the number of homegardens studied, types of species that were<br />
emphasized, size of the homegardens studied, reliance of homegardens for<br />
subsistence needs, and the traditional uses of the plants, among other factors as<br />
discussed below.<br />
A number of the studies shown in Table 2 also emphasize plant uses and<br />
management. For example, in Yucatán, Mexico, Rico-Gray et al. (1991) reported the<br />
uses of trees and shrubs from the tropical deciduous forests by the Yucatecan Maya.<br />
Despite the lack of important timber species in these forests, the authors conclude<br />
that management could lead to sustainable production of honey, deer, and building<br />
materials for houses. In homegardens of the Tehuacán-Cuicatlán Valley in Puebla,<br />
Mexico (Table 2), plants were categorized into three main groups: cultivated (68%),<br />
protected (10%), or spared (22%) (Blanckaertt et al., 2004). Cultivated plants are<br />
those that are sown or planted by the owner. Protected plants are those that are<br />
encouraged by the farmer, whether they are transplanted from zones outside the<br />
garden or grow spontaneously in the garden. The farmer may choose to protect or<br />
encourage the plant, for example, by supporting it or attaching it to a solid structure,<br />
or by putting stones around the plant. Spared plants are those that spontaneously<br />
grow in the garden and are not removed (Blanckaert et al., 2004).<br />
The high diversity in plant species and uses reported by Blanckaert et al. (2004)<br />
were found at 1217 m above sea level with a climate classified as semiarid to arid<br />
(total annual precipitation 395 mm). Theoretically, these conditions would place the<br />
region at the low end of the spectrum of potential plant species diversity. The most<br />
represented plant families were Cactaceae, Araceae, Liliaceae, Solanaceae, and<br />
Crassulaceae, reflecting the climatic characteristics as well as the preferences of<br />
the local farmers. Members of both Cactaceae and Solanaceae families in the<br />
homegardens are important edible plants. For instance, nopal (Opuntia spp. and<br />
other species of Cactaceae), chilli (Capsicum spp.), and tomato (Lycopersicum<br />
esculentum) (Solanaceae) are important ingredients of the Mexican diet.<br />
A possible explanation for the relatively large diversity of plants found in dry<br />
locations was advanced by Price (1989), who studied the characteristics of<br />
homegardens in five different ecological regions of Costa Rica (Table 2). The author<br />
found that homegardens were most important in regions of dry tropical forests<br />
because socioeconomic conditions were more difficult than in other regions of the<br />
country, making people rely more on homegardens for self-sustenance. In a semiarid<br />
region in eastern Cuba, Wezel and Bender (2003) also reported the importance of<br />
homegardens and their high species diversity (Table 2), with about 50% of the<br />
species consisting of fruit trees.<br />
Locally, plant diversity of homegardens can also be influenced by size of the<br />
homegardens. For example, in Nicoya, Costa Rica, Lok et al. (1998) found that the<br />
size of homegardens ranged from 0.1 to 1.4 ha with an average of 0.5 ha (Table 2).<br />
The smallest homegardens considered in the study had the highest diversity, with<br />
.<br />
F. MONTAGNINI
HOMEGARDENS OF MESOAMERICA<br />
M<br />
205 to 745 species and an average of 348 species per ha. In contrast, the larger<br />
homegardens had only an average of 96 species per ha, with less variability among<br />
gardens in comparison to the smaller homegardens that exhibited higher variability<br />
in species diversity.<br />
4.1. Importance for species domestication and conservation<br />
The high plant species diversity of homegardens in Mesoamerica makes them an<br />
important resource for ethnobotanical studies. Since many species in homegardens<br />
are encouraged or cultivated, the process of domestication of useful species has long<br />
taken place in homegardens. This is true for homegardens in other regions of<br />
the neotropics where they are intensely managed and crops are carefully selected for<br />
specific purposes. For example, the homegardens of Japanese emigrants in the<br />
Tomé-Açu settlement in Pará, in the eastern Amazon region of Brazil, have served<br />
as “banks” of potential crop species that had been gathered and closely observed by<br />
the family members (Yamada and Osaqui, 2006). The homegardens of Tomé-Açu<br />
functioned as individual validation facilities for farmers making decisions about<br />
planting new crops in their farms. Farmers also used homegardens for improvement<br />
and propagation of nursery stock.<br />
Several studies shown in Table 2 emphasize the role of homegardens as sites for<br />
domestication and preservation of useful species (Toledo et al., 1995; House and<br />
Ochoa, 1998; González-Soberanis and Casas, 2004, among others). In El Camalote,<br />
Copán (Honduras), House and Ochoa (1998) found several introduced species along<br />
with native species that belonged to natural forests of the region, and they stressed<br />
the importance of homegardens as genetic banks of ancient crops and as a research<br />
field for new varieties and cultivars. The diversity of traditional vegetables in the<br />
homegardens studied by these authors was outstanding, with many species that are<br />
also present in Guatemala and Mexico but that are absent in other parts of Honduras.<br />
They cite examples of several vegetables and fruits that today are almost exclusively<br />
found in the homegardens. Such is the case of the chayo (Cnidoscolus chaymansa),<br />
a popular green vegetable in Camalote (similar to spinach) but almost absent in the<br />
rest of Honduras. They also cite other unique species of vegetables and fruits that,<br />
again, are found only in the homegardens of Honduras and Guatemala.<br />
Other examples of domestication of crop species can be found in regions such as<br />
the Tehuacán-Cuicatlán valley in central Mexico, where the Maya cultures have a<br />
history of over 10 000 years (González-Soberanis and Casas, 2004). These authors<br />
studied the management and domestication of a fruit of the Sapotaceae family, the<br />
tempesquistle (Sideroxylon palmei). This fruit is consumed and commercialized in<br />
large quantities in the villages studied. Apparently, management of this species in<br />
homegardens has resulted in larger, better quality fruits than those of the wild<br />
populations, demonstrating the importance of domestication of plant species by the<br />
owners and managers of homegardens. This is a good example of a process of<br />
selection by local farmers that may be true for many other species in other homegarden<br />
settings too.<br />
<strong>Homegardens</strong> may have other positive effects on biodiversity, as they can serve<br />
as local refuges for plants and animals that otherwise may be threatened by human<br />
73
74<br />
or natural disturbances. For example, Griffith (2000) reported that during the 1998<br />
fires in Petén, Guatemala, homegardens and other agroforestry systems might have<br />
served as critical refuges for many forest species. Apparently, agroforestry farms<br />
attracted birds by virtue of their complex structure – similar to that of intact forest<br />
patches – they harbor insects, provide nesting sites, and offer protection from<br />
predators (Griffith, 2000). They were also attracted by the cultivated fruit trees,<br />
which may have provided some of the only food sources in the region after fire<br />
destroyed most of the surrounding vegetation. <strong>Homegardens</strong>, thus, can provide<br />
additional services as buffers for protecting local biodiversity in times of stress.<br />
5. SIGNIFICANCE FOR HOUSEHOLD FOOD SECURITY<br />
<strong>Homegardens</strong> can enhance food security in several ways, most importantly through:<br />
(1) direct access to a diversity of nutritionally rich foods, (2) increased purchasing<br />
power from savings on food bills and income from sale of garden products, and (3)<br />
fall-back food provision during periods of temporary food scarcity. In many parts of<br />
the world, homegardens supplement food supply for people, but in some cases,<br />
homegardens can yield basic staples, when they are large enough to plant sufficient<br />
quantities of tubers or cereals (Eibl et al., 2000; Wezel and Bender, 2003). In this<br />
regard, homegardens fulfill a very important social function, especially in a region<br />
like Mesoamerica where poverty and malnutrition co-exist. For example, in the<br />
Maya community of San Jose, Belize, traditional agroforestry systems including<br />
milpa, cacao (Theobroma cacao) under trees, and homegardens almost entirely meet<br />
the family needs for food and wood, and generate 62% of family income (Levasseur<br />
and Oliver, 2000).<br />
In contrast to other types of agroforestry and other productions systems,<br />
homegardens are very important for supplying the household with food products<br />
year-round (Budowski, 1990; Lok, 1998; Eibl et t al., 2000). Their principal<br />
goal is<br />
not to optimize production, as it could be in the rest of the farm, but to guarantee a<br />
minimum supply of different food products at all times of the year, functioning as a<br />
buffer in times of low income and food scarcity. Often, high value products from<br />
homegardens can be sold to purchase staple foods during periods of scarcity. In<br />
Central America, women play an important role in the management, maintenance,<br />
and sale of homegarden food products (Lok, 1998; Howard, 2006).<br />
5.1. Edible plant species<br />
F. MONTAGNINI<br />
As seen in the previous sections, homegardens in Mesoamerica are planted with a<br />
variety of species used for various purposes, including food, medicinal, ornamental,<br />
timber, construction, crafts, among others (Zaldívar et al., 2002) . In addition to their<br />
use for self-sustenance, many studies have indicated that the potential for cash sales<br />
from homegardens is highly important in their composition and management.<br />
Frequently, excess homegarden production is given away to relatives working in<br />
urban areas, thereby supporting food security in both urban and rural areas.
HOMEGARDENS OF MESOAMERICA<br />
M<br />
The importance of homegardens for household food security becomes greater in<br />
more extreme situations of poverty and isolation. In present day Cuba, homegarden<br />
products have contributed additional food to the basic provision such as bread, oil,<br />
flour, meat, and other products sold cheaply in government stores. After 1989, when<br />
the Soviet Union collapsed and dropped aid to Cuba, the economic situation<br />
worsened and food distribution declined precipitously. As it was imperative to find<br />
alternative sources of food supplies, farmers intensified homegarden production in<br />
order to feed their families (Wezel and Bender, 2003).<br />
Similar situations of low income and little assistance by government programs<br />
are common in several countries of Mesoamerica. In Nicaragua, one of the poorest<br />
countries of Central America, Méndez et al. (2001) found that families in Masaya<br />
obtained at least 40 different plant products from their homegardens (Table 2), as<br />
well as the benefit of space for working on handicrafts (a major source of income in<br />
Masaya), and socializing. People enjoyed meeting their neighbors and visitors in the<br />
homegarden because it was a pleasant area of their homes. Although dependence on<br />
homegardens varied according to specific conditions, they seemed to be a consistent,<br />
flexible resource used to meet a diversity of needs, although their main function was<br />
always to provide edible products for household consumption.<br />
Although Costa Rica probably has the best conditions of Mesoamerica in terms<br />
of average per capita income and social welfare programs, rural poverty and<br />
malnutrition persist there, especially among some indigenous groups living in<br />
remote areas. Chibchan Amerindians (Bribris, Cabecares and Guaymis) who live in<br />
reserves located in Talamanca and Coto Brus, in the south-central part of Costa<br />
Rica, practice slash-and-burn agriculture, and maintain polyculture fields or<br />
homegardens adjacent to their dwellings with a high diversity of plants (Zaldívar<br />
et al., 2002; Table 2). Both Bribris and Cabecares have lived in territories within the<br />
Talamanca Reserve for centuries, while the Guaymi migrated about 60 years ago<br />
from their ancestral territories in Panama. Most edible crops common to all<br />
settlements studied by Zaldívar et al. (2002) were native to the region, with the<br />
exception of plantains and bananas, ‘manzana de agua’ (water apple, Syzygium<br />
malaccense), oranges and mangoes. In other regions of Costa Rica also, homegardens<br />
are important for supplying food; they also serve as a buffer in times of<br />
harvest failures or economic depressions (Price, 1989).<br />
In the Chiriquí province of Panama, Lok and Samaniego (1998) found that<br />
among the Ngöbe (or Guaymi) indigenous populations, the homegarden was the<br />
system that provided the largest cash income and number of edible products for<br />
household consumption when compared with other farm activities. They studied 10<br />
farms with an average size of 6.7 ha each, of which about half a hectare was<br />
dedicated to homegardens. The Ngöbe grow annual food crops in plots where they<br />
also grow “fire-hardy” trees. These plots provide the basic food needs of the family<br />
(rice, maize, and beans) during much of the year. In the homegardens they grow<br />
about 100 plant species, of which 75 are woody species (trees, shrubs, and palms).<br />
Among the woody species most of them are fruit trees, including oranges, guayabas,<br />
avocados, and coconuts. Fruits are harvested for household consumption, and often<br />
are the sole source of food for the family in times of scarcity. Fruits are also a source<br />
of food for wildlife, especially birds that the Ngöbe hunt for food. About 80% of<br />
75
76<br />
land inhabited by the Ngöbe is of low productivity and is not suitable for<br />
commercial production of basic grains, as soils are low in organic matter and high in<br />
aluminium content. The cultivation of homegardens is one alternative that the Ngöbe<br />
families have successfully used to offset such edaphic constraints and/or to alleviate<br />
the problem of food shortage.<br />
5.2. Domestic animals<br />
F. MONTAGNINI<br />
Domestic animals are frequently found in the homegardens of Mesoamerica and<br />
Cuba. For example, in the Maya community of San Jose, Belize, poultry and pigs<br />
were found in about 80% of the households (Levasseur and Olivier, 2000).<br />
Likewise, in the Totonac backyard homegardens of Veracruz, Mexico, pigs,<br />
chickens, and small livestock were common (Del Angel-Perez and Mendoza, 2004).<br />
In a survey of 80 homegardens in the dry and humid regions of Costa Rica,<br />
Nicaragua, and Honduras, Wieman and Leal (1998) also noted chickens in 79% of<br />
the homegardens, pigs in 49%, and ducks in 10% of the households. In Cuba,<br />
animals such as pigs, sheep, chickens, and to a lesser extent ducks, rabbits, and<br />
turkeys abound in the homegardens (Wezel and Bender, 2003). Larger farm animals<br />
such as sheep, goats, and cows are often kept tethered on the nearby roadsides to<br />
permit grazing, or sometimes kept in small-fenced paddocks next to the house<br />
(C. Munford, pers. comm., October 2005).<br />
Small animals, in particular, represent a source of production of low-cost protein<br />
in homegardens, especially for the low-income households (Wieman and Leal,<br />
1998). Several small animals such as chickens, ducks, and rabbits also provide<br />
B-complex vitamins and minerals such as iron, calcium, and phosphorus. The small<br />
sizes of these animals also make their care and management, besides meat<br />
preparation (slaughtering, skinning, and cooking) relatively easy. Yet another<br />
advantage is the ease of selling the animals and their products in the local markets<br />
and their year-round production, unlike the orchard plant products which can be<br />
seasonal (Del Angel Pérez and Mendoza, 2004).<br />
Chickens are particularly important in the homegardens of the developing<br />
countries worldwide, primarily for their ability to generate cash income from the<br />
production of eggs, meat, and chicken manure. They also contribute to biological<br />
pest control by preying on insects and grubs, and facilitating household waste<br />
recycling. In the Totonac backyard homegardens of Veracruz, Mexico, chickens<br />
roamed free in about half of homegardens surveyed, although they are often penned<br />
at night (Del Angel-Perez and Mendoza, 2004). The families in Central America<br />
also consumed most of the chicken meat and eggs produced by them. In contrast,<br />
duck meat is not as much appreciated, as ducks are often considered pets. Overall,<br />
the home-raised livestock has high priority among the Totonac farmers, presumably<br />
because of the high value of these animals in the open market.<br />
Similarly, pigs are an important source of meat, despite the seasonality of<br />
production, mostly coinciding with festivities or special occasions. In the homegardens<br />
studied by Wieman and Leal (1998), an average of seven pigs were
HOMEGARDENS OF MESOAMERICA<br />
M<br />
found in the larger homegardens of Limón, Costa Rica and Paraíso, Honduras,<br />
and a smaller number in the smaller-sized homegardens of Masaya, Nicaragua.<br />
Ornamental plant nurseries, wherever present, deterred pig husbandry because of the<br />
potential damage to nursery plants.<br />
In general, local breeds of animals with high resistance to pests and diseases are<br />
used, and women take care of the animals (Lok and Samaniego, 1998). Whenever<br />
the domestic animals are likely to interfere with the cultivation of plants within the<br />
homegardens, they are enclosed or tied up. In the orchards dominated by trees, pigs<br />
and chickens roam freely, suggesting that the farmers disregard the understorey<br />
vegetation, while backyards in town often have animals in cages or in enclosed<br />
quarters to protect ornamental, medicinal, condiment, and ritual plants.<br />
5.3. Promotion of homegardens in food security and development projects<br />
<strong>Homegardens</strong> have long been used as a tool to promote household food security in<br />
many regions of the world, and especially as part of many educational and<br />
dissemination efforts by international aid agencies, local governments and nongovernment<br />
organizations (NGOs). For example, FAO has produced materials for<br />
their training package ‘Improving Nutrition through Homegardening’ (FAO 2001),<br />
featuring homegardens for food security in many regions of the world, including<br />
specific projects in Nicaragua, El Salvador, and Honduras. In Nicaragua,<br />
government subsidies, in combination with international aid, have been used for<br />
decades to promote homegardens as a means to guarantee basic household food<br />
security. For example, the Plan Alimentario Nacional (National Food Plan) with<br />
financial support from foreign-aid and local logistic and technical support from<br />
NGOs working in the region, has distributed seeds, working tools, and cooking<br />
utensils to families in need, mostly from the rural areas of semiarid regions<br />
(El Nuevo Diario, Managua, Nicaragua, April 3, 2002). Similar promotion of<br />
homegardens to alleviate poverty and ensure basic food supply in rural and urban<br />
areas is underway in Panama and El Salvador, again supported by local NGOs<br />
and international assistance (e.g., Food Safety Program in Tacuba, El Salvador,<br />
sponsored by World Vision, Canada). In Nicaragua, the Peace Corps of the USA<br />
established the Food Security Program after Hurricane Mitch in 1998, while other<br />
organizations such as the Red Cross integrated homegarden projects into larger ones<br />
directed to address the post-Mitch needs including natural disaster mitigation efforts<br />
(D. Craven, pers. comm., October 2005).<br />
In several locations of Mesoamerica, homegardens are often grown and managed<br />
as part of the communal development efforts. For example, in Diriamba, Nicaragua,<br />
community homegardens form part of a larger development program (POSAF,<br />
Program for Agroforestry Development and Environment) funded by the World<br />
Bank (Piotto et al., 2004). Similarly, in El Salvador and Nicaragua, homegardens are<br />
components of community development efforts in coffee <strong>coop</strong>eratives. They are<br />
assisted by local NGOs working on rural development and biodiversity conservation<br />
(Méndez and Bacon, 2005).<br />
77
78<br />
F. MONTAGNINI<br />
6. NUTRIENT CYCLING<br />
Efficient nutrient cycling is a key to the ecological sustainability of traditional<br />
homegardens, and species and structural diversity are critical to maintaining it<br />
through optimum use and transfer of carbon, water, and nutrients. Many traditional<br />
homegardens in Mesoamerica have survived for centuries despite many ecological,<br />
social, and political changes, justifying the claim that they are a sustainable land<br />
use system (e.g., the Maya homegardens of Yucatán Peninsula; Caballero, 1992).<br />
However, this cannot be generalized to all systems practiced by traditional peoples<br />
of the region. A comparison of land use and land clearing by Maya descendants and<br />
Hispanic populations in the Sierra de Lacandón National Park in Petén, Guatemala,<br />
is a case in point; not only agricultural land use by these two groups is very similar<br />
but also the impacts on land clearing are comparable (Carr, 2004). Population<br />
pressure and changes in other socioeconomic conditions thus strongly influence<br />
nutrient management and recycling, affecting the sustainability of homegardens. Yet<br />
many traditional societies retain the conventional wisdom on sustainable land<br />
management.<br />
In a study conducted in the northwestern and north-central regions of the<br />
Yucatán Peninsula of Mexico, Benjamin et al. (2001) hypothesized that Mayan<br />
farmers have been choosing tree associations and garden structures that maximize<br />
productivity and optimize nutrient cycling of the homegardens. At present, however,<br />
the Maya have ceased to use many of their earlier technologies that improved<br />
production, e.g., using raised beds and muck. Nevertheless, ‘modern’ Mayan<br />
homegardens still maintain relatively high yields using technologies of nutrient<br />
recycling such as mulching for residue management and fertilization (Benjamin<br />
et al., 2001). Soils in the region are very thin and contain rocks and calcium<br />
carbonates due to the shallow limestone bedrock. Low annual precipitation results in<br />
depleted surface and ground water resources, making large-scale irrigation a nonviable<br />
option. Benjamin et al. (2001) also noted that the Maya recognize appropriate<br />
tree species for such sites and know their growth characteristics; they also have<br />
the knowledge on appropriate nutrient management practices, which are applied in<br />
the design and management of homegardens. Irrigation timing, pruning, addition of<br />
ash to soils, and composting are some of the practices that Maya farmers use to<br />
enhance tree growth and survival, resulting in high fruit production with less<br />
investment in leaf biomass.<br />
Sweeping and burning of litter in homegardens results in the export of<br />
substantial amounts of nutrients, decreasing the effectiveness of nutrient cycling.<br />
Ash is recycled in the homegardens, although not uniformly. However, soils had<br />
high concentrations of organic matter. If litter were not removed, potential nitrogen<br />
contributions from litter to the homegardens would be very high (Benjamin et al.,<br />
2001).<br />
Nutrient addition through the litter of nitrogen-fixing species is also a practice<br />
used in many homegardens in Mesoamerica. The Maya communities of San Jose,<br />
Belize, use the litter of Gliricidia sepium, a tree species native to Mesoamerica, to<br />
fertilize their homegardens (Levasseur and Oliver, 2000). In addition, practices that<br />
are aimed to controlling soil erosion also contribute to nutrient recycling through
HOMEGARDENS OF MESOAMERICA<br />
M<br />
soil and nutrient conservation. The Totonacs in Coxquihui, Veracruz, Mexico,<br />
perceive soil loss as the most serious hazard to their traditional homegardens, and<br />
therefore, have sought to control erosion by retaining a continuous canopy cover,<br />
and using litter for mulching among other soil conservation practices (Del Angel-<br />
Pérez and Mendoza, 2004).<br />
The small size of homegardens allows for the application of intensive<br />
management practices that can improve nutrient recycling and lead to higher<br />
productivity. In Tacuba, El Salvador, farmers often open small trenches (about 30<br />
cm deep, few meters long, and set perpendicular to the direction of the slope) in<br />
their homegardens (Fig. 3). They drop household residues as well as prunings and<br />
other organic materials in the trenches. This avoids losses of residues that otherwise<br />
could be washed down the slope during the rains. They change the location of the<br />
trenches in the area of the homegarden so that eventually residues are recycled all<br />
over the homegarden area (pers. obs.).<br />
Figure 3. Recycling of household residue in homegardens in Tacuba, El Salvador. Farmers<br />
dig small trenches set perpendicular to the direction of the slope, where they deposit<br />
household residues, prunings and other organic material, to avoid losses of residues down the<br />
slope.<br />
79
80<br />
F. MONTAGNINI<br />
Manure of small animals is valuable as a nutrient source for the homegardens.<br />
This may be a localized effect as chickens or pigs often wander free in portions of<br />
the homegarden and their manure falls near cultivated plants. It can also be part of a<br />
specific management strategy, as chicken manure in regions of Costa Rica is used to<br />
fertilize small patches planted with corn that is used to feed the chickens as well as<br />
other animals of the homegarden (author’s pers. obs.).<br />
Vermiculture, or growing earthworms in worm boxes to use the castings for<br />
fertilizing homegarden soils, is used in many parts of Central America to increase<br />
the productivity of vegetable gardens and fruit trees. The high production of worm<br />
castings by certain earthworm species (e.g., the red Californian earthworm,<br />
Allophora caliginosa) is a source of cheap fertilizer for staple crops such as corn and<br />
sorghum (Sorghum bicolor). In rural areas of Nicaragua, some families sell the<br />
worm castings as organic manure (D. Craven, pers. comm., November 2005).<br />
As mentioned above, in Mesoamerica the traditional agricultural knowledge<br />
existing in many regions that still carry the influence of ancient Mayan indigenous<br />
peoples includes management practices that improve nutrient cycling. Some<br />
management practices can be redirected or improved to optimize plant productivity<br />
in homegardens. Improved litter management and knowledge of the relative nutrient<br />
content of the litter from different species when used as mulch or compost may be<br />
one avenue for improving both water and nutrient cycling and homegarden<br />
production. Composting of homegarden litter, instead of burning it, would augment<br />
the amounts of nutrients recycled in the system. Water retention, by adding organic<br />
matter via compost, would help to improve water availability for plants, especially<br />
important in homegardens located in subhumid and semiarid regions of Mesoamerica.<br />
Long-rotation production systems such as agroforests and homegardens can also<br />
sequester sizeable quantities of carbon in plant biomass and in long-lasting wood<br />
products (Albrecht and Kandji, 2003; Montagnini and Nair, 2004; Kumar, 2006).<br />
Many of the traditional homegardens already described share ecological characteristics<br />
and management practices that make them efficient in the use of solar<br />
radiation and carbon, and allow high levels of productivity. Soil carbon sequestration<br />
constitutes another realistic option achievable in homegardens.<br />
7. CONCLUSIONS<br />
The region of Mesoamerica suffers from social and environmental problems due to<br />
overpopulation and rural poverty. Under such conditions, homegardens have<br />
traditionally fulfilled and still provide an important function in terms of ensuring a<br />
basic food supply for the family. This is especially important in the remote areas<br />
such as in indigenous reserves or in other rural settings in the relatively more<br />
impoverished countries of the region.<br />
Mesoamerican homegardens are quite diverse, with a complex vertical and<br />
horizontal structure that includes plants for food, ornamental, medicinal and other<br />
purposes. Mesoamerican homegardens are important reservoirs of local biodiversity<br />
and have a prominent role in the domestication of useful species.
HOMEGARDENS OF MESOAMERICA<br />
M<br />
Domestic animals in homegardens of Mesoamerica contribute to increased food<br />
security. Animal manure also contributes to nutrient recycling. The inclusion of<br />
domestic animals in homegardens is vital to ensure a more sustained protein supply.<br />
However, they require an investment for the care and management of animals that<br />
would be relatively large for the poor, rural households.<br />
Mesoamerica was the home of the ancient Maya civilization, whose descendants<br />
still practice sustainable agriculture and manage homegardens in ways that increase<br />
the efficiency of the capture of solar radiation, increase productivity and improve<br />
nutrient cycling. Soil organic matter in homegardens can be increased by several<br />
practices of residue management. It is important that such sustainable management<br />
practices be retained to ensure homegarden sustainability. <strong>Homegardens</strong> of Mesoamerica<br />
also contribute to environmental services such as carbon sequestration, even<br />
though globally their role may be minimal due to their small land area.<br />
ACKNOWLEDGEMENTS<br />
Dylan Craven, Philip Marshall, Quint Newcomer, and Tatsuhiro Ohkubo provided<br />
useful comments and insights to this manuscript. Dylan Craven also helped with the<br />
literature review and editing.<br />
ENDNOTE<br />
1. CCAD (Comisión Centroamericana de Ambiente y Desarrollo). 2003. Estado<br />
del Sistema Centroamericano de Areas Protegidas: informe de síntesis regional.<br />
Comisión Centroamericana de Ambiente y Desarrollo (Central American<br />
Comisión for Environment and Development). San José, Costa Rica. 33p.<br />
REFERENCES<br />
Albrecht A. and Kandji S.T. 2003. Carbon sequestration in tropical agroforestry systems.<br />
Agric Ecosyst and Environ 99: 15 – 27.<br />
Atran S. 1993. Itza Maya tropical agroforestry. Curr Anthropol 34: 633 – 700.<br />
Atran S., Medin D., Ross N., Lynch E., Coley J., Ucan Ek’ E., and Vapnarsky V. 1999. Folk<br />
ecology and commons management in Maya lowlands. Proc Natl Acad Sci USA 96:<br />
7598 – 7603.<br />
Barrera A., Gómez-Pompa A., and Vázquez-Yanes C. 1977. El manejo de las selvas por los<br />
Mayas: sus implicaciones silvícolas y agrícolas. Biotica 2(2): 47 – 61.<br />
Beach T. and Dunning N.P. 1995. Ancient Maya terracing and modern conservation in the<br />
Peten rain forest of Guatemala. J Soil Water Conservation 50: 138 – 145.<br />
Benjamin T.J., Montañez P.I., Jiménez J.J.M. and Gillespie A.R. 2001. Carbon, water and<br />
nutrient flux in Maya homegardens in the Yucatán Península of Mexico. Agroforest Syst<br />
53: 103 – 11.<br />
Blanckaert I., Swennen R.L., Paredes Flores M., Rosas López R., and Lira Saade R. 2004.<br />
Floristic composition, plant uses and management practices in homegardens of San Rafael<br />
Coxcatlán, Valley of Tehuacán, Mexico. J Arid Environ 57: 39 – 62.<br />
Budowski G. 1990. Home gardens in <strong>Tropical</strong> America: a review. In: Landauer K. and Brazil<br />
M. (eds), <strong>Tropical</strong> home gardens, pp 3 – 8. United Nations University, Tokyo.<br />
81
82<br />
F. MONTAGNINI<br />
Caballero J. 1992. Maya homegardens: past, present and future. Ethnoecologica 1(1): 35 – 54.<br />
Carr, D.L. 2004. Ladino and Q’eqchi Maya land use and land clearing in the Sierra de<br />
Lacandon National Park, Peten, Guatemala. Agric Hum Values 21: 67 – 76.<br />
Casas A., Valiente-Baunet A., Viveros J.L., Dávila P., Lira R., Caballero J., Cortés L., and<br />
Rodríguez I. 2001. Plant resources of the Tehuacán Valley, Mexico. Econ Bot 55:<br />
129 – 166.<br />
De Clerck F.A.J. and Negreros-Castillo P. 2000. Plant species of traditional homegardens of<br />
Mexico as analogs for multistrata agroforests. Agroforest Syst 48: 303 – 317.<br />
Del Angel-Perez A.L. and Mendoza M.A. 2004. Totonac homegardens and natural resources<br />
in Veracruz, Mexico. Agric Hum Values 21: 329 – 346.<br />
De Las Salas G. 1987. Suelos y ecosistemas forestales con énfasis en América tropical.<br />
Instituto Interamericano de Cooperación paraa la Agricultura (IICA), San José, 479p.<br />
Dommergues Y.R. 1987. The role of biological nitrogen fixation in agroforestry. In: Steppler<br />
H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development, pp 245 – 272.<br />
ICRAF, Nairobi.<br />
Eibl B., Fernández R., Kozarik J.C., Lupi A., Montagnini F. and Nozzi D. 2000. Agroforestry<br />
systems with Ilex paraguariensis (American Holly or yerba mate) and native timber trees<br />
on small farms in Misiones, Argentina. Agroforest Syst 48: 1 – 8.<br />
Food and Agriculture Organization (FAO) 2001. Improving nutrition through home<br />
gardening. A training package for preparing field workers in Southeast Asia. Food and<br />
Agriculture Organization of the United Nations, Rome, 171p.<br />
Fedick S.L. and Morrison B.A. 2004. Ancient use and manipulation of landscape in the<br />
Yalahau region of the northern Maya lowlands. Agric Hum Values 21: 207 – 219.<br />
Gillespie A.R., Knudson D.M. and Geilfus F. 1993. The structure of four home gardens in the<br />
Peten, Guatemala. Agroforest Syst 24: 157 – 170.<br />
González-Soberanis C. and Casas A. 2004. Traditional management and domestication of<br />
tempesquistle, Sideroxylon palmeri (Sapotaceae) in the Tehuacan-Cuicatlán Valley,<br />
Central México. J Arid Environ 59: 245 – 258.<br />
Griffith D.M. 2000. Agroforestry: a refuge for tropical biodiversity after fire. Conserv Biol<br />
14: 325 – 326.<br />
Guindon C. 1996. The importance of forest fragments to the maintenance of regional<br />
biodiversity in Costa Rica. In: Schelhas J. and Greenberg R. (eds), Forest patches in<br />
tropical landscapes, pp 168 – 186. Island Press, Washington, D.C.<br />
Harvey C.A. and Haber W.H. 1999. Remnant trees and the conservation of biodiversity in<br />
Costa Rican pastures. Agroforest Syst 44: 37 – 68.<br />
Harvey C., Alpizar F., Chacón M. and Madrigal R. 2005. Assessing linkages between<br />
agriculture and biodiversity in Central America. Historical overview and future<br />
perspectives. Mesoamerican and Caribbean region, Conservation Science Program. The<br />
Nature Conservancy (TNC), San José, 138p.<br />
Holdridge L.R. 1987. Ecología basada en Zonas de Vida. Instituto Interamericano de<br />
Cooperación para la Agricultura (IICA), San José, 216p.<br />
House P.H. and Ochoa L. 1998. La diversidad de especies útiles en diez huertos en la aldea de<br />
Camalote, Honduras. In: Lok R. (ed.), Huertos caseros tradicionales de América Central:<br />
características, beneficios e importancia, desde un enfoque multidisciplinario, pp 61 – 84.<br />
Centro Agronómico <strong>Tropical</strong> de Investigación y Enseñanza (CATIE), Turrialba.<br />
Howard P.L. 2006. Gender and social dynamics in swidden and homegardens in Latin<br />
America In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 159 – 182. Springer Science, Dordrecht.<br />
Kumar B.M. 2006. Carbon sequestration potential of tropical homegardens In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 185 – 204. Springer Science, Dordrecht.
Levasseur V. and Olivier A. 2000. The farming system and traditional agroforestry systems in<br />
the Maya community of San Jose, Belize. Agroforest Syst 49: 275 – 288.<br />
Lok R. 1998. El huerto casero tropical tradicional en América Central. In: Lok R. (ed.),<br />
Huertos caseros tradicionales de América Central: características, beneficios e importancia,<br />
desde un enfoque multidisciplinario, pp 7 – 28. Centro Agronómico <strong>Tropical</strong> de<br />
Investigación y Enseñanza (CATIE), Turrialba.<br />
Lok R. and Samaniego G. 1998. La valorización sociocultural del huerto y del café con<br />
árboles entre la población Ngöbe de Chiriquí, Panamá. In: Lok R. (ed.), Huertos caseros<br />
tradicionales de América Central: características, beneficios e importancia, desde un<br />
enfoque multidisciplinario, pp 185 – 213. Centro Agronómico <strong>Tropical</strong> de Investigación y<br />
Enseñanza (CATIE), Turrialba.<br />
Lok R., Wieman A. and Kass D. 1998. Influencia de las características del sitio y el acceso al<br />
agua en huertos de la Península de Nicoya, Costa Rica. In: Lok R. (ed.), Huertos caseros<br />
tradicionales de América Central: características, beneficios e importancia, desde un<br />
enfoque multidisciplinario, pp. 29 – 59. Centro Agronómico <strong>Tropical</strong> de Investigación y<br />
Enseñanza (CATIE), Turrialba.<br />
Méndez V.E. and Bacon C. 2005. Medios de vida y conservación de la biodiversidad arbórea:<br />
las experiencias de las <strong>coop</strong>erativas cafetaleras en El Salvador y Nicaragua. LEISA<br />
Revista de Agroecologia 20 (4): 27 – 30.<br />
HOMEGARDENS OF MESOAMERICA<br />
M<br />
Méndez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Miller R.P., Penn Jr. J.W. and van Leeuwen J. 2006. Amazonian homegardens: their<br />
ethnohistory and potential contribution to agroforestry development. In: Kumar B.M. and<br />
Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agro-<br />
forestry, pp 43 – 60. Springer Science, Dordrecht.<br />
Montagnini F. and Nair P.K.R. 2004. Carbon sequestration: An under-exploited<br />
environmental benefit of agroforestry systems. Agroforest Syst 61: 281 – 295.<br />
Myers N., Mittermeier R.A., Mittermeier C.G., da Fonseca G.A.B. and Kent J. 2000.<br />
Biodiversity hotspots for conservation priorities. Nature 403: 853 – 858.<br />
National Academy of Sciences (NAS). 1979. <strong>Tropical</strong> legumes: Resources for the future.<br />
Board of Science and Technology for International Development, National Research<br />
Council, Washington, DC, 332p.<br />
National Academy of Sciences (NAS). 1980. Firewood crops. Shrubs and tree species for<br />
energy production. Board of Science and Technology for International Development,<br />
National Research Council, Washington, DC, 236p.<br />
Noble I.R and Dirzo R. 1997. Forests as human-dominated ecosystems. Science 277:<br />
522 – 525.<br />
Piotto, D., Montagnini F., Kanninen M., Ugalde L. and Viquez E. 2004. Forest plantations in<br />
Costa Rica and Nicaragua: performance off species and preferences of farmers.<br />
J Sustainable For 18: 59 – 77.<br />
Price N.W. 1989. The tropical mixed garden in Costa Rica. PhD Thesis. University of British<br />
Columbia, Vancouver, 403p.<br />
Rico-Gray V., Chemas A. and Mandujano S. 1991. Uses of tropical deciduous forest species<br />
by the Yucatecan Maya. Agroforest Syst 14 (2): 149 – 161.<br />
Richards P.W. 1996. The tropical rainforest: An ecological study (2 nd ed.). Cambridge<br />
University Press, London, 450p.<br />
Sanchez P.A. 1976. Properties and management of soils in the tropics. John Wiley and Sons,<br />
New York, 618p.<br />
83
84<br />
F. MONTAGNINI<br />
Steinberg M.K. 1998. Political ecology and cultural change: impacts on swidden-fallow<br />
agroforestry practices among the Mopan Maya in southern Belize. Prof Geogr 50:<br />
407 – 417.<br />
Toledo V.M., Batis A.I., Becerra R. Martinez E. and Ramos C.H. 1995. The useful forest:<br />
quantitative ethnobotany of the indigenous groups of the humid tropics of Mexico.<br />
Interciencia 20: 177 – 187.<br />
Turner II B.L. and Harrison P.D. 1981. Prehistoric raised-field agriculture in the Maya<br />
lowlands. Science 213: 399 – 405.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
Wieman A. and Leal D. 1998. La cría de animales menores en los huertos caseros. In: Lok R.<br />
(ed.), Huertos caseros tradicionales de América Central: características, beneficios e<br />
importancia, desde un enfoque multidisciplinario, pp 85 – 115. Centro Agronómico<br />
<strong>Tropical</strong> de Investigación y Enseñanza (CATIE), Turrialba.<br />
Yamada M. and Osaqui H.M.L. 2006. The role of homegardens for agroforestry development:<br />
lessons from a Japanese-Brazilian settlement in the Amazon. In: Kumar B.M. and Nair<br />
P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry,<br />
pp 299 – 316. Springer Science, Dordrecht.<br />
Zaldivar M.E., Rocha O.J., Castro E. and Barrantes R. 2002. Species diversity of edible<br />
plants grown in homegardens of Chibchan Amerindians from Costa Rica. Hum Ecol 30:<br />
301 – 316.
SECTION 2<br />
STRUCTURE, FUNCTION, AND DYNAMICS<br />
OF HOMEGARDENS
CHAPTER 6<br />
HOMEGARDEN DYNAMICS<br />
IN KERALA, INDIA<br />
A. PEYRE 1,2 , A. GUIDAL 1,2 , K.F. WIERSUM 2 *, AND<br />
F. BONGERS 1<br />
1 Forest Ecology and Forest Management group and 2 Forest and Nature<br />
Conservation Policy group, Wageningen University, The Netherlands; *E-mail:<br />
<br />
Note: Adapted from: Peyre A., Guidal A., Wiersum K.F. and Bongers F. 2006. Dynamics of<br />
homegarden structure and function in Kerala, India. Agroforestry Systems 66: 101 – 115.<br />
Keywords: Biodiversity, Functional differentiation, Multispecies systems, Social adaptability,<br />
Socioeconomic change.<br />
Abstract. <strong>Homegardens</strong> in Kerala, India, have long been important multipurpose agroforestry<br />
systems that combine ecological and socioeconomic sustainability. These traditional<br />
homegardens, however, are subject to changes consequent to various on-going socioeconomic<br />
transformations. The study of structural and functional dynamics of homegardens offers an<br />
opportunity to understand the trends in socioeconomic sustainability in relation to their<br />
ecological sustainability. These dynamics were studied in a survey of 30 homegardens. Based<br />
on a cluster analysis of tree/shrub species density and a subsequent grouping using<br />
homegarden size, six homegarden types were differentiated, and these were assessed for<br />
structural, functional, and managerial characteristics, besides their dynamics. Four<br />
development stages of homegardens were found along a gradient from traditional to modern<br />
homegardens. Fifty percent of the homegardens still displayed traditional features, whereas<br />
33% incorporated modern practices. The process of modernization includes a decrease of the<br />
tree/shrub diversity, a gradual concentration on a limited number of cash crop species, an<br />
increase of ornamental plants, a gradual homogenization of homegarden structure and an<br />
increased use of external inputs. A traditional homegarden combining multispecies<br />
composition and intensive management practices could, however, offer an alternative<br />
development path to modern homegardens in adapting homegardens to changing<br />
socioeconomic conditions.<br />
87<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 87–103.<br />
© 2006 Springer. Printed in the Netherlands.
88<br />
A. PEYRE ET AL.<br />
1. INTRODUCTION<br />
<strong>Homegardens</strong> are recognized worldwide as an epitome of a sustainable agroforestry<br />
system (Torquebiau, 1992; Kumar and Nair, 2004). From a system-dynamics point<br />
of view, the concept of sustainability includes two main dimensions (Wiersum,<br />
1995): ecological sustainability (in the sense of keeping within ecological stability<br />
domains) and social sustainability (in the sense of adjusting to social dynamics).<br />
Most studies on sustainability of homegardens have been focused on ecological<br />
sustainability, while social sustainability has been given much less systematic<br />
attention (Torquebiau, 1992; Kumar and Nair, 2004). Social sustainability may<br />
relate either to the social acceptability of homegardens within the livelihood systems<br />
of rural producers or to the ability of homegardens to adjust to socioeconomic<br />
changes. The structure and composition of homegardens can well be adjusted to<br />
various livelihood conditions such as size of landholdings, role of homegardens<br />
within the overall farming-system and degree of commercialization. However,<br />
homegardens are not static, but have evolved over centuries; thanks to adaptive<br />
abilities of farmers in responding to changing rural and livelihood conditions<br />
(Wiersum, 2006). Traditionally, the homegardens mainly served to produce<br />
vegetables, fruits and other crops, which supplemented the staple food crops<br />
produced on open croplands. With the advent of commercialization, often an<br />
increase in selected cash crops such as coconut (Cocos nucifera) or rubber (Hevea<br />
brasiliensis) has been observed. The shift from subsistence agriculture to market<br />
economy often implies drastic structural and functional modifications, including a<br />
homogenization of the homegarden structure and use of external inputs (Kumar and<br />
Nair, 2004).<br />
Several authors have voiced concern that these developments result in the loss of<br />
relevance of the homegardens and threaten their future development. Recently the<br />
question was even raised whether the homegardens are becoming irrelevant or even<br />
extinct (Kumar and Nair, 2004). The expressed fears that the traditional, diverse and<br />
ecologically sustainable homegardens will gradually dissolve into monospecific<br />
agricultural systems with uncertain sustainability are in stark contrast to the earlier<br />
ideas on homegardens as having a promising future (Soemarwoto, 1987). The<br />
maintenance of multispecies and multistrata agroforests is deemed worthwhile<br />
because of the growing interest in developing multifunctional land use systems,<br />
which contribute not only to production objectives, but also to the objectives of<br />
biodiversity and environmental conservation. In order to maintain the positive<br />
characteristics of the traditional homegardens it is therefore necessary to develop<br />
improved homegardens that counterbalance the ongoing trend of homogenization.<br />
In order to better understand whether there is scope for such an alternative<br />
development path, and whether it is possible to adapt homegarden systems to the<br />
changing rural conditions while still maintaining the positive features of the<br />
traditional homegardens, it is necessary to study the trends in homegarden dynamics<br />
in detail. Up until the present, most homegarden studies have focused mainly on<br />
species inventories or system description (Nair, 2001) and still little attention has<br />
been given to their structural and functional evolution. In the past, differences<br />
between homegardens were mostly described based on characteristics such as size,
structure (vertical stratification, diversity indices) or socioeconomic factors (level of<br />
inputs, subsistence/commercial production). Only recently, studies have been undertaken<br />
to systematically classify the structure of homegardens using analytical<br />
methods such as cluster analysis common to vegetation science (Leiva et al., 2002;<br />
Quiroz et al., 2002; Mendez et al., 2001; Tesfaye Abebe, 2005). These methods<br />
offer good opportunities for obtaining a systematic insight into different types of<br />
homegardens. The further evaluation of these different types in respect to socioeconomic<br />
conditions, under which they evolved, can provide useful insights into the<br />
development trends of homegardens.<br />
<strong>Homegardens</strong> have traditionally been managed and adopted by farmers rather<br />
than through agroforestry research (Nair, 2001). Consequently, an interesting<br />
question is whether all farmers are following similar homegarden development<br />
trends, or whether farmers are following different pathways in maintaining their<br />
homegardens. The recent advances in using statistical methods for classification of<br />
homegarden systems provides a good basis for assessing whether there exist<br />
differences in homegarden types and evaluating whether different types follow<br />
different development trends.<br />
Based on these considerations, the present study was undertaken with the<br />
objective of assessing the nature of dynamics of homegarden characteristics. It<br />
focused on the following questions:<br />
1. What different types of homegarden are present in the study area and what<br />
are their characteristics?<br />
2. What changes in homegarden structure, function, and management<br />
characteristics took place during the past decade?<br />
3. What conclusion can be drawn regarding the position of the different<br />
homegarden types on an evolutionary axis?<br />
2.1. Study site<br />
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 89<br />
2. MATERIALS AND METHODS<br />
The study was conducted in Palakkad district, Kerala, India, which is one of the<br />
tropical regions where concerns about the future of homegardens have been raised 1 .<br />
In this region, the value of homegardens as multipurpose production systems<br />
combining ecological and socioeconomic sustainability is well-recognized (Nair and<br />
Sreedharan, 1986; Jose and Shanmugaratnam, 1993; Kumar et al., 1994). Various<br />
authors have voiced concern that socioeconomic changes and related adoption of<br />
modern managerial systems cause a negative conversion process of homegardens in<br />
this region (Jose and Shanmugaratnam, 1993; John and Nair, 1999). Several<br />
government development programs want to assist the farmers to raise their cash<br />
incomes and therefore promote the conversion of homegardens towards cashcropping<br />
systems by providing loans and subsidies for rubber or other cash-crop<br />
cultivation. Moreover, government controls on timber production discourage the<br />
growing of timber in homegardens.
90<br />
In view of these developments, Kerala offers a good opportunity to study the<br />
development trends in agroforestry systems. The case study was carried out in two<br />
panchayaths (administrative units: Mundur and Puduparyaram) of Palakkad district<br />
in central Kerala. The region is characterized by a tropical humid climate with a<br />
monsoonal pattern of rainfall. The topography is rolling to hilly and main soil types<br />
are Dystric Nitosols (FAO, 1977).<br />
2.2. Data collection<br />
Within the two study panchayaths, a stratified sample of 30 farm households was<br />
selected. The sample was stratified according to total landholding size, i.e., small<br />
(< 1 ha; n = 10), medium (1 to 2 ha; n = 10) and large (> 2 ha; n = 10). The<br />
households were selected based on information from a local rural development<br />
organization (Integrated Rural Technology Centre, Mundur, Palakkad) and from<br />
referrals of initial respondents.<br />
In the homegardens of each household, a detailed survey of the composition and<br />
management practices was made. The survey consisted of an inventory of tree and<br />
shrub species and a count of all individuals per species. Only presence was recorded<br />
for herbs and (bi)-annuals. The species were classified according to their use into the<br />
categories: fruits and nuts, staple food, beverages and stimulants, spices, timber and<br />
firewood, medicinal products, religious plants, ornamentals, multipurpose species<br />
with more than four uses, and others. Rubber was the only species used exclusively<br />
as a cash crop and classified as such. During the survey, information was also<br />
collected on the management practices for individual species based on the approach<br />
developed by Wiersum and Slingerland 2 . In this approach, five main practices are<br />
distinguished: controlled utilization, protection, and maintenance, stimulation of<br />
desired products, regeneration, and interface management. The first four categories<br />
represent an increasing input of human energy per unit of land (Wiersum, 1997).<br />
Additional information concerning the homegarden size, the overall strategy in<br />
homegardens orientation (subsistence or commercial) and management inputs was<br />
collected using structured interviews. In these interviews, additional information<br />
was collected on changes in homegarden structure and management during the<br />
preceding ten years. This concerned both changes in homegarden composition and<br />
spatial arrangements (including homegarden size) as well as in management<br />
practices (changes in vegetation structure, production characteristics, and chemical<br />
input use).<br />
2.3. Data processing and analysis<br />
A.PEYRE ET AL.<br />
A hierarchical cluster analysis was applied for classification of the 30 homegardens<br />
using tree/shrub species density (number of f individuals per species per unit area) as
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 91<br />
the main variable. Chi-square was used as distance or similarity measure and<br />
between-group average linkage method. Nine clusters were distinguished of which<br />
five consisted of only one homegarden each. Those five “single” clusters were<br />
reclassified based on homegarden size into two new types. Thus, a group of four<br />
small homegardens and a “group” of one big homegarden emerged. These six<br />
homegarden types were assessed with respect to their structural, functional and<br />
management characteristics as well as dynamics.<br />
Structural characteristics: Four parameters were used to assess the structural<br />
attributes: homegarden size (land area including the house), total density of trees per<br />
homegarden, and species richness and evenness (except for species that could not be<br />
counted)—computed using Shannon’s and Simpson’s diversity indices (following<br />
Huston, 1994). Since only three of the six types had a sufficient number of<br />
homegardens, statistical analysis was only applied to differences among these<br />
homegarden types. Differences were tested using ANOVA for all the parameters<br />
except for number of species and tree density, as populations were not normally<br />
distributed even after transformation. In this case, a non-parametric Kruskall and<br />
Wallis test was applied.<br />
Functional characteristics: Two parameters were used to assess the functional<br />
characteristics: the proportions of mean number of trees per use-category and the<br />
differentiation in home- or cash-orientation in production. Relative contribution of<br />
each use group was calculated and compared within each homegarden type. Annual<br />
staple food crops and ‘other’ crops were not included in this analysis; coconut and<br />
rubber were treated as separate categories in view of their high value according to<br />
both farmers’ opinions and actual situation.<br />
Management characteristics: Management was characterized in respect to<br />
management intensity, spatial arrangement, and use of inputs. Assessment of the<br />
management intensity was based on a detailed assessment of the management<br />
practices for the seven most common and preferred species: rubber, coconut, arecanut<br />
(Areca ( catechu),<br />
mango (Mangifera indica), jackfruit (Artocarpus ( heterophyllus),<br />
teak<br />
(Tectona grandis) and neem ( (Azadirachta indica).<br />
It was characterized on a<br />
comparative scale according to the technique of Wiersum and Slingerland 2 . The<br />
characterization of management inputs was based on an assessment of the internal<br />
and external inputs applied in cultivating the seven tree species.<br />
Homegarden dynamics: The dynamics of each homegarden type were qualitatively<br />
assessed according to the changes in homegarden size, vegetation structure (introduction<br />
of new species, changes in respect to ornamental and medicinal plants, and<br />
changes in spatial arrangements), and production characteristics (change of homegarden<br />
orientation and evolution of chemical input use).<br />
The data were analyzed using the statistical package SPSS 10.0 (SPSS Inc.).<br />
Based on structure, functions, management, and dynamics, the different homegarden<br />
types were arranged along a gradient from traditional l to modern homegardens.
92<br />
3.1. Distinction in homegarden types<br />
A. PEYRE ET AL.<br />
3. RESULTS<br />
Based on the cluster analysis using a dissimilarity index of 12.2 as a cut-off point,<br />
the 30 selected homegardens were categorized into nine clusters with different<br />
patterns of tree/shrub species density (Fig. 1). As five clusters consisted of only one<br />
Figure 1. Hierarchical classification of 30 homegardens in Kerala, India.<br />
homegarden each, a qualitative assessment was made to further delineate different<br />
homegarden types. Four clusters (clusters 4, 5, 6 and 7) which were similar in<br />
respect of their very small size (0.12 to 0.2 ha) were combined. Cluster 8 was
maintained as a specific type due to its large size (0.81 ha) and specific structure.<br />
Consequently, the nine clusters were regrouped into six homegarden types for<br />
further analysis (Fig. 1).<br />
3.2. Structural characteristics<br />
The different homegarden types showed important variations in all their structural<br />
characteristics except for the number of species (Table 1). Although the average<br />
number of species in the various homegarden types ranged from 17 to 51, in types 1,<br />
2, 4, and 6, the average number of species per homegarden were relatively similar.<br />
Types 2, 3, and 4 are small homegardens, whereas types 5, 6, and 1 are much larger.<br />
Types 1, 2, and 3 have a much lower tree density than types 4, 5, and 6. Types 1, 3,<br />
and 6 have lower species diversities (higher Simpson and lower Shannon indices)<br />
compared to types 2, 4, and 5. Finally, types 1 and 6 have lower evenness values<br />
than the other garden types. This indicates that in these homegarden types,<br />
production is oriented toward fewer species compared to types 2, 4, and 5. Further<br />
statistical tests were applied on types 1, 2, and 3 (Table 1). Type 1 is significantly<br />
larger than types 2 and 3. The three types are significantly different in respect to<br />
their number of species, but have similar tree/shrub densities per homegarden.<br />
Concerning the diversity indices, type 1 is statistically less diverse and has a lower<br />
evenness compared to type 2. Type 3 is intermediate.<br />
Attributes<br />
Homegarden<br />
size (ha)<br />
Number of<br />
species<br />
Density<br />
(No./ha)<br />
Simpson’s<br />
index<br />
Shannon’s<br />
index<br />
Evenness<br />
1<br />
(n = 8)<br />
0.72 a<br />
(0.117)<br />
27.1<br />
(3.47)<br />
555.5 a<br />
(57.99)<br />
0.35 a<br />
(0.077)<br />
0.79 a<br />
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 93<br />
Table 1. The structural characteristics of six homegarden types, Kerala, India.<br />
2<br />
(n = 11)<br />
0.40 b<br />
(0.076)<br />
28.7<br />
(1.33)<br />
449.0 a<br />
(54.9)<br />
0.08 b<br />
(0.007)<br />
1.24 b<br />
Homegarden types Tests<br />
3<br />
(n = 4)<br />
0.24 b<br />
(0.057)<br />
17.7<br />
(3.09)<br />
621.3 a<br />
(128.9)<br />
0.20 ab<br />
(0.028)<br />
0.89 a<br />
(0.108) (0.020) (0.092)<br />
0.56 a<br />
0.86<br />
(0.060)<br />
b<br />
0.72<br />
(0.013)<br />
b<br />
(0.052)<br />
4<br />
(n = 4)<br />
0.14<br />
(0.020)<br />
27.5<br />
(3.77)<br />
1105.8<br />
(137.7)<br />
0.09<br />
(0.015)<br />
1.21<br />
(0.076)<br />
0.84<br />
(0.018)<br />
5<br />
(n = 1)<br />
0.81<br />
(na)<br />
51.0<br />
(na)<br />
1671.6<br />
(na)<br />
0.09<br />
(na)<br />
1.32<br />
(na)<br />
0.77<br />
(na)<br />
6<br />
(n = 2)<br />
1.01<br />
(0.200)<br />
24.0<br />
(3.00)<br />
1387.9<br />
(46.6)<br />
0.51<br />
(0.068)<br />
0.50<br />
(0.087)<br />
0.36<br />
(0.049)<br />
Type F/Chi2<br />
Values in parentheses represent the standard error (na = not applicable).<br />
Values with different letters among homegardens are significantly different.<br />
Tests: A = ANOVA, KW = Kruskall Wallis; F value for ANOVA, Chi-square values for<br />
Kruskall & Wallis tests and P = probability level of significance.<br />
P<br />
A 4.04 0.034<br />
KW 6.14 0.046<br />
KW 2.74 0.254<br />
A 9.33 0.001<br />
A 13.18 0.001<br />
A 16.65 0.001
94<br />
In general, there is a tendency that with an increase in size of homegardens [from<br />
type 4 (very small) to types 2 and 3 (small) to type 1 (medium) and type 6 (very<br />
big)], there is an increase in the Simpson’s diversity index, a decrease in the<br />
Shannon’s diversity, and a decrease in the evenness index. Only homegarden type 5<br />
does not fit into this pattern; this big homegarden has a low Simpson’s diversity<br />
index, a high Shannon’s diversity index, and a high evenness index.<br />
3.3. Functional characteristics<br />
A.PEYRE ET AL.<br />
The various homegarden types differ in functional characteristics (Fig. 2). A range<br />
from five to nine use-categories was present in the homegarden types. Fruits and<br />
nuts, spices, timber, and coconut, are present in all homegarden types. In two types,<br />
only one use group consists of more than 50% of all trees: rubber in type 1 and<br />
beverage in type 6. The relatively less important use categories are the ones of<br />
religious, medicinal, and multipurpose trees.<br />
Figure 2. Functional characteristics of six different homegarden types, Kerala, India (% of<br />
number of individuals in each functional use group).<br />
The different types of homegardens can be arranged along a gradient from<br />
predominately single commodity production to intensive multiple cropping. Type 1<br />
is predominantly focused on rubber production, types 3 and 4 are characterized by a<br />
mixture of fruit trees and coconuts, type 6 had a mixture of fruit trees and beverage<br />
crops with some additional spices and timber trees, type 2 was a mixture of coconut,<br />
fruit trees, and timber trees, and type 5 by an intensive mixture of timber trees,<br />
spices, fruit trees, and beverage crops. These characteristics are related to the<br />
differences in whether farmers are oriented toward cash income generation or home
consumption. Farmers managing type 1 and 6 are cash-oriented as reflected by the<br />
dominance of rubber and beverage producing crops or stimulant producing trees<br />
respectively. In these gardens, cash crops represent more than half of the total<br />
number of trees. Also the farmer managing homegarden 5 is cash-production<br />
oriented, but in this case there is no clear dominance of any crop. The managers of<br />
homegarden type 3 are focused on both cash income generation (coconuts) and<br />
subsistence production (fruits). The homegarden types 2 and 4 are both home<br />
consumption oriented. These homegardens are small in contrast to the cash-oriented<br />
types 5 and 6, which are larger in size. The garden type 1 with highest production<br />
specialization in rubber production is of medium size.<br />
3.4. Management characteristics<br />
Most management practices concern the manipulation of the tree environment rather<br />
than the tree itself. Sanitary pruning, rejuvenation pruning, canopy pruning to<br />
increase light penetration and cutting low branches are seldom, whereas weeding,<br />
fertilization, and crop spacing are more common (Table 2). In particular, cash crops<br />
are subjected to a variety of management practices. The most intensively managed<br />
species are coconut, rubber and arecanut; their cultivation includes use of chemical<br />
fertilizers and insecticides, systematic weeding, organic fertilization, and row<br />
arrangement of trees. They are also relatively often protected from competitors and<br />
are the only crops that receive watering. Fruit trees and neem receive less attention<br />
(selective weeding, some application of organic manures). The valuable timber<br />
species, teak receives no particular attention to increase its productivity.<br />
The different homegarden types can be arranged along a gradient of management<br />
intensity. The small-sized types 4 and 2 are on one end of this gradient characterized<br />
by low to medium management with a concentration on internal inputs and with<br />
random arrangement of trees. On the other extreme of the gradient, the medium- to<br />
big sized homegarden types 1, 5, and 6 are subject to a more intensive management<br />
with use of both internal and external inputs such as chemical fertilizers, insecticides<br />
and purchased seedlings. In this case, row planting is dominant. The small-sized<br />
type 3 has intermediate characteristics, with only a medium intensity of management,<br />
but with a dominant spatial arrangement in row. In summary, the smaller<br />
homegarden types are managed at a lower intensity than the larger ones, but their<br />
production is more diverse. Type 5 has the distinctive feature of being a large garden<br />
with very intensive management but low use of external inputs; this homegarden is<br />
oriented at multiple-production.<br />
3.5. Homegarden dynamics<br />
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 95<br />
During the past decade, there has been hardly any change in homegarden size.<br />
However, several changes in the structure and function occurred; these varied for the<br />
different homegarden types (Table 3). Spices (black pepper or Piper nigrum) and<br />
ornamental species are the only use categories that have been introduced in all<br />
homegarden types except in types 5 and 3 where they were already present.
96<br />
Table 2. Management practices of seven common species in different homegarden types.<br />
Inputs<br />
Inputs<br />
Inputs<br />
Management<br />
Management<br />
Management<br />
A.PEYRE ET AL.<br />
Species/management HG types<br />
1 2 3 4 5 6<br />
presence 8/8 0/11 0/4 0/4 1/1 0/2<br />
latex tapping 8 - - - 1 -<br />
weeding 6 - - - 1 -<br />
removing competition 4 - - - 0 -<br />
sanitary pruning 0 - - - 1 -<br />
cut low branches 0 - - - 1 -<br />
spatial arrangement 2 - - - 2 -<br />
Int organic fertilization 8 - - - 1 -<br />
Ext<br />
bought seedlings<br />
chemicals<br />
7<br />
8<br />
-<br />
-<br />
-<br />
-<br />
-<br />
-<br />
1<br />
0<br />
-<br />
-<br />
presence 8/8 11/11 4/4 4/4 1/1 2/2<br />
nut harvesting 8 11 4 4 1 2<br />
weeding 8 6 3 0 1 2<br />
watering 0 4 2 2 0 2<br />
ringing 8 9 4 0 0 2<br />
sanitary pruning 0 0 0 0 1 0<br />
removing competition 4 6 2 0 0 0<br />
leaf harvesting 0 5 2 0 0 2<br />
canopy prunings 0 0 0 0 1 0<br />
spatial arrangement 2 2 2 2 2 2<br />
organic fertilization 7 10 4 3 1 2<br />
Int<br />
mulching<br />
seeding<br />
8<br />
0<br />
10<br />
0<br />
4<br />
0<br />
3<br />
0<br />
0<br />
0<br />
1<br />
2<br />
nursery 0 0 2 0 0 1<br />
Ext<br />
bought seedlings<br />
chemicals<br />
5<br />
5<br />
7<br />
0<br />
2<br />
2<br />
4<br />
0<br />
1<br />
1<br />
0<br />
0<br />
presence 3/8 1/11 4/4 0/4 0/1 2/2<br />
nut harvesting 3 1 4 - - 2<br />
weeding 3 1 0 - - 2<br />
watering 2 0 0 - - 2<br />
ringing 3 1 2 - - 1<br />
sanitary pruning 0 0 0 - - 1<br />
removing competition 0 0 0 - - 1<br />
spatial arrangement 2 2 1/2 - - 1/2<br />
organic fertilization 3 1 3 - - 2<br />
Int<br />
mulching<br />
seeding<br />
2<br />
2<br />
0<br />
0<br />
2<br />
0<br />
-<br />
-<br />
-<br />
-<br />
0<br />
1<br />
nursery 0 0 1 - - 1<br />
Ext<br />
bought seedlings<br />
chemicals<br />
0<br />
2<br />
1<br />
0<br />
0<br />
0<br />
-<br />
-<br />
-<br />
-<br />
1<br />
0
MANGO<br />
JACKFRUIT<br />
TEAK<br />
Inputs<br />
Inputs<br />
Inputs<br />
Management<br />
Management<br />
Management<br />
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 97<br />
Species/management H G types<br />
1 2 3 4 5 6<br />
presence 7/8 11/11 4/4 4/4 1/1 2/2<br />
fruit harvesting 7 11 4 4 1 2<br />
sanitary pruning 2 5 2 0 1 1<br />
rejuvenation pruning 0 0 0 0 1 0<br />
canopy pruning 0 0 0 0 1 0<br />
lopping 2 2 2 0 1 0<br />
weeding 0 0 0 0 1 0<br />
cutting low branches 0 0 0 0 1 0<br />
spatial arrangement 4 4 4 4 4 4<br />
organic fertilization 0 0 0 0 1 0<br />
seeding 3 3 0 0 0 0<br />
Int<br />
nursery 0 3 3 0 1 0<br />
protecting natural regeneration 0 0 0 2 0 1<br />
plant cuttings 0 0 0 0 0 1<br />
Ext<br />
bought seedlings<br />
chemicals<br />
3<br />
0<br />
5<br />
3<br />
0<br />
0<br />
3<br />
0<br />
0<br />
0<br />
0<br />
0<br />
presence 8/8 10/11 4/4 3/4 1/1 2/2<br />
fruit harvesting 8 10 4 3 1 2<br />
sanitary pruning 3 3 2 0 1 1<br />
rejuvenation pruning 0 0 0 0 1 0<br />
canopy pruning 0 0 0 0 1 0<br />
lopping 0 2 0 0 1 0<br />
weeding 0 0 0 0 1 0<br />
cutting low branches 0 2 0 0 1 0<br />
spatial arrangement 4 4 4 4 4 4<br />
organic fertilization 0 0 0 0 1 0<br />
Int<br />
seeding<br />
nursery<br />
4<br />
0<br />
5<br />
0<br />
0<br />
0<br />
2<br />
0<br />
0<br />
1<br />
1<br />
0<br />
protecting natural regeneration 2 0 2 0 0 1<br />
Ext<br />
bought seedlings<br />
wildings<br />
0<br />
0<br />
0<br />
3<br />
0<br />
0<br />
0<br />
0<br />
0<br />
0<br />
0<br />
0<br />
presence 6/8 10/11 1/4 3/4 1/1 2/2<br />
sanitary pruning 2 2 0 0 1 2<br />
rejuvenation pruning 0 0 0 0 1 0<br />
canopy pruning 0 0 0 0 1 0<br />
lopping 3 2 0 0 1 2<br />
weeding 0 2 0 0 1 1<br />
cutting low branches 0 0 0 0 1 1<br />
coppicing 0 0 0 0 1 1<br />
spatial arrangement 4 4 1 4 4 1/4<br />
organic fertilization 0 2 0 0 1 0<br />
Int<br />
nursery 0 2 0 0 1 1<br />
protecting natural regeneration 4 3 0 2 0 0<br />
Ext bought seedlings 4 3 1 0 0 1
98<br />
NEEM<br />
Inputs<br />
Management<br />
A.PEYRE ET AL.<br />
Species/management HG types<br />
1 2 3 4 5 6<br />
presence 5/8 5/11 0/4 2/4 1/1 0/2<br />
leaf harvesting 3 5 - 0 1 -<br />
sanitary pruning 0 2 - 0 1 -<br />
rejuvenation pruning 0 0 - 0 1 -<br />
lopping 0 0 - 0 1 -<br />
cutting low branches 0 0 - 0 1 -<br />
spatial arrangement 1 4 4 4 1/4 4<br />
organic fertilization 0 0 - 0 1 -<br />
Int<br />
nursery 0 0 - 0 1 -<br />
protecting natural regeneration 5 3 - 1 0 -<br />
Ext bought seedlings 0 0 - 1 0 -<br />
Presence: a/b a = number of homegardens studied; b = number of homegardens with species;<br />
(-) tree is not present; HG = homegardens, Int = internal, Ext = external; Legend for spatial<br />
arrangements: 1 = borders, 2 = rows, 3 = strips, 4 = scattered.<br />
Ornamentals are usually cultivated around and in front of the house and along the<br />
paths. Black pepper is usually associated with palm trees in order to benefit from<br />
their soil management and inputs. Some farmers reported difficulties to harvest the<br />
palm nuts without damaging the pepper vines. Also some other support trees such as<br />
Erythrina spp. were introduced. In five out of the six homegarden types, fruit trees<br />
have also been introduced. They are usually cultivated close to the house, except for<br />
big trees such as mango or jackfruit or when planted in a large-scale. Another<br />
change concerns the medicinal plants. In the homegarden types 1, 5, and 6, many<br />
farmers have partially removed the medicinal species.<br />
Few structural and functional changes have occurred in types 2 and 4, especially<br />
when compared to types 1 and 6. The large majority (93%) of the homegardens of<br />
types 2 and 4 are still subsistence-oriented, just as they were 10 years ago. Crop<br />
introductions do not concentrate on any specific species or use and are of low<br />
intensity (less than 50 individuals per species). These homegardens have preserved<br />
the traditional features; they still have a multistoried structure, high diversity and<br />
low dependency on external inputs. In contrast, 60% of farmers managing types 1<br />
and 6 have shifted to a cash strategy with a modernized management oriented<br />
toward a few cash crops such as rubber, arecanut, and coffee. The introduction of<br />
these commercial crops resulted in important structural and functional changes. The<br />
canopy became less stratified and species diversity was reduced, notably in respect<br />
to species producing fruits and nuts, timber, and medicines. This caused a reduction<br />
in the multiple functions of homegardens. This change was most dominant in case of<br />
increased rubber cultivation, as this species is always grown as a monoculture.<br />
Coconut and arecanut are often still intercropped. Moreover, 70% of the farmers<br />
increased their use of chemical inputs.<br />
No clear pattern could be deduced concerning the dynamics of the homegarden<br />
types 3 and 5. Although the production pattern of type 3 changed, its vegetation<br />
structure did not undergo any fundamental modification and the predominant spatial<br />
arrangement of trees remained in rows. The owner of type 5 follows a long-term<br />
cash strategy oriented toward timber production. The farmer has been able to follow
the market demand by introducing more rubber and arecanut trees. However, these<br />
introductions did not affect the structural characteristics and vegetation structure.<br />
Table 3. Species introductions and changes in spatial arrangements in the period 1993 – 2003 in<br />
six homegarden types, Kerala, India.<br />
Garden<br />
types<br />
Introduced<br />
uses/species<br />
Type 4 Spices (pepper)<br />
Fruits (Citrus,<br />
guava)<br />
Beverage (coffee)<br />
Ornamentals<br />
Type 2 Spices (pepper)<br />
Fruits (Citrus,<br />
guava, Annona,<br />
papaya)<br />
Ornamentals<br />
Type 3 Spices (pepper)<br />
Fruits (guava, jack,<br />
cashew, papaya)<br />
Type 5 Beverage (arecanut)<br />
Cash (rubber)<br />
Ornamentals<br />
Type 6 Timber (teak)<br />
Ornamentals<br />
Beverage (arecanut,<br />
coffee)<br />
Spices (pepper)<br />
Fruits (banana)<br />
Type 1 Spices (pepper)<br />
Fruits (guava,<br />
Citrus)<br />
Ornamentals<br />
Cash (rubber)<br />
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 99<br />
Rate of<br />
introduction<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
low<br />
high<br />
high<br />
high<br />
low<br />
low<br />
low<br />
high<br />
Spatial arrangements of trees<br />
10 years ago Nowadays<br />
random random<br />
random random<br />
row row<br />
partly<br />
random,<br />
partly rows<br />
random row<br />
random row<br />
partly<br />
random,<br />
partly rows<br />
Low = less than 50 individuals introduced in total; High = more than 50 individuals<br />
introduced in total.<br />
3.6. Classification of homegarden types on an evolutionary axis<br />
Based on their structural and functional characteristics and dynamics, the different<br />
homegarden types can be arranged along a gradient from traditionall to modernized<br />
homegardens (Table 4). The homegarden types 2 and 4 are relatively small and have<br />
a high diversity and a random arrangement of trees. Few changes occurred during<br />
the past decade and the traditional features of homegardens have been preserved
100<br />
(high diversity, multi-storied canopy, and multi-production). These homegardens are<br />
oriented toward subsistence production and few products are sold. The management<br />
practices are predominantly based on internal inputs, although in type 2 some<br />
external inputs are also used. Based on these characteristics, they can be<br />
characterized as traditional l homegardens. These traditional homegardens can be<br />
contrasted with homegarden types 1 and 6, which can be characterized as “modern.”<br />
In these modern homegarden types, farmers have adopted a cash-orientation and<br />
have introduced several new management practices. In these relatively big<br />
homegardens the production became oriented at a few cash crops which are<br />
systematically arranged in rows. In the case of rubber, part of the homegarden is<br />
even transformed into single species plantation. Also, the use of external inputs<br />
(purchased seedling, chemical fertilizers, and insecticides) has increased.<br />
Table 4. Ordination of homegarden types along a gradient from traditional to modernized<br />
homegarden, Kerala, India.<br />
HG<br />
categories<br />
Type (s) No. of<br />
HGs<br />
Size Orientation Nature of<br />
production<br />
Tree/shrub<br />
diversity<br />
Traditional 2,4 15 (very) small home multiple high<br />
Adapted<br />
traditional<br />
6 1 big cash multiple high<br />
Incipient 3 4 small home and multiple medium<br />
modern<br />
cash<br />
Modern 5,1 10 medium to<br />
very big<br />
cash mono low<br />
HG = homegardens.<br />
A.PEYRE ET AL.<br />
Type 3 can be considered as incipient modern type as it shares both traditional<br />
and modern characteristics. This homegarden type consists of small homegardens<br />
with medium diversity, and involves a low management intensity that depends<br />
predominantly on external inputs. Although type 5 is characterized by its cash<br />
orientation including introduction of new cash crops such as rubber and systematic<br />
spacing of trees, it still maintains the multispecies composition of the traditional<br />
homegardens. The garden is very intensively managed, but mostly with internal<br />
inputs by using organic fertilization and mulching for soil management and by<br />
regenerating trees by protecting natural regeneration, seeding, and using local plant<br />
material such as plant cuttings. Thus, although this homegarden was adapted to the<br />
modern cash economy, it maintained several characteristics of the traditional<br />
homegardens.<br />
4. DISCUSSION AND CONCLUSIONS<br />
This study shows that homegardens should not be considered as being static. Rather,<br />
their composition and management are gradually evolving in response to the
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 101<br />
socioeconomic dynamics. Only 50% of all respondents still followed traditional<br />
homegarden management practices, whereas 33% of all respondents have adopted<br />
modern practices by increasingly moving towards concentrated cash crop production<br />
and use of external inputs. Traditional homegardens were mostly of small size, while<br />
modern homegardens are much larger. This parameter should not be interpreted as<br />
the only, or main, feature influencing the development path of homegardens. Other<br />
factors, such as the role of the homegarden in the overall farming system and the<br />
degree to which a household has access to off-farm employment and income<br />
(Wiersum, 2006) might be of more importance. Unfortunately, these factors could<br />
not be taken into account in the present study.<br />
Our data reinforce the general fears regarding the loss of traditional characteristics<br />
of homegardens and their gradual demise into cash crop production systems<br />
(Kumar and Nair, 2004). Because of the rise of market economy, agriculture in<br />
Palakkad region of Kerala is currently struggling to find new intensification<br />
strategies. Although traditional Kerala homegardens are reputed to be sustainable in<br />
both biophysical and socioeconomic terms, they do gradually change from a<br />
traditional type to a more modern one. This process of modernization often brings<br />
with it a decrease of the tree/shrub diversity, a gradual concentration on a limited<br />
number of cash-crop species, gradual homogenization of homegarden structure and<br />
increased use of external inputs.<br />
Interestingly, however, one farmer in our sample had combined an increased<br />
orientation at cash crop production with the maintenance of a high species diversity<br />
and use of internal rather than external inputs. This example shows that there is no<br />
single uniform trend towards the modernization of homegardens in Kerala, but that<br />
alternative pathways exist. Moreover, this example also shows that traditional<br />
ecological features ensuring ecological sustainability of homegardens could still be<br />
maintained in modernized homegardens. This suggests that it might be possible to<br />
identify new development policies that aim at optimal combination of ecological and<br />
productive features of the homegardens rather than optimizing only cash crop<br />
production.<br />
Although the study was focused on ascertaining trends in tree composition<br />
resulting from the process of commercialization, other trends influencing the composition<br />
of the homegarden vegetation were also observed (Wiersum, 2006). These<br />
included an increase in the use of ornamental plants and an increase in staple food<br />
production. The trend in gradual replacement of functional plants to ornamentals has<br />
also been observed in cases where people became richer. The gradual increase in<br />
staple food production was specifically found in cases where homegardens were the<br />
last remaining farming unit of poor households. Unfortunately, little attention has<br />
been given towards systematically studying under which set of conditions these<br />
different trends in homegarden development occur, to what extent they are<br />
interrelated, how they are related towards changes in livelihood conditions, and what<br />
their impact on biodiversity is.<br />
Our study further shows that it is incorrect to assume a uniform development<br />
pattern for all homegardens, rather different pathways in homegarden development<br />
may co-exist. At present rural areas are subject to many socioeconomic changes<br />
(Ashley and Maxwell, 2001). The notion of homegardens being sustainable needs
102<br />
A. PEYRE ET AL.<br />
therefore to be specified in respect to ecological and social sustainability. Whereas<br />
the concept of ecological sustainability is time-independent, the concept of social<br />
sustainability includes the notion of agroforestry systems adjusting in a timely<br />
fashion to changing rural conditions. With respect to the potential of traditional<br />
agroforestry systems such as homegardens, the focus in assessing social sustainability<br />
should not only be on the question of whether the system fits into the<br />
traditional farming and livelihood systems, but also on the question of whether these<br />
agroforestry systems can be adjusted to modern rural conditions while still maintaining<br />
their features of ecological sustainability. Our study shows that research based on<br />
detailed assessments of the actual dynamics in the features of traditional agroforestry<br />
systems is rewarding. Such studies may indicate that different developments<br />
trajectories are being followed. The understanding of these development pathways and<br />
the factors involved offers good scope for the identification of options for further<br />
modification of agroforestry systems.<br />
ACKNOWLEDGEMENTS<br />
The fieldwork for this research was accommodated by the Integrated Rural<br />
Technology Center, Mundur, Palakkad, Kerala. Dr. Unnikrishnan provided valuable<br />
background information and important assistance to the organization of the survey.<br />
Additional information on the status of homegardens was provided by Dr. B. Mohan<br />
Kumar, Kerala Agricultural University, Dr. S. Sankar, Kerala Forest Research<br />
Institute and M/s. K.P. Ouseph and M. Shetty of the Kerala Forest Department.<br />
ENDNOTES<br />
1. The trends in homegarden structure and composition in the research area were<br />
discussed in an International workshop on agroforestry and natural resource<br />
management, organized in 2002 by the Centre for Rural Development and<br />
Appropriate Technology, Cochin University of Science and Technology, in<br />
association with the Integrated Rural Technology Center, Mundur, Palakkad,<br />
Kerala, India.<br />
2. Wiersum K.F. and Slingerland M. 1996. Use and management of two<br />
multipurpose tree species (Parkia biglobosa and Detarium microcarpum) in<br />
agrisilvopastoral land use systems in Burkina Faso. Wageningen Agricultural<br />
University, Antenne sahelienne. Document de projet No. 41.<br />
REFERENCES<br />
Ashley C. and Maxwell S. 2001. Rethinking rural development. Dev Pol Rev 19: 395 – 425.<br />
FAO. 1977. FAO-UNESCO Soil Map of the World 1:5000000. Volume VII, South Asia.<br />
UNESCO, Paris.<br />
Huston M.A. 1994. Biological diversity. The coexistence of species on changing landscapes.<br />
Cambridge University Press. 681p.<br />
John J. and Nair M.A. 1999. Socio-economic characteristics of homestead farming in south<br />
Kerala. J Trop Agric 37: 107 – 109.
HOMEGARDEN DYNAMICS IN KERALA, K INDIA I 103<br />
Jose D. and Shanmugaratnam N. 1993. Traditional homegardens of Kerala, a sustainable<br />
human ecosystem. Agroforest Syst 24: 203 –213.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Kumar B.M., George S.J. and Chinnamani S. 1994. Diversity, structure and standing stock of<br />
wood in the homegardens of Kerala in peninsular India. Agroforest Syst 25: 243 –262.<br />
Leiva J.M., Azurdia C., Ovanda W., Lopez E. and Ayala H. 2002. Contributions of<br />
homegardens to in situ conservation in traditional farming systems – Guatemalan<br />
component. In: Watson J.W. and Eyzaguirre P.B. (eds), <strong>Homegardens</strong> and in situ<br />
conservation of plant genetic resources in farming systems. Proceedings of the Second<br />
International homegarden workshop, Witzenhausen, Germany, pp 56 – 72. International<br />
Plant Genetic Resources Institute, Rome.<br />
Méndez V.E., Lok R., and Somarriba E. 2001. Interdisciplinairy analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Nair M.A. and Sreedharan C. 1986. Agroforestry farming systems in the homesteads of<br />
Kerala, southern India. Agroforest Syst 4: 339 – 363.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Quiroz C., Gutierrez M., Rodriguez D., Perez D., Ynfante J., Gamez J., Perez de Fernandez<br />
T., Marques A. and Pacheco W. 2002. <strong>Homegardens</strong> and in situ conservation of agrobiodiversity<br />
– Venezuelan component. In: Watson J.W. and Eyzaguirre P.B. (eds),<br />
<strong>Homegardens</strong> and in situ conservation of plant genetic resources in farming systems.<br />
Proceedings of the Second International homegarden workshop, Witzenhausen, Germany,<br />
pp 73 – 82. International Plant Genetic Resources Institute, Rome.<br />
Soemarwoto O., 1987. <strong>Homegardens</strong>: a traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry, a decade of development,<br />
pp 157 – 172. ICRAF, Nairobi.<br />
Tesfaye Abebe 2005. Diversity in homegarden agroforestry systems of southern Ethiopia.<br />
Wageningen University, the Netherlands, <strong>Tropical</strong> Resource Management Paper No. 59,<br />
143p.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Wiersum K.F. 1995. 200 years of sustainability in forestry: lessons from history. Environ<br />
Manage 19: 321 – 329.<br />
Wiersum K.F. 1997. Indigenous exploitation and management of tropical forest resources: an<br />
evolutionary continuum in forest – people interactions. Agric Ecosyst Environ 63: 1 – 16.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.
CHAPTER 7<br />
STRUCTURE AND DYNAMICS<br />
OF COCONUT-BASED AGROFORESTRY<br />
SYSTEMS IN MELANESIA:<br />
A CASE STUDY FROM<br />
THE VANUATU ARCHIPELAGO<br />
N. LAMANDA 1 *, E. MALÉZIEUX 1 , AND P. MARTIN 2<br />
1 CIRAD UMR SYSTEM, Agro M, Bâtiment 27, 2 place viala, 34060 Montpellier,<br />
France; *E-mail: . 2 INA P-G département AGER,<br />
bâtiment EGER BP 01, 78850 Thiverval-Grignon, France<br />
Keywords: Cocos nucifera, Cropping system dynamics, Melanesian agriculture, Vegetation<br />
structure analysis.<br />
Abstract. Coconut (Cocos nucifera)-based agroforestry systems hold promise as a sustainable<br />
land use activity in the Melanesian islands, where food dependency on foreign sources and<br />
land shortages are increasing dramatically. This chapter describes the dynamics of these<br />
smallholder production systems in the Malo Island of northern Vanuatu (Melanesia), where a<br />
dual economy operates in which resources are dedicated to both subsistence and commercial<br />
production. The floristic elements found in the coconut plantations were typical of those<br />
described in the humid tropical homegardens elsewhere, with an average of 12 tree species<br />
per plot. Mean Shannon Weaver index was 1.57 with the vertical profile of vegetation having<br />
one-to-five strata. Although the coconut palms dominate these production systems, in certain<br />
cases other trees may dominate it. Situations in coconut plots evolve throughout the<br />
development phase of the palms. Based on that, five types of smallholder coconut-based<br />
agroforestry systems were recognized, which falls into two main evolutionary patterns: (1) a<br />
perennial occupation of the cultivated land by coconut trees, because of coconut replanting,<br />
and (2) a gradual return to tree fallow in which the coconut palms gradually disappear<br />
because of changes in the complex multistrata vegetation.<br />
1. INTRODUCTION<br />
In the Melanesian archipelago of Vanuatu, about 80% of the estimated 0.2 million<br />
population lives in rural areas and are involved in agriculture (Labouisse, 2004). The<br />
105<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 105–121.<br />
© 2006 Springer. Printed in the Netherlands.
106 N. LLAMANDA<br />
ET AL.<br />
traditional farming systems are shifting cultivation with long fallows (food gardens)<br />
and cultivation of coconut palms (Cocos nucifera) with a mixture of other species.<br />
The coconut palm incidentally is known as the “tree of life” in the Pacific islands<br />
because of its multiple uses. The staple food crops in the multistory food gardens<br />
include root and tuber crops, for example, yam (Dioscorea spp.) and taro (Colocasia<br />
esculenta), often closely associated with other species such as banana (Musa spp.),<br />
island cabbage ( (Abelmoschus spp.), or cassava (Manihot esculenta) and numerous<br />
tree species ( (Artocarpus altilis,<br />
Barringtonia edulis, etc.). Some farmers also<br />
undertake pig breeding, mainly due to social considerations (Bonnemaison, 1996).<br />
During the 20th century, however, development of large “coconut estates” by<br />
the Europeans became a dominant land use activity and was rapidly followed by the<br />
evolution of a large number of smallholder plantations that substantially altered the<br />
indigenous farming systems in Vanuatu (Barrau, 1955; Clarke and Thaman, 1993;<br />
Bonnemaison, 1996). In particular, such coconut plantations became dominant in the<br />
northern islands of the archipelago where the agroclimatic conditions and market<br />
opportunities were ideally suited for coconut production. Furthermore, over a period<br />
of time, the intercropped, smallholder food gardens, with young coconut trees<br />
planted after bush or tree fallow clearing (Weightman, 1989) have evolved into<br />
complex farming systems in which coconut is associated with numerous other<br />
species and/or cattle grazing. Because of the development of coconut plantations,<br />
often on the best agricultural lands, food gardens were pushed farther from villages<br />
and onto the marginal lands (Clarke and Thaman, 1993; Bonnemaison, 1996).<br />
Concomitantly, forests or old tree-fallows were also converted into gardens and<br />
coconut plantations.<br />
At present about 60% of the cultivated area in Vanuatu is occupied by coconut<br />
plantations and copra production is still the major source of income for the northern<br />
Vanuatu’s rural population, despite the downward trend in copra prices worldwide<br />
during the past decade (Labouisse, 2004). With an increasing population that may<br />
double over the next 30 years, food dependency on external sources and pressure on<br />
natural resources may increase, and farmers and agricultural extension services are<br />
expressing concern about how to improve the current cropping systems. As a result,<br />
a diagnosis of the performance and sustainability of the existing situations in the<br />
coconut farms is needed in order to manage and prepare for the intensification of<br />
these systems. Moreover, the existing situations are the result of mixed species<br />
vegetation developing in plots managed with farmers’ practices, and are, in turn<br />
dependent on local agro-ecological conditions. This chapter analyzes the dynamics<br />
of these complex coconut-based land use systems and the evolution of its vegetation<br />
structure over time.<br />
2. MATERIALS AND METHODS<br />
2.1. Study area and selection of sample plots<br />
The study site was situated on Malo Island (15°40’S, 167°10’E) in northern Vanuatu<br />
that has a dual economy, in which resources are dedicated to both subsistence and
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
107<br />
commercial production (Allen, 2000). Malo Island, which covers about 180 km², is<br />
located southeast of the Santo Island, 20 km from Luganville (Santo Island), the<br />
second-ranking urban center of the archipelago (Fig. 1). This island, with its highly<br />
fertile soils and an equatorial climate tempered by oceanic influence (Quantin,<br />
1982), offers optimum conditions for coconut cultivation (IRHO, 1969). Coincidentally,<br />
copra production is the main source of income to the inhabitants.<br />
Figure 1. Location of the study area, Malo Island, Vanuatu.<br />
The experimental approach involved characterizing the structure and dynamics<br />
of the smallholder coconut-based agroforestry systems. It consisted of four steps: (1)<br />
selection of sample plots representing the diverse situations existing in the study
108<br />
area; (2) description of the selected plots that included a farmer survey and plot<br />
observations; (3) classification and grouping of the monitored plots according to<br />
their vegetation structure; and (4) correlation studies to gain insights into the<br />
temporal dynamics of the cropping systems.<br />
Malo island was first stratified according to its biophysical and socioeconomic<br />
characteristics (soil type, climate, and market opportunity) using a participatory<br />
mapping technique (Caron, 1997). The bibliographical data (Quantin, 1982;<br />
Weightman, 1989; Allen, 2000) were supplemented with historical data gathered<br />
through interviews of the local chiefs, farmers, and extension officers, chosen for<br />
their knowledge of Malo and its agriculture. Soil and climate characteristics,<br />
population density and origin, roads and marketing areas were also mapped on the<br />
basis of these bibliographical and interview data.<br />
By linking the historical aspects and the biophysical characteristics of Malo<br />
(Lamanda et al., 2004), two distinct production areas were identified: the western<br />
area and the central eastern area. A native population and heterogeneous biophysical<br />
conditions characterize the western area. The predominant soil types are coral<br />
limestone in the coastal region and clay soils on the hills. In this area, the<br />
households are nested within villages and the farm fields of each family are<br />
scattered over large areas (i.e., the fields are often more than half an hour’s walk<br />
from the household). Infrastructures are also concentrated in the villages, including<br />
the main dispensary, markets and roads and a rapid sea-link with the second-ranking<br />
town of the archipelago (Luganville) where most of the harvested produce are<br />
marketed. The central eastern area is characterized by a population essentially<br />
composed of migrants from other islands, and with more homogeneous soil<br />
characteristics (clay soils with good agronomic potential). The habitat is scattered<br />
and the coconut plots are located around the houses. Subsistence food gardens are<br />
also cultivated, sometimes far from the household depending on land availability.<br />
The population density is lower than that in the western area with estimates 1 of 250<br />
inhabitants per km 2 in Avunatari, the main village of the western area, and 15<br />
inhabitants per km² in the central eastern area during 1997.<br />
In the main villages of each production area, farmers were interviewed in order<br />
to select the sample plots. The objective was to produce a sample that captures the<br />
diversity of situations existing in the coconut plots of Malo Island (i.e., different<br />
stages of development of coconut and/or different intercropping situations). We<br />
defined a plott in this study by evaluating the vegetation structure and stage of<br />
development of the coconut palms; accordingly, two or more plots could be<br />
distinguished within a farmer’s field. The fieldwork was carried out in 2002 – ‘03<br />
when 191 coconut plots were sampled and described. These plots (116 plots in the<br />
western area and 75 in the central eastern area) represented different stages of<br />
coconut development over an 85-year period and involved different species<br />
associations.<br />
2.2. Description of the selected plots<br />
N. LLAMANDA<br />
ET AL.<br />
A farmers’ survey was conducted in each selected plot in order to assess the<br />
management history of the plots from coconut pre-planting to the present. The
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
109<br />
survey covered aspects such as when and how the plantation was established<br />
(biological material, original planting patterns, how fallow was destroyed, or used:<br />
e.g., forest or tree fallow), what are the changes in associated vegetation types, e.g.,<br />
presence of food gardens or cacao (Theobroma cacao) trees, when coconut palms<br />
began to produce, present management of the plots, and its evolution through time.<br />
The plots were located and their area was calculated using a geographical<br />
positioning system (GPS) when the canopy provided a clear signal; otherwise, direct<br />
measurements were used. All tree and crop species present in each plot were<br />
identified and grouped according to their nature and uses. Density was calculated by<br />
counting the individuals (i) on three sub-plots of size 900 m 2 each for species<br />
regularly planted such as coconut and cacao, and (ii) on the total plot area for other<br />
species. Species richness (number of species) and the Shannon Weaver index of<br />
species diversity (Krebs, 1985) were calculated collectively and separately for trees<br />
and other species.<br />
Horizontal distribution of species was assessed visually and the planting pattern<br />
of coconut and cacao evaluated using a 0 (no visible planting pattern) to +++<br />
(systematic pattern) scale. The distance between rows and trees in a row was<br />
measured for 10 coconut and cacao trees each in order to assess the planting pattern.<br />
Location of the large trees was determined using GPS. The vertical differentiation of<br />
the vegetation profile was first assessed visually and then supplemented by height<br />
measurements obtained for all tree species with individuals taller than 1.5 m.<br />
2.3. Classification of the monitored plots<br />
Structural groups (or vegetation types) were constructed based on the responses to a<br />
hierarchical set of questions concerning vegetation structure in the plot: (1) major<br />
species in the plot, (2) extent of species diversity, (3) horizontal distribution of<br />
major species and species groups, (4) vertical differentiation of the vegetation<br />
(canopy) profile, (5) status of the major species in the vegetation profile, and (6)<br />
dominant species or species groups. Many of these structural parameters, however,<br />
showed a continuous gradation; consequently, the structural groups identified in the<br />
classification scheme also constituted a continuum.<br />
2.4. Dynamics of coconut-based agroforestry systems<br />
A matrix of ‘cropping situations’ that combined the structural groups vs. time was<br />
constructed in order to position the situations described in the coconut plots in a<br />
temporal scheme. Time was represented by the development stages of coconut trees.<br />
Based on literature reference and the information provided by the farmers, four<br />
distinct stages of development of the coconut palms were recognized: (1) juvenile<br />
stage (0 to 7 years), (2) low productive stage (8 to 15 years), (3) productive stage (16<br />
to about 60 years), and (4) senescent stage (over 60 years).<br />
All monitored plots were then positioned in the matrix according to the structural<br />
groups and the development stages of the palms, which was crosschecked through<br />
farmer survey, especially about the date of plantation. The cropping situations were
110<br />
then linked to the management histories of the plots, and expressed as a succession<br />
of cropping situations during coconut development representing its temporal dynamics.<br />
A ‘cropping situation’ defined by the intersection in the matrix of a structural group<br />
and a development stage could be represented by different plots.<br />
3.1. Vegetation structure of coconut plots<br />
N. LLAMANDA<br />
ET AL.<br />
3. RESULTS<br />
Vegetation characteristics of the experimental area are presented in Table 1. The<br />
major tree species found are coconut and cacao. Mean density of coconut palms in<br />
the smallholder plantations was 148 trees per ha, which is close to the 143 trees per<br />
ha recommended by local extension services. Density ranged from 11 to 744 palms<br />
per ha, and higher densities were common especially when two generations of<br />
coconut palms coexisted in the same plot. Mean density for cacao was 209 trees per<br />
ha, with a maximum of 1053, which was indeed less than the density recommended<br />
by the extension services (1111 cacao trees intercropped with 143 coconuts per ha).<br />
Mean size of the coconut plots was 1 ha (range 0.01 to 4 ha) and the smallest plots<br />
were mainly the food gardens associated with juvenile coconut palms.<br />
Table 1. Vegetation characteristics of coconut plots on Malo Island (Vanuatu).<br />
Vegetation characteristics Mean Min Max<br />
Number of species 15 4 40<br />
Number of tree species 12 0 28<br />
Number of semi-perennial herbs 3 0 12<br />
Shannon Weaver index (total) 1.57 0.14 2.81<br />
Shannon Weaver index (trees) 1.5 0 2.88<br />
Shannon Weaver index (semi-perennial herbs) 0.61 0 1.94<br />
Coconut planting density (all generations; no. per ha) 164 11 744<br />
Coconut planting density (number per ha) 148 0 457<br />
Number of tree species per ha<br />
(coconut and cocoa not included)<br />
223 3 2733<br />
Ratio of coconut palms-to-total tree species 0.5 0.05 0.99<br />
Cacao tree planting density (number per ha) 209 0 1053<br />
Age of first generation coconut palms (years) 35 planting 84<br />
Coconut field area (ha) 1 0.01 4<br />
In addition to the two main species mentioned, 90 other useful species were<br />
identified in the monitored plots (a list of conspicuous species with their local names<br />
and uses are given in Appendix I). According to their habit and uses, the species<br />
were grouped as ‘trees’ (49 species mainly with fruit trees such as Mangifera indica
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
or Barringtonia edulis and/or timber trees such as Hibiscus tiliaceus or Pometia<br />
pinnata) and ‘semi-perennial food crop species’ (41 species). The mean number of<br />
cultivated species per plot was 16, with a minimum of four and a maximum of 40<br />
species (Table 1). The most represented species were fruit trees such as Artocarpus<br />
altilis, B. edulis s and M. indica, found in 68, 60, and 55% of the monitored plots<br />
respectively. Semi-perennial food crops such as Musa spp., Carica papaya, Xanthosoma<br />
sagittifolium, and Dioscorea nummularia were also frequently intercropped along<br />
with coconuts (in about 40% of the monitored plots).<br />
The number of trees intercropped with coconut showed a wide range (3 to 2733<br />
trees per ha); consequently, the proportion of f coconut palms relative to other tree<br />
species was highly variable. Indeed, the ratio of the coconut tree density to the<br />
density of other tree species ranged from 0.05 to 0.99 (with a mean of 0.5), which<br />
illustrates the high floristic diversity of the coconut plots (Table 1). Consistent with<br />
this, the Shannon Weaver index collectively for tree and crop species ranged from<br />
0.14 to 2.81 (mean =1.57). Furthermore, species diversity was higher for trees than<br />
for the semi-perennial food crops (mean Shannon Weaver index of 1.50 for trees and<br />
0.60 for food crops). The extent of mixing food crops also varied substantially, with<br />
situations ranging from plots cropped with juvenile coconuts along with mixed food<br />
gardens, to plots in which only a few taro or banana plants were grown.<br />
3.2. Horizontal and vertical l structure of vegetation<br />
111<br />
Theoretically, the horizontal distribution of a species could be ‘systematic’ (with a<br />
repeated pattern), ‘distorted’ (if altered by the death or cutting down of certain<br />
individuals in the systematic pattern), ‘random’ (without any definite pattern), or<br />
‘patchy’ (presence of groups or clusters; Fig. 2). Our observations indicate that the<br />
horizontal distribution of coconut and cacao trees in the sample plots was mostly<br />
systematic or distorted. This is because the coconut trees were mostly planted in a<br />
square pattern with a mean distance of 7.7 m between trees (CV = 14.5%). Likewise,<br />
the cacao trees were interplanted between the coconut rows with a mean distance of<br />
5.3 m between cacao trees (extension recommendations are 9 m for coconuts and 3<br />
m for intercropped cacao). The horizontal distribution of other trees was mostly<br />
random, without any clear geometrical arrangement. Semi-perennial food crop<br />
species mainly had a patchy distribution pattern either on the boundaries or between<br />
tree species depending on species and their cultivation requirements. For instance,<br />
strong yam (Dioscorea nummularia) was often found close to large trees that<br />
provided supports for the vines, while banana plants were clustered in pure or mixed<br />
stands.<br />
As regards to vertical organization of the components, a multistrata arrangement<br />
with one-to-five strata depending on the number and architecture of the tree species<br />
was discernible. Coconut palms often formed the dominant component, but were<br />
sometimes dominated, especially when they were young and/or before the fallow<br />
clearing. Complex situations in which coconuts were both dominant and dominated<br />
were also noted when forest trees (dominant) were combined with food crops<br />
(dominated). Natural regeneration of tree/shrub species constituted the lower strata<br />
along with the food crop species. When cattle grazing under coconut and fruit trees
112<br />
was practiced, the naturally regenerating tree/shrub species of interest were<br />
protected by the farmers.<br />
The horizontal distribution of a species could be :<br />
Regular<br />
Distorted<br />
Randomised<br />
Patchy<br />
Horizontal distributions of each major species and species group<br />
are overlapped to obtain a map of the horizontal vegetation in the plot<br />
Figure 2. Different types of horizontal vegetation distribution on Malo Island, Vanuatu.<br />
3.3. Structural groups<br />
N. LLAMANDA<br />
ET AL.<br />
The monitored plots were finally classified into 14 structural groups (Fig. 3) and<br />
their principal attributes are summarized in Table 2. Similar structural groups were<br />
observed both in the western and central eastern areas and for the sole coconut and<br />
coconut+cacao systems. However, four structural groups not described on Malo but<br />
existing on other Vanuatu islands were also included in the classification scheme for<br />
the sake of comprehensiveness; these include the coconut estates where the palm<br />
was cultivated as a single species, possibly intercropped with cacao or associated<br />
with cattle grazing (I0A I , I0B I , II0A I , II0B I ; Fig. 3). In contrast to these, the smallholder<br />
plots were generally characterized by significant species diversity.<br />
3.4. Temporal dynamics of coconut-based cropping systems<br />
Five coconut-based agroforestry systems were identified and the dynamics of their<br />
vegetation structure during coconut development phases were reconstructed (Fig. 4).<br />
Different structural groups can be noted for a given stage in the coconut cycle.<br />
Productive coconut plots could also be classified into several structural groups, thus<br />
illustrating the profound variability in vegetation structure and farmers’ practices<br />
associated with coconut production in Malo Island (Fig. 5).<br />
The evolutionary pathway starts as a system involving fruit trees, coconut palms,<br />
and food gardens planted together in a tree fallow that has been selectively managed.
1<br />
2<br />
3<br />
4<br />
5<br />
6<br />
Coconut trees tr<br />
1<br />
2<br />
Coconut and d cocoa c<br />
trees<br />
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
3<br />
no vertical<br />
differentiation<br />
4 5 6<br />
regular ular<br />
no vertical<br />
differentiation<br />
4 5 6<br />
altered<br />
no specific cific<br />
diversity ersit<br />
significant significan specific<br />
diversity er<br />
regular ular<br />
5<br />
4 5<br />
vertical<br />
differentiation<br />
food foood<br />
crop s<br />
6<br />
youn you g<br />
coconuts coconut<br />
vertical<br />
differentiation on<br />
4<br />
vertical v<br />
dominant<br />
5 6<br />
cocoas<br />
differentiation<br />
diff<br />
regular ar<br />
5<br />
3<br />
4<br />
altered<br />
dominant<br />
no specific peci<br />
diversity divers<br />
6<br />
cocoas<br />
2<br />
5 6<br />
significant specific pe<br />
diversity<br />
no vertical al<br />
differentiation atio<br />
4<br />
dominant<br />
5<br />
cocoas<br />
6<br />
vertical ver<br />
Coconut trees<br />
differentiation ren<br />
regular gul<br />
Fruit trees<br />
Food crops<br />
Coconut trees<br />
replanting<br />
Cocoa trees<br />
What are the major species in the plot ?<br />
What species diversity is there ?<br />
What is the horizontal distribution of<br />
the major species and of the species group ?<br />
What is the vertical differentiation of the<br />
vegetation profile ?<br />
What is the status of the major tree species<br />
in the vertical profile ?<br />
What are the dominated species<br />
or species groups ?<br />
3<br />
4<br />
5<br />
no vertical tica<br />
differentiation enti<br />
no vertical tica<br />
differentiation ntia<br />
altered ered<br />
dominant nant<br />
dominated omina<br />
6<br />
6<br />
6<br />
dominant and a<br />
dominated ed<br />
cocoas ocoas<br />
altered ered<br />
no vertical<br />
cocoas coc and<br />
differentiation<br />
d<br />
6<br />
food crops<br />
dominant dom<br />
4 5<br />
6<br />
dominated omin<br />
food crops and<br />
young coconuts uts<br />
6<br />
dominant ant and<br />
dominated ed<br />
6<br />
Figure 3. Classification of coconut plots for Malo (Vanuatu) in structural groups.<br />
I0A<br />
I0B<br />
I2<br />
I1A<br />
I1B<br />
I1C<br />
I4A<br />
I4B<br />
I3A<br />
I3B<br />
II 0A<br />
II 0B<br />
II 2<br />
II 1A<br />
II 1B<br />
II 1C<br />
3A<br />
II 3B<br />
Illustrat ions C irad: N . Lam anda, M . D uport al<br />
113
Structural<br />
groups<br />
Monitored<br />
plots (no.)<br />
Table 2. Characteristics of structural groups for Malo Island (Vanuatu).<br />
Number of tree<br />
species per plot<br />
Number of semiperennial<br />
crop<br />
species per plot<br />
Structural attributes of vegetation<br />
Shannon<br />
Weaver index<br />
Coconut tree<br />
density<br />
(no. per ha)<br />
Cacao tree<br />
density<br />
(no. per ha)<br />
Tree density other<br />
than coconut and<br />
cocoa (no. per ha)<br />
mean CV% mean CV% mean CV% mean CV% mean CV% mean CV%<br />
I1A 22 13 33 1 251 1.13 48.8 165 29 0 469 81 163<br />
A<br />
I1B 41 12 29 0 - 1.35 42.6 126 30 1 384 120 134<br />
I1C 19 15 27 5 45 2.01 23.3 107 26 5 436 244 99<br />
I2 15 9 57 5 78 1.66 24.1 218 50 11 190 129 54<br />
I3A 17 9 51 7 42 1.58 26.6 210 57 3 141 206 84<br />
A<br />
I3B 10 7 23 1 218 1.36 33.5 332 56 0 0 1017 77<br />
I4A 4 13 13 2 141 1.31 39.6 240 100 0 0 108 60<br />
A<br />
I4B 4 14 38 8 29 1.86 27.7 198 81 1 200 60 76<br />
II1A 6 15 40 2 92 1.44 46.3 144 26 280 87 167 165<br />
A<br />
II1B 5 17 34 1 100 1.49 23.1 150 29 128 49 51 35<br />
II1C 12 17 30 5 64 2.03 22.9 134 32 249 90 172 55<br />
II2 8 15 45 3 77 1.62 39.1 197 33 125 187 198 52<br />
II3A 14 11 38 6 47 1.74 29.4 131 63 332 88 341 103<br />
A<br />
II3B 11 14 42 5 47 1.92 21.6 150 56 173 97 366 91<br />
114<br />
N. LLAMANDA<br />
ET AL.
115<br />
As the palms attain the bearing stage, cattle is introduced into the plantation and<br />
grazed until the coconut trees become senescent (cropping system I). I With the<br />
coconut palms becoming still older (~60 years) and that their planting pattern gets<br />
distorted, food crops are introduced and/or a new generation of coconut trees<br />
interplanted along with the first generation palms. Cacao might also be intercropped<br />
along with coconuts at the beginning. However, as the cacao trees die eventually,<br />
cattle might be introduced into the plantation, and this system (System III) I then<br />
evolves like the previous one; alternately food crops can be inter-planted (System<br />
IV). V<br />
When the tree fallows are not managed, however, the coconut palms would be<br />
dominated by other woody perennial components (systems III and V where cacao<br />
was intercropped with coconut). ‘Key situations’ of these systems, corresponding to<br />
the trajectories for the juvenile coconut stage are I3A I , I2 I , II3A I and II2 I (Fig. 4).<br />
Moreover, at the beginning of the senescent stage if gaps arise between coconut<br />
trees through altered planting patterns, it could be utilized in different ways (I1B I and<br />
I ).<br />
II1B<br />
Cropping system II<br />
Cropping system I<br />
Cropping system III<br />
Cropping system IV<br />
Cropping system V<br />
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
I3A I2<br />
I1A I1B<br />
II3A II2<br />
II1A II1B<br />
0 5 20 60 100<br />
I3B<br />
I1C<br />
I4A<br />
I4B<br />
II1C<br />
II3B<br />
Coconut trees<br />
Fruit trees<br />
Food crops<br />
Coconut trees<br />
replanting<br />
Cocoa trees<br />
years after coconut<br />
tree planting<br />
Illustrations Cirad: N. Lamanda, M. Duportal<br />
Figure 4. Temporal dynamics of coconut-based agroforestry systems on Malo Island, Vanuatu.
116<br />
N. LLAMANDA<br />
ET AL.<br />
4. DISCUSSION<br />
The coconut plots on Malo Island had species diversity levels close to those noted in<br />
the multistrata agroforestry systems of the humid tropics, i.e., an average number of<br />
12 tree species per plot with a mean Shannon Weaver index of 1.57 (for a complete<br />
review of the reported floristic elements in homegardens see Kumar and Nair, 2004).<br />
Figure 5. Some structural groups described on Malo Island (Vanuatu) [I I 1A:<br />
Systematic<br />
planting of coconut trees (++) and no vertical differentiation of the vegetation profile;<br />
mixtures of coconut palms (Cocos nucifera), Mangifera indica a and Hibiscus tilaceus can,<br />
however, be seen in the rear end. I 3B:<br />
Coconut plots with significant species diversity.<br />
Distorted planning pattern and a vertical differentiation of the vegetation profile where<br />
coconut trees are dominant (e.g., a first generation of coconut trees dominates a younger<br />
generation and Annonaa spp. at the first level of the picture) and dominated (mainly by forest<br />
trees). I 1C:<br />
Coconut trees with a significant species diversity. Distorted planning pattern and<br />
a vertical differentiation of the vegetation profile. Food crops such as Musaa spp. constitute<br />
the lower stratum of the vegetation profile].<br />
A vertical stratification involving one to five strata is also characteristic of the<br />
structure of homegardens, one of the most common agroforestry systems in the<br />
tropics (Fernandes and Nair, 1986; Kumar and Nair, 2004). The vegetation structure
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
reported for the smallholder coconut plots on Malo Island is similar to that described<br />
for many smallholder coconut production systems in other locations, e.g., cattle<br />
grazing in the Pacific islands (Nair, 1983; Clarke and Thaman, 1993), multistory<br />
mixed species systems involving coconut and another cash crop such as cacao, or<br />
with food crops in South Asia, and in particular in India (for a comprehensive<br />
review of the agroforestry systems with coconut, see Nair, 1979; 1983; 1989).<br />
The situation of coconut plots was, however, not static and evolved throughout<br />
its development phases. We discerned five dynamic phases for the smallholder<br />
coconut-based agroforestry systems that corresponded to five major trajectories (Fig.<br />
4) from which two evolutionary patterns could be deduced. First, a perennial<br />
occupation of the cultivated land by the coconut palms, because of coconut<br />
replanting in cropping systems I, IIII and IV. V In these systems, the tree fallows<br />
gradually evolved into situations with one-to-three strata and a new coconut stand<br />
could be established in the original pattern after about 60 years of coconut<br />
cultivation. Second, a gradual return to tree fallows where coconut trees could<br />
gradually disappear because of the evolution of complex multistrata vegetation in<br />
which other tree species dominate (coconut-based agroforestry systems III and V). V<br />
This pattern of evolution could lead to a new cultivation cycle depending on the<br />
agroecological impacts (especially on soil fertility) and fallow duration (e.g., the<br />
food gardens).<br />
The smallholder coconut-based agroforestry systems have various economic/<br />
social functions too: (1) generating cash flow by copra and/or cocoa production,<br />
which incidentally, is the main source of income for most Maloese, (2) contribution<br />
to food security by producing fruits, nuts, leaves, roots, etc., that are a substantial<br />
source of food supply – and some of which have high nutritional value, thus adding<br />
to the nutritional security, (3) an inheritance pattern with plantations being passed<br />
down to the children, (4) a social function with copra harvesting by a working group<br />
called ‘kompagny”, and (5) a cultural role with the production of decorative,<br />
medicinal/‘magic’ species. Cattle grazing in coconut plots is also associated with (1)<br />
generation of cash income, (2) food production that constituted an important source<br />
of animal proteins, (3) a social function with cattle slaughtered for marriage and<br />
funeral ceremonies, (4) weed control and (5) a nutrient recycling function, e.g.,<br />
grazing and nutrient addition through dung and urine.<br />
In addition, the coconut-based agroforestry systems provide for ecological<br />
functions such as carbon sequestration (see Kumar, 2006); efforts to quantify this<br />
substantial potential of the plantations of Vanuatu are currently underway. Yet<br />
another advantage is in situ agrobiodiversity conservation, especially the high intraspecific<br />
variability and genetic diversity at plot level. For instance, seven named<br />
types of coconut trees were reported per ha of the smallholder plantations in Vanua<br />
Lava, another northern island of Vanuatu (Caillon, 2005). And a wide variety of<br />
breadfruit (Artocarpus ( altilis)<br />
is also cultivated in Vanuatu (~132 types; Walter,<br />
1989).<br />
Cultivating food crops in coconut gardens also might reduce the impact on tree<br />
fallows or forests by reducing the rate at which these are being cleared for food<br />
production. Furthermore, it represents a sustainable way of intensifying the current<br />
cropping practices. That is, with only 25% of the light and space being used by the<br />
117
118<br />
N. LLAMANDA<br />
ET AL.<br />
coconut palms, resources are often under-exploited in mature coconut plantations.<br />
Intercropping might be the best option for effectively utilizing these resources (Nair,<br />
1979; 1983). In the current context of Melanesian agriculture (with land shortages<br />
and a downward trend in the profitability of copra production), agroforestry, thus,<br />
appears to be a very attractive option for intensification of the smallholder coconut<br />
production systems, and in particular, the old plantations. Nowadays, in the western<br />
area of Malo, where there are land shortages due to human pressure, food crops such<br />
as banana, papaya, island cabbage, or strong yam are already being introduced on<br />
the farm boundaries, or in the distorted planting patterns of coconut trees in the older<br />
(~60 year-old) plantations (I1C I C in Figs. 3, 4 and 5). This situation, which is found at<br />
the senescent stage of coconut palms and in areas with high human population<br />
pressure, may constitute an innovation and a valid alternative to current land use<br />
problems.<br />
More intensive use of the older coconut plantations is possible by intercropping<br />
food crops or species with high economic value such as vanilla (Vanilla planifolia),<br />
that are adapted to the level of resources usually available under the canopy of the<br />
coconut palms. These species can provide a significant source of income or food.<br />
Moreover, food crops can be sold in the local markets that offer considerable<br />
potential for development and expansion due to the increase in urban populations. In<br />
isolated areas such as Melanesian islands, however, developing the production of<br />
species with high economic potential should be linked to niche-marketing<br />
opportunities and extension facilities to certify the quality attributes/organic origin<br />
of the produce.<br />
Existing smallholder copra production systems are also more complex than the<br />
large European coconut estates. Yet the development of copra production has led<br />
to a simplification of the pre-existing smallholder systems, a phenomenon called<br />
agrodeforestation (Clarke and Thaman, 1993), which had dramatic consequences on<br />
many Pacific islands. Therefore, it should be accompanied by another process of<br />
agroforestation, to avoid environmental disasters.<br />
6. FUTURE DIRECTIONS<br />
Characterizing the existing smallholder coconut-based agroforestry systems and<br />
their dynamics constitutes the first step towards evaluating their agro-ecological and<br />
agro-economical potentials, which is required to guide the future of these systems.<br />
Key situations, such as those where food crops are reintroduced into the coconut<br />
plots, are currently being studied to assess the possibility of more intensive use of<br />
the old coconut plantations. In particular, soil fertility levels (with organic matter<br />
indicators) and light availability in the vegetation profile and root occupation are<br />
being measured to estimate the degree to which various biophysical resources are<br />
used. Future studies should also take into account differences in soil fertility due to<br />
topographic differences (coral limestone in coastal area versus clay soils on hills),<br />
and economic evaluation of the coconut-based agroforestry systems to ensure that<br />
they match the farmers’ goals.
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
ACKNOWLEDGEMENTS<br />
119<br />
We are grateful to the Vanuatu Research and Training Centre (VARTC) and the<br />
Vanuatu Agriculture Extension Services and the Farming Support Association (FSA)<br />
for their logistical support and <strong>coop</strong>eration. We would also like to express our<br />
gratitude to the Maloese farmers for their warm welcome and infinite patience.<br />
ENDNOTE<br />
1. Allen M.G. 2001. Change and continuity: Land use and agriculture on Malo<br />
Island, Vanuatu. Master of science. Australian National University, Canberra,<br />
201p.<br />
REFERENCES<br />
Allen M. 2000. Subsistence or Cash Cropping ? Food Security on Malo Island, Vanuatu. In:<br />
Bourke R., Salisbury M.G., and Allen J.C. (eds), Food security for Papua New Guinea -<br />
Proceedings of Papua New Guinea ffood<br />
and nutrition 2000 Conference, pp 100 – 111.<br />
ACIAR Proceeding. PNG University of Technology, Canberra.<br />
Barrau J. 1955. L’agriculture vivrière mélanésienne. Commission du Pacifique Sud. Nouméa,<br />
206p.<br />
Bonnemaison J. 1996. Gens de Pirogues et Gens de la Terre- Les fondements géographiques<br />
d’une identité, l’archipel du Vanuatu- Livre 1. Orstom (eds). Paris, 460p.<br />
Caillon S. 2005. Pour une conservation dynamique de l’agrobiodiversité: gestion locale de la<br />
diversité variétale d’un arbre “des Blancs” (cocotier, Cocos nucifera L.) et d’une plante<br />
“des ancêtres” (taro, Colocasia esculenta (L.) Schott) au Vanuatu. Doctorat, Géographie,<br />
Université d’Orléans, Orléans, 789p.<br />
Caron P. 1997. Le zonage régional à dire d’acteurs. Connaître, représenter, planifier, agir, une<br />
méthodologie expérimentée dans le Nordeste du Brésil. In: Quelle géographie au CIRAD?<br />
Montpellier 10: 145 – 156.<br />
Clarke W.C. and Thaman R.R. (eds). 1993. Agroforestry in the Pacific Islands: systems for<br />
sustainability. United Nation University, Tokyo, 297p.<br />
Fernandes E.C.M. and Nair P.K.R. 1986. An evaluation of the structure and function of<br />
tropical homegardens. Agroforest Syst 21: 279 – 310.<br />
Institut de Recherches des Huiles (IRHO) 1969. Accroissement de la production de coprah<br />
aux Nouvelles Hébrides. Institut de Recherches des Huiles et Oléagineux (IRHO), Paris,<br />
154p.<br />
Krebs C.J. 1985. Ecology: the experimental analysis of distribution and abundance. Harper<br />
and Row Publisher, New York, 686p.<br />
Kumar B.M. 2006. Carbon sequestration potential of tropical homegardens. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 185 – 204. Springer Science, Dordrecht.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Labouisse J-P. 2004. Systèmes agraires et économie du cocotier au Vanuatu: historique et<br />
perspectives. Journal de la Société des Océanistes (CNRS-Musée de l’Homme), Paris,<br />
118: 11 – 33.<br />
Lamanda N., Malézieux E. et Martin P. 2004. Organisation spatiale et dynamique des<br />
systèmes de culture à base de cocotiers (Cocos nucifera L.) dans une île mélanésienne.<br />
Cahiers Agricultures 6: 459 – 467.
120<br />
N. LLAMANDA<br />
ET AL.<br />
Nair P.K.R. 1979. Intensive multiple cropping with coconuts in India. Paul Parey,<br />
Berlin/Hamburg, 148p.<br />
Nair P.K.R. 1983. Agroforestry with coconuts and other tropical plantation crops. In: Huxley<br />
P.A. (ed.), Plant research and agroforestry, pp 79 – 102. ICRAF, Nairobi.<br />
Nair P.K.R. (ed.). 1989. Agroforestry systems in the tropics. Kluwer, Dordrecht, 664p.<br />
Quantin P. 1982. Atlas des sols et de quelques données du milieu naturel: Atlas and<br />
exploratory notes. Orstom (eds), Paris, 37p.<br />
Walter A. 1989. Notes sur les cultivars d’arbres à pain à Vanuatu. Journal de la Société des<br />
Océanistes (CNRS-Musée de l’Homme), Paris, 1/ 2: 3 – 18.<br />
Weightman B. 1989. Agriculture in Vanuatu: An historical review. The British Friends of<br />
Vanuatu, Cheam, 320p.<br />
APPENDIX 1<br />
List of local, scientific names and uses of the conspicuous species reported in<br />
coconut plots on Malo Island, Vanuatu.<br />
List of conspicuous species Uses<br />
Scientific name Local name<br />
(bishlama)<br />
Abelmoschus manihot aeland<br />
kappish<br />
⊕ ⊕<br />
Ananas comosus pineapple ⊕ ⊕<br />
Sold Food Timber Fuel Used<br />
every<br />
day<br />
Eaten<br />
by<br />
animals<br />
Magic<br />
and<br />
medicinal<br />
uses<br />
Annona spp. korrosol ⊕ ⊕ ⊕ ⊕<br />
Artocarpus altilis breadfruit ⊕ ⊕ ⊕ ⊕ ⊕<br />
Barringtonia edulis navele ⊕ ⊕ ⊕ ⊕<br />
Canarium indicum nangaie ⊕ ⊕ ⊕ ⊕ ⊕<br />
Carica papaya paw paw ⊕ ⊕ ⊕ ⊕<br />
Citrus grandis pomelos ⊕ ⊕ ⊕<br />
Citrus limon lemon ⊕ ⊕ ⊕ ⊕<br />
Citrus reticulata mandarine ⊕ ⊕ ⊕<br />
Dioscorea spp. soft yam ⊕ ⊕ ⊕<br />
Discorea nummularia strong yam ⊕ ⊕<br />
Erythrina variegata narara ⊕ ⊕<br />
Helicona indica leaf lap lap ⊕ ⊕<br />
Hibiscus tiliaceus bourrao ⊕ ⊕ ⊕ ⊕ ⊕ ⊕<br />
Inocarpus fagiferus namambé ⊕ ⊕ ⊕<br />
Macaranga spp. navenue ⊕ ⊕ ⊕ ⊕<br />
Mangifera indica mango ⊕ ⊕ ⊕<br />
Manihot esculenta manioc ⊕ ⊕<br />
Metroxylon warburghii natangora ⊕ ⊕ ⊕<br />
Musa spp. banana ⊕ ⊕ ⊕ ⊕<br />
Pometia pinnata nandao ⊕ ⊕ ⊕ ⊕ ⊕
COCONUT-BASED AGROFORESTRY IN MELANESIA M<br />
Psidium guajava guava ⊕ ⊕ ⊕<br />
Saccharum officinarum sugarcane ⊕ ⊕<br />
Spondias dulis naos ⊕ ⊕ ⊕<br />
Vanilla<br />
planifolia/tahitensis<br />
Xanthosoma<br />
sagittifolium<br />
vanilla ⊕<br />
taro Fiji ⊕ ⊕ ⊕<br />
121
CHAPTER 8<br />
DIVERSITY AND DYNAMICS<br />
IN HOMEGARDENS OF SOUTHERN<br />
ETHIOPIA<br />
TESFAYE ABEBE 1 , K.F. WIERSUM 2 *, F. BONGERS 3<br />
AND F. STERCK 3<br />
1 Debub University, Awassa College of Agriculture, Ethiopia. 2 Forest and Nature<br />
Conservation Policy group, and 3 Forest Ecology and Management group,<br />
Wageningen University, The Netherlands; *E-mail: <br />
Keywords: Adaptability, Species composition, Socioeconomic change, Sustainability.<br />
Abstract. Most homegarden studies have focused on Asia, where homegardens constitute a<br />
component of a spatially separated farming system consisting of cultivated fields with staple<br />
food and/or commercial crops away from homes complemented by the homegardens with<br />
supplementary crops such as fruits and vegetables surrounding residential houses. In the<br />
highlands of East and Central Africa, another type of homegarden is found in the form of an<br />
integrated farming system within itself and without additional cultivated fields. In these<br />
‘integral’ homegardens, not only supplementary crops such as fruits and vegetables, but also<br />
staple food crops and cash crops are grown. The enset (Enset ventricosum) and coffee (Coffea<br />
arabica) homegarden system in southern Ethiopia is a typical example of such integral<br />
homegardens. An assessment of 144 of these homegardens was made to gain insights into<br />
their structure and vegetation composition and the relation between composition and<br />
geographic and socioeconomic factors. Four specific garden types are identified, which vary<br />
in commercial crop composition and diversity. These variations are related to farm size and<br />
access to roads and markets, and illustrate the dynamic character of homegardens. Overall, the<br />
diversity of the integral homegarden system seems to be somewhat lower than that of the<br />
‘complementary’ homegarden systems in Asia, probably due to the inclusion of light<br />
demanding staple food crops and a relatively large number of commercial crops. The dynamic<br />
pathways of the integral homegarden systems because of commercialization appear similar to<br />
reported trends in the ‘complementary’ homegarden systems in Asia. Although the<br />
composition of the homegardens is influenced by socioeconomic dynamics, overall the<br />
Ethiopian homegardens can be characterized as being ecologically and socioeconomically<br />
sustainable. This can be attributed not only to species diversity but also to the presence of two<br />
keystone species—coffee and enset.<br />
123<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 123–142.<br />
© 2006 Springer. Printed in the Netherlands.
124 TESFAYE T ABEBE ET AL.<br />
1. INTRODUCTION<br />
<strong>Homegardens</strong> have commonly been characterized as biodiverse and sustainable land<br />
use systems (Soemarwoto, 1987; Torquebiau, 1992; Kumar and Nair, 2004).<br />
Recently, it has been acknowledged that this does not mean that the structure and<br />
composition of homegardens should be assumed as being stable (Kumar and Nair,<br />
2004). From an ecological point of view, the production processes are not<br />
necessarily negatively affected by changes in vegetation structure and composition,<br />
if the nutrient cycling processes, hydrological conditions, and synergetic relations<br />
are not compromised. From a social point of view, the concept of sustainability<br />
incorporates the notion of adaptation to social change (Peyre et al., 2006). Similar to<br />
any land use system, homegardens are faced with constant pressure of change<br />
brought about by demographic, economic, technological, and social dynamics, and<br />
they are constantly adapted to changing livelihoods. Several studies in Asia indicate<br />
that with commercialization, often a gradual change from subsistence to commercial<br />
crops occurs in homegardens, while the crop diversity decreases (Michon and Mary,<br />
1994; Kumar and Nair, 2004; Peyre et al., 2006; Abdoellah et al., 2006).<br />
Most homegardens studies are focused on gardens that constitute a component of<br />
a spatially separated farming system consisting of cultivated fields away from<br />
homes complemented by the homegardens surrounding residential houses. In such<br />
multi-locational farming systems, homegarden production is mostly supplementary<br />
to the staple food production and mainly focuses on vegetables, fruits, and<br />
condiments (Wiersum, 2006; Soemarwoto, 1987; Hoogerbrugge and Fresco, 1993).<br />
The notion of tropical homegardens as components of integrated farming systems,<br />
which also include cultivated fields for staple food production, prevails in much of<br />
the homegarden literature. These ‘complementary’ homegardens typically consist of<br />
small (0.01 to 1 ha) plots around houses with a more or less randomly organized<br />
cropping pattern. A part of the garden may be devoted to ornamentals or tree crops.<br />
As these homegardens complement other components of the overall farming system,<br />
crop diversity and homegarden dynamics are influenced by the nature and<br />
characteristics of the other components of the overall farming system (Stoler, 1978;<br />
Karyono, 1990). However, in the highlands of East and Central Africa, a somewhat<br />
different type of homegardens exists in the form of an integrated farming system<br />
within itself without additional cultivated fields (Tesfaye Abebe, 2005). These<br />
‘integral’ homegardens consist of medium-scale (0.4 to ~3 ha) multipurpose farm<br />
fields around homes that form the principal means of livelihood for the households.<br />
In these gardens, not only supplementary crops such as vegetables, fruits,<br />
condiments, and/or medicinal crops, but also staple food crops and cash crops are<br />
cultivated. The motivating factor for this multiplicity of species is that farmers have<br />
no or very little additional land devoted to specialized types of production, for<br />
instance cereals. Consequently, these homegardens function as a total rather than a<br />
partial farming system. Most of the homegardens in the highlands of East Africa<br />
belong to this category (Fernandes et al., 1984; Okigbo, 1990; Oduol and Aluma,<br />
1990; Rugalema et al., 1994; Tesfaye Abebe, 2005). They have been much less<br />
intensively studied than the ‘complementary’ homegardens of Asia. An interesting<br />
question is whether the diversity and dynamics as observed in the ‘complementary’
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
125<br />
homegardens are also present in these ‘integral’ homegardens. This question is<br />
examined in this chapter by analyzing the structure and composition of the<br />
homegardens of southern Ethiopia as an example.<br />
2. HOMEGARDENS IN SOUTHERN ETHIOPIA<br />
The traditional agroforestry homegardens of southern Ethiopia are located at<br />
altitudes of 1500 to 2300 m above sea level where moisture and temperature<br />
conditions are favorable for agriculture. These gardens are popularly known as<br />
‘enset-coffee homegardens’ after the two major perennial crops dominating this<br />
system (Fig. 1; Tesfaye Abebe, 2005). Enset [Enset ventricosum (Welw.)<br />
Cheesman], sometimes called false-banana, is a multipurpose crop that provides<br />
subsistence food for about 10 million people in Ethiopia (Bezuneh and Feleke, 1966;<br />
Desalegn Rahmato, 1995; Almaz Negash, 2001). Because of the possibility to<br />
harvest this perennial crop in times of famine, it has been termed as a ‘tree against<br />
Figure 1. Coffee (Coffea arabica)-enset (Enset ventricosum) m homegarden in southern<br />
Ethiopia. This ‘integral’ homegarden is not leveled as usual in Asian homegardens (Photo:<br />
Tesfaye Abebe).<br />
hunger’ (Brandt et al., 1997). Coffee (Coffea arabica) is also a native crop, which is<br />
not only grown for household use, but also as a cash crop. Other components of this<br />
agroforestry system include roots and tubers, fruits, vegetables, cereals, spices, and<br />
other crops such as the stimulant chat (Chata edulis). Moreover, livestock is kept in<br />
the gardens and different tree species are grown to serve productive as well as
126<br />
TESFAYE T ABEBE ET AL.<br />
ecological functions. Structurally, the gardens resemble the coffee-banana<br />
agroforestry systems of Uganda (Oduol and Aluma, 1990) and northern Tanzania<br />
(Fernandes et al., 1984; Rugalema et al., 1994; Soini, 2005) with enset taking the<br />
position of banana.<br />
The enset-coffee homegardens have for centuries supported very dense<br />
populations in the mid-altitude highlands of southern Ethiopia (Kippie Kanshie,<br />
2002). Although some studies have been made on the system (Westphal, 1975;<br />
Okigbo, 1990; Tessema Chekun, 1997; Zemede Asfaw and Zerihun Woldu, 1997),<br />
still only limited information is available about the (variations in) diversity and<br />
composition as well as the dynamics of the system. For instance, the gardens have<br />
been mostly described as being predominantly subsistence-based, although the<br />
presence of coffee and chat is indicative of the fact – and the authors’ experiences<br />
support this – that these gardens are also used for commercial production.<br />
3. RESEARCH ON STRUCTURE AND COMPOSITION OF SOUTHERN<br />
ETHIOPIAN HOMEGARDENS<br />
In order to assess the structure and composition of the enset-coffee homegardens as<br />
well as the main factors influencing them, a study was conducted in the Sidama<br />
Figure 2. Map of Sidama administrative zone (southern Ethiopia) showing the study areas<br />
with location of selected woredas (or districts) and names of selected Peasant Associations<br />
within each woreda.
127<br />
administrative region of southern Ethiopia during 1999 to 2002 (Tesfaye Abebe,<br />
2005). This region is one of the most densely populated areas of the country with a<br />
population density of 320 persons km –2<br />
m . The most important agroecological zone in<br />
the area is locally known as Gammoje (Sidama) or Woyna-Dega (Amharic). This<br />
zone is situated between 1500 and 2300 m above sea level, and characterized by a<br />
moist to subhumid warm subtropical climate with average annual rainfall of 1000 to<br />
1800 mm, and a mean temperature of 15 to 20 o C. The dominant soils are Eutric<br />
Nitosols (corresponding to Alfisols in the USDA soil taxonomy). Within this zone,<br />
detailed data on homegarden composition were collected from 144 homegardens<br />
located in 12 different Peasant Associations (PA = smallest Ethiopian administrative<br />
unit) distributed over four Woredas or districts (Fig. 2). The administrative units<br />
were selected purposefully in order to systematically cover the range of geographic<br />
conditions in the study area; and, within each administrative unit, the homegardens<br />
were selected randomly. For each homegarden, data were collected on the size and<br />
layout and all species (except spontaneously grown weeds) were inventoried and<br />
enumerated. Through farmer’s interviews, data were also collected on physical<br />
and socioeconomic characteristics of the farms, such as altitude, distance to markets<br />
and roads (physical data collection in the field), and on household characteristics<br />
such as family size, labor force, age and educational status. The interviews also<br />
served to collect data on the production of various crops and their market prices.<br />
Tesfaye Abebe (2005) gives further details on that.<br />
4. STRUCTURE AND COMPOSITION OF HOMEGARDENS IN SIDAMA<br />
REGION<br />
4.1. Structure<br />
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
The size of the 144 selected homegardens varied from 0.18 to 7.46 ha with a mean<br />
size of 0.90 ha. These homegarden holdings included residential areas and<br />
specialized grazing areas (with mean share of 14% of the holding size), cultivated<br />
lands (mean 82%), and sometimes some specialized woodlots (average 4%; Fig. 3).<br />
A major variable influencing plot size was the wealth status of the households with<br />
average values of 0.55, 1.46 and 2.75 ha for poor, middle income, and rich farmers<br />
(according to the local Peasant Association classification) respectively. Coffee and<br />
enset dominate in over 50% of the homegarden area, while the other crops occupy<br />
much smaller areas. Cash crops such as chat, sweet potato (Ipomoea batatas) and<br />
pineapple (Ananas ( comosus)<br />
often are grown in special zones.<br />
Within the homegardens, the vegetation structure was not uniform; often zones<br />
distinguished by specific crop combinations were found. For instance, zones dominated<br />
by coffee mixed with fruit and other trees, enset, and miscellaneous auxiliary crops;<br />
zones dominated by enset mixed with vegetables and miscellaneous trees; zones<br />
with maize (Zea mays) mixed with other food crops; zones with cash crops such as<br />
chat; and residential and grazing zones. The diversity of homegardens can, therefore,
128<br />
not only be assessed based on species composition, but also of the area share of<br />
main crop components.<br />
Figure 3. Mean area share of major homegarden components in Sidama administrative zone,<br />
southern Ethiopia.<br />
4.2. Species composition<br />
TESFAYE T ABEBE ET AL.<br />
Overall, 198 species of cultivated crops (78) and trees (120) were recorded from the<br />
144 homegardens in four woredas. Within each woreda, the total number of plant<br />
species present in homegardens varied from 84 to 159 (Table 1) demonstrating<br />
significant intra-regional variations. The mean number of plant species per<br />
homegarden was 37, with values ranging from 15 to 78. Appendix 1 gives an<br />
overview of the recorded crop species. In addition to species diversity, a high level<br />
of genetic diversity was found with respect to the two major crops, enset and coffee,<br />
being represented by 42 and 24 cultivars respectively. <strong>Homegardens</strong> also included<br />
seven livestock species: cattle, goats, sheep, donkeys, horses, mules, and poultry<br />
(mainly chicken).<br />
Homegarden composition can also be characterized by the diversity of functional<br />
crop types. Besides miscellaneous tree species, 10 functional groups of plants were<br />
recognized: fruit crops (24%), root and tuber crops (16%), vegetables (15%),<br />
stimulant crops (10%), cereals (9%), pulses (6%), spices and condiments (5%), oil<br />
crops (3%), medicinal crops (3%), and miscellaneous crops (9%). Each functional<br />
crop type was represented by 3 to 15 species. Cereals and root/tuber crops provide<br />
carbohydrate-rich staple foods; fruits, vegetables, pulses, spices/condiments, and<br />
medicinal crops mostly yield supplementary food and household products; the<br />
stimulants and oil crops mostly serve as cash crops. Miscellaneous tree species<br />
provide fuel- and construction wood and serve as shade trees. This combination of
different functions of crops, coupled with the presence of livestock in the<br />
homegardens, illustrates its character of forming an integrated farming system.<br />
129<br />
Table 1. Number of crop, tree, and livestock species in the homegardens of four woredas in<br />
southern Ethiopia.<br />
Woreda (n) Crop Tree Total useful Livestock<br />
species species plant species species<br />
Dara (36) 56 72 128 5<br />
Aleta Wondo (48) 64 95 159 6<br />
Dale (36) 57 94 151 5<br />
Awassa Zurya (24) 33 51 84 6<br />
Overall combined 78 120 198 7<br />
n = number of homegardens sampled.<br />
4.3. Homegarden types<br />
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
In addition to species composition, the extent of area under major crops varied<br />
significantly among different geographic zones. Four homegarden types could be<br />
Figure 4. Detrended correspondence analysis (DCA) scatter plots of tree composition of the<br />
farms. The four homegarden types are indicated with different symbols and indicated by a<br />
different letter, and are spatially separated. A, Type 1 Enset-coffee-maize type; B, Type 2<br />
Enset-chat-maize-coffee type; C, Type 3 Enset-coffee-sweet potato type; D, Type 4 Ensetcoffee-maize-chat-pineapple<br />
type.
130<br />
TESFAYE T ABEBE ET AL.<br />
identified based on the extent of area under major crops in different PAs (Tesfaye<br />
Abebe, 2005). This comparative deductive assessment of crop data for the four types<br />
is compared with the results of a Detrended Correspondence Analysis of tree species<br />
present in the homegardens. DCA reduces the multidimensional space of a speciesabundance<br />
matrix into a two-dimensional one. We used DCA as implemented in<br />
CANOCO 1 . The first axis represents the main variation in species composition, the<br />
second axis the main variation once the first axis variation is removed. The<br />
homegarden types are indicated in the DCA graph (Fig. 4). The spatial separation of<br />
the homegarden types in the graph indicates that the two methods (deductive<br />
assessment of crop data and DCA of tree species data) resulted in a similar<br />
categorization into four homegarden types having the following characteristics (see<br />
Tables 2 and 3):<br />
1. The enset-coffee-maize type. In a large part of the research area (almost 60% of<br />
all inventoried homegardens), the homegardens belong to this original type in<br />
which coffee and enset dominate on about 75% of the farmland. In addition,<br />
maize is grown on about 10% of the land. As reflected by their high wood<br />
volume, trees form an important component in the system (Appendix I). Species<br />
diversity is relatively high with a mean of 41 cultivated crop and tree species.<br />
These homegardens are predominantly subsistence-oriented with enset and<br />
maize serving as main staple food crops and coffee serving as a cash crop. The<br />
overall financial value (based on production amounts and market values) of the<br />
combined annual yields amount to Birr 5084 ha –1 , which is relatively low (1<br />
Birr ~ 0.1 US$).<br />
2. The enset-coffee-maize-sweet potato type is present in 8% of all sampled<br />
homegardens. It is even more subsistence-oriented than the first homegarden<br />
type. The share of the staple crop enset is relatively lower than in type 1.<br />
Instead, farmers produce mainly maize and sweet potato as staple foods. The<br />
proportion of land devoted to coffee as a cash crop is much lower than in type 1.<br />
Some farmers are cultivating eucalyptus (Eucalyptus spp.) as an alternative cash<br />
crop. This homegarden type has the highest species richness (43) of crops and<br />
trees. The overall financial value of the annual yields was lowest with Birr 4362<br />
ha –1 .<br />
3. The enset-chat-maize-coffee type is found in 16% of all sampled homegardens.<br />
It is much more cash-oriented than the types 1 and 2. Staple food production<br />
dominates in 56% of the land with maize occupying more land than enset<br />
production. The importance of coffee as a cash crop is low, and chat has taken<br />
over this role. The diversity of plant species is relatively low, however, the<br />
number of livestock per farm is the highest (3.4) of all types. This can be<br />
attributed to the higher farm income of farmers (overall annual financial value<br />
of all crops = Birr 6802 ha –1 ) which enables them to buy cows and feed them to<br />
produce milk for home consumption and the market.<br />
4. The enset-coffee-maize-chat-pineapple type was represented in 8% of all<br />
sampled homegardens. This homegarden type accommodates a relatively<br />
balanced proportion of the different major crops. This garden type has the<br />
lowest area share of grazing and housing lands, and the highest proportion of<br />
croplands, where enset production dominates. The staple food crops enset,
131<br />
maize, and sweet potato occupy 41% of the land area against 46% for the cash<br />
crops. In addition to coffee and chat, pineapple is an important cash crop.<br />
Species diversity of this type is relatively low, but higher than in type 3. The<br />
overall financial value of all crop annual yields is high with Birr 6809/ha.<br />
Table 2. Area share of main crops in different homegarden types of four woredas in southern<br />
Ethiopia.<br />
Homegarden type (n)<br />
Area coverage of different crops 1 (%)<br />
enset coffee maize chat sweet<br />
potato<br />
pineapple<br />
others<br />
Enset-coffee-maize (84) 29.1 46.5 10.5 0.6 1.2 0.3 12.2<br />
Enset-coffee-maize-sweet<br />
potato (12)<br />
17.2 27.2 33.0 0.8 10.6 0 11.2<br />
Enset-chat-maize-coffee<br />
(24)<br />
24.8 13.7 31.6 19.8 1.4 0 8.7<br />
Enset-coffee-maizepineapple<br />
and chat (24)<br />
23.5 31.1 12.2 6.5 5.3 8.5 13.1<br />
Mean 26.4 36.6 16.4 4.5 2.6 1.6 11.9<br />
n=number of homegardens sampled. 1 Percentage area coverage of different crops was<br />
calculated considering the crop areas only. That t is, residential and grazing areas and separate<br />
woodlots were not included in the calculation. Overall area share including these are shown in<br />
Fig. 3.<br />
Homegarden types (n)<br />
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
Table 3. Composition of different homegarden types of four woredas in southern Ethiopia.<br />
Number of crop<br />
and tree species<br />
Number of<br />
livestock<br />
species<br />
Number of<br />
livestock<br />
(TLU ha –1 )<br />
Mean SD Mean SD Mean SD<br />
Enset-coffee-maize (84) 41 a<br />
12.3 2.3 a 0.9 2.1 b 1.9<br />
Enset-coffee-maize-sweet<br />
potato (12)<br />
43 a<br />
12.2 2.0 a 0.4 1.9 b 1.0<br />
Enset-chat-maize-coffee (24) 25 b 5.6 2.1 a 0.7 3.4 a 3.6<br />
Enset-coffee-maize-chatpineapple<br />
(24)<br />
30 b 7.9 2.2 a 0.6 1.7 b 1.6<br />
Mean 37 12.0 2.2 0.8 2.2 2.2<br />
F test ( (p)<br />
< 0.001 ns < 0.05<br />
n = number of homegardens sampled; SD = standard deviation; TLU = tropical livestock unit;<br />
ns = not significant. Homegarden types with different letters differ significantly (F test and<br />
Duncan’s Multiple Range test, p < 0.05); p = probability level of significance.
132<br />
TESFAYE T ABEBE ET AL.<br />
4.4. Factors influencing presence of different homegarden types<br />
The different types of homegardens were not evenly distributed over woredas<br />
(Tesfaye Abebe, 2005): type 2 was found in only one PA of Dale woreda, type 3<br />
was found in Awasa Zurya woreda only, type 4 was found in one PA in Dara and in<br />
one PA in Aleta Wondo. Type 1 was most extensively found, in three woredas. This<br />
indicates that the presence of different homegarden types cannot be explained by<br />
variation in physical conditions only; but socioeconomic conditions might account<br />
for a significant extent of such variations. For instance, the homegardens of type 1<br />
are located far from major roads, while homegardens of type 4 have good access to<br />
roads, which facilitates the sale of homegarden products. Homegarden type 3 is<br />
located in areas with a very high population density, which necessitated an increase<br />
in staple food production (e.g., maize). The impact of several ecological and<br />
socioeconomic factors on homegarden composition was further tested by means of<br />
multiple step-wise regressions between crop and tree diversity and possible<br />
explanative factors. The factors that were included in this analysis were: altitude and<br />
slope of the farm, farm size, farm labor force, involvement in off-farm activities and<br />
distance to major roads and markets (Tesfaye Abebe, 2005). Among these, the<br />
following two factors emerged as the most important determinants of homegarden<br />
diversity (Table 4).<br />
Farm size: Although a decrease in farm size was not significantly correlated with<br />
overall crop diversity, it negatively affected the relative proportion of a homegarden<br />
covered by cash crops, indicating how smallholders give priority to produce food<br />
crops rather than cash crops. Also, species richness of trees and livestock decreased<br />
with decreasing farm size. Small landholders grew the same number of crop species<br />
as the large holders; but with increasing land size, farmers increased the number of<br />
tree species. The density of dominant native timber and multipurpose species such<br />
as Podocarpus falcatus, Cordia africana and Milletia ferruginea also decreased with<br />
decreasing farm size, while that of fast-growing eucalyptus increased because of the<br />
need for wood for home consumption as well as for income generation.<br />
Access to major roads: Although access to highways did not significantly<br />
correlate with overall crop diversity, it affected significantly the area share of the<br />
major crops. The share of annual crops, mainly maize, increased at the expense of<br />
enset. Also the importance of the new cash crops chat and pineapple increased,<br />
while the traditional cash crop coffee declined. Proximity to major roads also<br />
negatively affected the richness in tree species. The share of native and multipurpose<br />
trees declined with increased road access, but the share of eucalyptus increased. This<br />
reflects the ability of eucalypts to grow fast and produce wood for consumption as<br />
well as income generation.<br />
These two factors are, however, not static, but depend on socioeconomic<br />
development. They can logically be related to the processes of population growth<br />
and commercialization. It can, therefore, be concluded that under the current local<br />
conditions, these two developments have a major impact on the dynamics in<br />
homegarden structure and composition. In respect to the first factor, it should be<br />
remembered that agroforestry homegardens of southern Ethiopia already carry a<br />
very dense population of 300 to 600 persons per km 2 . Its high growth rate (2.2%) is
133<br />
likely to increase the fragmentation of farmlands. The resulting increasingly smaller<br />
farms may lead to a reduction of the perennial crop and tree components as well as<br />
livestock. Regarding commercialization, it appears that the access to road networks<br />
often results in a gradually greater emphasis on commercial crops and crop<br />
specialization in homegardens. Consequently, the share of the perennial crops and<br />
native tree species tend to decline with proximity to highways and, hence, access to<br />
markets.<br />
Table 4. Multiple linear regression of species richness and number of livestock on physical<br />
and socioeconomic environments of homegardens in southern Ethiopia.<br />
Factors<br />
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
Species richness<br />
Crops Trees Livestock<br />
No. of<br />
livestock<br />
(TLU)<br />
Physical environment<br />
Altitude (1520 – 2040 m above sea level) ns ns ns 0.19*<br />
Slope (0 – 45%) ns 0.14* ns ns<br />
Socioeconomic environment<br />
Distance to markets (0.04 – 6.0 km) 0.17* ns ns ns<br />
Distance to highway (0.02 – 26 km) ns 0.35*** 0.17* ns<br />
Farm size (0.18 – 7.46 ha) ns 0.42*** 0.28*** 0.48***<br />
Farm labor force (2 – 12) 0.18* ns ns ns<br />
Population density (2 – 35 inhabitants/ha of<br />
farmland)<br />
–<br />
0.20* –0.17* ns ns<br />
Involvement in off-farm work (yes/no) ns 0.14* ns ns<br />
Parenthetical values under “factors” denote the range for each parameter. R 2 values species<br />
richness of crops, trees and livestock were 0.15, 0.53 and 0.11 respectively and that of TLU<br />
was 0.48; * = p < 0.05; ** = p < 0.01; *** = p
134<br />
here as well. In the first place, the differences in lowland/highland location may<br />
impact species diversity with humid lowland homegardens having more biodiversity<br />
than highland homegarden systems (Wiersum, 2006). In the second place, one<br />
should take care in comparing the diversity figures, because of differences in the<br />
types of plant species considered. Some reports considered all plant species<br />
including ornamentals and sometimes weeds. For instance, Mendez et al. (2001)<br />
reported a total of 324 plant species in the homegardens of Nicaragua, out of which<br />
180 (56%) were ornamentals. Likewise, 219 plant species occurred in one village in<br />
West Java and 60 (27%) were ornamentals. As mentioned earlier, in the present<br />
study only deliberately grown crops and trees were recorded. Due to such<br />
differences in inventory, broad comparisons of species diversity in homegardens in<br />
agroecologically and socioeconomically different regions have several drawbacks,<br />
and should be considered as providing only indicative information.<br />
Ecological<br />
zone<br />
Humid<br />
lowlands<br />
Table 5. Species richness of selected homegardens in the tropics.<br />
Location Total no. of<br />
plant species<br />
West Java,<br />
Indonesia<br />
219 (60<br />
ornamentals)<br />
Average no.<br />
of plants per<br />
homegarden<br />
Sources<br />
56 Soemarwoto (1987);<br />
Soemarwoto and<br />
Conway (1991)<br />
39 De Clerck and<br />
Castillo (2000)<br />
70 (22–106) Mendez et al. (2001)<br />
Quintanana Roo, 150 useful<br />
Mexico<br />
plants<br />
Nicaragua 324 (180<br />
ornamentals)<br />
Santa Rosa , 168 35 (18–74) Padoch and De Jong<br />
Peruvian Amazon<br />
(1991)<br />
Humid Kandy, Sri Lanka 125 (93 usable) 46 (37–65) Perera and Rajapakse,<br />
lowlands<br />
1991<br />
to mid Kerala, India 127 woody 22 Kumar et al. (1994)<br />
altitudes<br />
species<br />
Highlands Chagga, Tanzania 111 (58 woody na Fernandes et al.<br />
and 53 herbs)<br />
(1984)<br />
Bukoba, Tanzania 57 na Rugalema et al.<br />
(1994)<br />
Wolayita and 60 14.4 Zemede-Asfaw and<br />
Gurage, southern<br />
Zerihun-Woldu<br />
Ethiopia<br />
(1997)<br />
Sidama, southern 198 crop and 37 (15–78) Tesfaye Abebe (2005)<br />
Ethiopia tree species<br />
na = not available.<br />
TESFAYE T ABEBE ET AL.
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
135<br />
Regarding the pattern in changing homegarden composition in the Sidama region<br />
in relation to decreasing plot size and commercialization respectively, similar trends<br />
have also been observed in ‘complementary’ homegarden systems (Kumar and Nair,<br />
2004; Peyre et al., 2006; Wiersum, 2006). For the ‘integral’ Chagga homegardens in<br />
Tanzania, changing livelihoods due to dynamics in socioeconomic conditions as<br />
well as market prices for garden products are reported to affect the composition of<br />
the gardens (Soini, 2005). We hypothesize that these trends are stronger in the<br />
‘integral’ homegarden system compared to the ‘complementary’ type because the<br />
former essentially incorporates cash crops, while this is not necessarily the case in<br />
the latter.<br />
6. IMPLICATIONS OF HOMEGARDEN COMPOSITION AND DYNAMICS<br />
FOR SUSTAINABILITY<br />
The description of the homegardens in southern Ethiopia illustrates that their<br />
structural characteristics are similar to the general features of tropical homegardens.<br />
The multispecies composition is often considered as a basic feature contributing to<br />
sustainability (Kumar and Nair, 2004). However, the presence of different types of<br />
homegardens illustrates that the homegarden composition is not always similar, but<br />
that it varies in response to socioeconomic differences and changes. Thus, when<br />
considering the sustainability of homegardens, a differentiation between ecological<br />
sustainability and socioeconomic sustainability seems warranted (Peyre et al., 2006).<br />
6.1. Homegarden composition and ecological sustainability<br />
Many studies have discussed the relation between species diversity in homegardens<br />
and their ecological sustainability (Soemarwoto, 1987; Torquebiau, 1992; Kumar<br />
and Nair, 2004). In the case of the Sidama homegardens, the presence of animal<br />
species is also a noteworthy phenomenon. Although such presence has been noted in<br />
several studies (e.g., Soemarwoto, 1987; Okafor and Fernandes, 1987), the animal<br />
component of homegardens is often neglected. However, our data demonstrate that<br />
livestock form an important component of the system (see also the Mesoamerican<br />
gardens described by Montagnini, 2006). In addition to their economic contribution<br />
by fulfilling various functions such as providing food in the form of milk and meat,<br />
traction and transport, they also play an important ecological role providing manure<br />
for the improvement of soil fertility and crop productivity. The animals contribute<br />
towards the maintenance of a closed nutrient cycling system with minimum dependence<br />
on external inputs such as fertilizers. The plant species diversity contributes toward<br />
the maintenance of animals. For instance, in the dry season, when fodder grass is in<br />
short supply, the animals are fed with immature thinned-out plants and leaves of<br />
enset, banana and other plants, as well as crop residues.<br />
Thus, within the Sidama homegardens not only the diversity in crop species and<br />
related multilayered vegetation system, but also the inclusion of animals in the<br />
system contributes toward their ecological sustainability. Moreover, the gardens also<br />
demonstrate that the stability of the system should not exclusively be related to its
136<br />
TESFAYE T ABEBE ET AL.<br />
diversity, but can also be attributed, at least in part, to the specific characteristics of<br />
the two main components: enset and coffee. As an evergreen perennial crop, enset<br />
gives a permanent shade to understorey crops, including coffee. Soil management is<br />
facilitated by the use of enset residues as a mulching material. Coffee is an ideal<br />
complementary crop to enset. Not only is it architecturally and ecologically<br />
compatible with enset, but the harvest of both enset and coffee involve only selected<br />
plant parts and do not involve major export of soil nutrients. Thus, enset and coffee<br />
can be considered as keystone species contributing to ecological sustainability of the<br />
system. In ecological studies, the role of keystone species in maintaining ecosystem<br />
stability has received some attention (Mills et al., 1993; Khanina, 1998), but the<br />
notion of keystone species has still received little attention in agroforestry research.<br />
6.2. Homegarden composition and socioeconomic sustainability<br />
The maintenance of high species diversity in the Sidama homegardens also contributes<br />
to socioeconomic stability. As in other homegarden systems, the diversity of crop, tree,<br />
and livestock species with different uses and production cycles enables year-round<br />
production of different products, reduces risk of production failure, allows spreading of<br />
labor-use and flexibility, and enables efficient cycling of locally available resources, thus<br />
reducing dependence on external inputs (Kumar and Nair, 2004). In addition, the<br />
Sidama homegardens also incorporate several specific features in respect to<br />
socioeconomic sustainability. They not only have high species diversity, but also a<br />
high diversity in functional crop types; notable is the presence of both staple food<br />
crops and cash crops in addition to the more usual supplementary homegarden<br />
crops. The basic food crops (enset and maize), which are rich in carbohydrates, are<br />
supplemented by pulses, vegetables, fruits, and animal products that provide<br />
proteins, fats, and vitamins, and by trees that provide resources for construction and<br />
household energy. Also cash crops are incorporated in the homegarden, not only<br />
coffee, but also chat and pineapple. The proportion of subsistence and cash crops is<br />
often adjusted to meet the household requirements. Moreover, the spreading of risk<br />
from crop failures is not only facilitated by the crop diversity, but also by the<br />
inclusion of enset. The flexibility in harvesting enset for staple food production has<br />
been indicated as one of the main reasons why the Southern Highlands are relatively<br />
free of hunger (Desalegn Rahmato, 1995; Brandt et al., 1997).<br />
Thus, similar to the ecological sustainability, the socioeconomic sustainability<br />
cannot only be explained by species diversity, but also by the specific features of the<br />
two key species enset and coffee. Enset is both a food crop and a provider of<br />
different products such as fiber and fodder. It is therefore ideally suited to lowexternal<br />
input agricultural production systems, while its high productivity and<br />
multiple functions provide sustenance for a very dense population which is often<br />
two to three times higher than that in the cereal-based systems found in other parts<br />
of Ethiopia. Moreover, due to its perennial production cycle, enset can serve well as<br />
an excellent drought-relief crop. Coffee serves as a main cash crop supplementing<br />
the mainly subsistence-oriented enset production. The combined production allows<br />
for a good safety net in times of crop or market failures. Also, processing and<br />
marketing of coffee creates employment for many people. Consequently, not only
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
from an ecological point of view, but also from a socioeconomic point of view,<br />
coffee and enset can be considered as keystone species.<br />
6.3. Impact off system dynamics on sustainability<br />
137<br />
Even if the Sidama homegardens can be characterized as being sustainable, this does<br />
not mean that they do not change. The system is affected by decreasing farm size<br />
resulting from population growth and increased commercialization. The shift from<br />
the traditional enset-coffee systems towards inclusion of other food and cash crops<br />
has diversified the diet and increased household incomes. But the expansion of<br />
open-field food crops, such as maize and sweet potato, and of monocultural cash<br />
crops, such as chat and pineapple, are not only causing a gradual loss of species<br />
diversity and tree biomass, but also a gradual decrease in the dominance of the two<br />
key species enset and coffee. This results in a gradual reduction of the ecological<br />
benefits of these integrated and complex systems, e.g., by decreasing soil cover and<br />
thus increasing erosion hazards, as well as a reduction of the keystone enset species<br />
serving as ‘a tree against hunger’ in favor of quickly producing cash crops. Although<br />
the hazards of such changes in vegetation structure and composition could<br />
potentially be offset by more intensive management practices including use of<br />
external inputs, in case that no proper adaptive management activities are undertaken<br />
this may threaten the long-term sustainability of the homegardens. In view of the<br />
call for stimulating new development of forest-analogous land use systems<br />
combining production and biodiversity conservation (Wiersum, 2004), attempts<br />
should be made to integrate new crops into the existing multistory systems without<br />
affecting its biodiverse nature and without losing essential keystone species.<br />
7. CONCLUSIONS<br />
The enset-coffee homegarden system can be considered as an integral homegarden<br />
system as it forms a spatially delineated farming system in contrast to the more<br />
commonly studied homegarden systems that are spatially complementary to<br />
cultivated fields and have a supplementary role to the overall family farming system.<br />
Nonetheless, both types of systems have several common features. The diversity of<br />
crops that are predominantly perennial in nature, with high diversity of trees and the<br />
presence of livestock allow a multitude of ecological interactions among the<br />
homegarden components and allow ecological sustainability. Moreover, the species<br />
richness combined with presence of different functional crop groups permit a<br />
balanced year-round production of both subsistence and cash crops.<br />
The enset-coffee homegarden systems also have some characteristics that are<br />
different from those of the more common spatially integrated (or supplementary)<br />
homegarden systems. Due to the absence of additional fields for staple food production<br />
or cash crop cultivation, the enset-coffee systems form a sort of “complete” farming<br />
system, producing a much higher proportion of basic food and cash crops than in the<br />
‘complementary’ homegarden systems. The system is characterized by the presence<br />
of two crops, enset and coffee, which are keystone species, due to their important
138<br />
TESFAYE T ABEBE ET AL.<br />
economic and ecological roles. The large number of varieties of both species reflects<br />
their great importance in the system. The combination of these two, mutually<br />
compatible, native perennial crops and their dominance in the systems are essential<br />
features of these homegardens.<br />
In a similar manner as reported on complementary homegarden systems, the<br />
recent developments in land use systems resulted from increasing commercialization<br />
and continuing population growth affects the enset-coffee homegarden system. The<br />
growing population requiring basic foods has resulted in a gradual replacement of<br />
enset by annual staple food crops. The advent of commercialization has resulted in<br />
the development of new lucrative cash crops such as chat and pineapple requiring<br />
monoculture-cropping practices. These patterns have led to the decline in the areas<br />
of enset, coffee, and other trees. The decline in the share of these perennial<br />
components and their replacement particularly with annual crops could reduce some<br />
of the multiple benefits derived from these integrated and complex traditional<br />
systems, but the impact of such changes on long-term sustainability of the system is<br />
speculative, at best.<br />
Within homegarden studies, little attention has been given to differences in<br />
homegarden structure and function in relation to their position either in the overall<br />
farming system or to the role of keystone species in respect to the sustainability of<br />
these systems. As demonstrated by the features of the Sidama homegarden systems,<br />
these aspects deserve further research attention.<br />
ENDNOTE<br />
1. Ter Braak C.J.F. and Smilauer P. 1998. CANOCO reference manual and user’s<br />
guide to CANOCO for Windows: Software for canonical community ordination<br />
(version 4). Centre for Biometry, CPRO-DLO, Wageningen, 351p.<br />
REFERENCES<br />
Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo S. and Parikesit. 2006.<br />
Commercialization of homegardens in an Indonesian village: vegetation composition and<br />
functional changes. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A<br />
time-tested example of sustainable agroforestry, pp 233 – 250. Springer Science,<br />
Dordrecht.<br />
Almaz Negash. 2001. Diversity and conservation of enset (Enset ventricosum (Welw.)<br />
Cheesman) and its relation to household food and livelihood security in southwestern<br />
Ethiopia. PhD dissertation, Wageningen University, Wageningen, 247p.<br />
Bezuneh T. and Feleke A. 1966. The production and utilization of the genus Ensete in<br />
Ethiopia. Econ Bot 20: 65 – 70.<br />
Brandt S.A., Spring A., Hiebsch C., McCabe J.T., Endale Tabogie, Mulugeta Diro, Gizachew<br />
Wolde Michael, Gebre Yntiso, Masyoshi Shigeta and Shiferaw Tesfaye. 1997. The tree<br />
against hunger. American Association for the Advancement of Science, Washington, DC,<br />
56p.<br />
De Clerck F.A.J. and Negreros-Castillo P. 2000. Plant species of traditional Mayan<br />
homegardens of Mexico as analogs for Multistrata agoforests. Agroforest Syst 48:<br />
303 – 317.
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
139<br />
Desalegn Rahmato 1995. Resilience and vulnerability: Enset agriculture in southern Ethiopia.<br />
J Ethiopian Stud 28: 23 – 51.<br />
Fernandes E.C.M., Oktingati A. and Maghembe J. 1984. The Chagga homegardens: a<br />
multistoryed agroforestry cropping system on Mt. Kilimanjaro, Northern Tanzania.<br />
Agroforest Syst 2: 73 – 86.<br />
Hoogerbrugge I.D. and Fresco L.O. 1993. Homegarden systems: Agricultural characteristics and<br />
challenges. International Institute for Environment and Development, Gatekeeper series no.<br />
39, London, 23p.<br />
Karyono 1990. <strong>Homegardens</strong> in Java: their structure and function. In: Landauer K. and Brazil<br />
M. (eds), <strong>Tropical</strong> home gardens, pp 138 – 146. United Nations University Press, Tokyo.<br />
Khanina L. 1998. Determining keystone species. Conservation Ecology 2(2):R2 [online]:<br />
URL:http:// www.consecol.org/Journal/vol2/iss2/resp2 (last accessed: December 21,<br />
2005).<br />
Kippie Kanshie T. 2002. Five thousand years of sustainability? A case study on Gedeo land<br />
use. PhD dissertation, Wageningen University, Wageningen, 295p.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Kumar B.M, George S.J. and Chinnamani S. 1994. Diversity, structure and standing stock of<br />
wood in the homegardens of Kerala in peninsular India. Agroforest Syst 25: 243 – 262.<br />
Mendez V.E., Kok L. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use, and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96<br />
Michon G. and Mary F. 1994. Conversion of traditional village gardens and new economic<br />
strategies of rural households in the area of Bogor, Indonesia. Agroforest Syst 25:<br />
31 – 58.<br />
Mills L.S., Soule M.E. and Doak D.F. 1993. The keystone-species concept in ecology and<br />
conservation. BioScience 43: 219 – 224.<br />
Montagnini F. 2006. <strong>Homegardens</strong> of Mesoamerica: biodiversity, food security, and nutrient<br />
management. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-<br />
tested example of sustainable agroforestry, pp 61 – 84. Springer Science, Dordrecht.<br />
Oduol P.A. and Aluma J.R.W. 1990. The banana (Musa spp.)-Coffea robusta: traditional<br />
agroforestry system of Uganda. Agroforest Syst 11: 213 – 226.<br />
Okafor J.C. and Fernandes E.C.M. 1987. Compound farms of southeastern Nigeria: A<br />
predominant agroforestry homegarden system with crops and small livestock. Agroforest<br />
Syst 5: 153 – 168.<br />
Okigbo B.N. 1990. <strong>Homegardens</strong> in tropical Africa. In: Landauer K. and Brazil M. (eds),<br />
<strong>Tropical</strong> home gardens, pp 21 – 40. United Nations University Press, Tokyo.<br />
Padoch C. and Jong W. de 1991. The house gardens of Santa Rosa: Diversity and variability<br />
in an Amazonian agricultural system. Econ Bot 45: 166 –175.<br />
Perera A.H. and Rajapakse R.M.N. 1991. A baseline study of Kandian forest gardens of Sri<br />
Lanka: structure, composition and utilization. For Ecol Manag 45: 269 – 280.<br />
Peyre A., Guidal A., Wiersum K.F. and Bongers F. 2006. Homegarden dynamics in Kerala,<br />
India. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 87 – 103. Springer Science, Dordrecht.<br />
Rugalema G.H., Okting’ati A. and Johnson F.H. 1994. The homegarden agroforestry systems<br />
of Bukoba district, Northwestern Tanzania. 1. Farming systems analysis. Agroforest Syst<br />
26: 53 – 64.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: A traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development,<br />
pp 157 – 170. International Council for Research in Agroforestry, Nairobi.
140<br />
TESFAYE T ABEBE ET AL.<br />
Soemarwoto O. and Conway G.R. 1991. The Javanese homegarden. J Farming Syst Res Extn<br />
2: 95-118.<br />
Soini E. 2005. Changing livelihoods on the slopes of Mt. Kilimanjaro, Tanzania: challenges<br />
and opportunities in the Chagga homegarden system. Agroforest Syst 64: 157 – 167.<br />
Stoler A. 1978. Garden use and household economy in rural Java. Bull Indonesian Econ Stud<br />
14: 85 – 101.<br />
Tesemma Chekun 1997. The culture of coffee in Ethiopia. Agroforest Today 9: 19 – 21.<br />
Tesfaye Abebe 2005. Diversity in homegarden agroforestry systems of southern Ethiopia.<br />
Wageningen University, the Netherlands, <strong>Tropical</strong> Resource Management Paper No. 59,<br />
143p.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Westphal E. 1975. Agricultural Systems in Ethiopia. Centre for Agricultural Publication and<br />
Documentation, Wageningen, 278p.<br />
Wiersum K.F. 2004. Forest gardens as an ‘intermediate’ land use system in the nature-culture<br />
continuum: characteristics and future potential. Agroforest Syst 61: 123 – 134.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.<br />
Zemede Asfaw and Zerihun Woldu 1997. Crop associations of homegardens in Welayita and<br />
Gurage in southern Ethiopia. Sinet: Ethiopian J Sci 20: 73 – 90.<br />
APPENDIX I<br />
List of crop species in Sidama homegardens listed under their main functional crop<br />
group, sorted by frequency (% of homegardens in which the species is found, out of<br />
a total of 144).<br />
Scientific name Family English common Freque<br />
Roots and tubers<br />
name ncy (%)<br />
Enset ventricosum (Welw.) Cheesman Musaceae enset, false 100<br />
Dioscorea alata L. Dioscoreaceae yam 59<br />
Colocasia esculenta (L.) Schoot Araceae taro 51<br />
Ipomoea batatas (L.) Lam. Convolvulaceae sweet potato 42<br />
Manihot esculenta Cranz. Euphorbiaceae cassava 8<br />
Solanum tuberosum L. Solanaceae potato 6<br />
Beta vulgaris L. Chenopodiaceae beet root 5<br />
Daucus carota L. Apiaceae carrot 2<br />
Dioscorea bulbifera L.<br />
Vegetables<br />
Dioscoreaceae aerial yam 1<br />
Brassica integrifolia (West.) O.E. Brassicaceae kale 99<br />
Cucurbita pepo L. Cucurbitaceae pumpkin 83<br />
Capsicum frutescens L. Solanaceae hot pepper 43<br />
Brassica oleraceae L. Brassicaceae Ethiopian kale 33<br />
Lycopersicon esculenta L. Solanaceae tomato 16
HOMEGARDENS OF SOUTHERN ETHIOPIA<br />
141<br />
Capsicum annuum L. Solanaceae chilly 12<br />
Solanum villosum L. Solanaceae African 9<br />
Allium cepa L. Alliaceae shallot 3<br />
Brassica oleracea L. var. capitata Brassicaceae cabbage 3<br />
Lactuca sativa L. Asteraceae head lettuce 2<br />
Allium porrum L. Alliaceae leek 2<br />
Allium sativum L.<br />
Pulses<br />
Alliaceae garlic 1<br />
Phaseolus vulgaris L. Fabaceae common bean 99<br />
Phaseolus lunatus L. Fabaceae lima bean 30<br />
Vicia faba L. Fabaceae faba bean 3<br />
Pisum sativum L. Fabaceae pea 2<br />
Cajanus cajan (L.). Mill.<br />
Cereals<br />
Fabaceae pigeon pea 2<br />
Zea mays L. Poaceae maize 100<br />
Sorghum bicolor (L.) Moench Poaceae sorghum 31<br />
Eragrostis tef (Zucc.) Trotter Poaceae tef 6<br />
Hordeum vulgare L. Poaceae barley 2<br />
Triticum sativum L.<br />
Fruits<br />
Poaceae wheat 2<br />
Persea americana Mill. Lauraceae avocado 88<br />
Musa paradisiaca L. Musaceae banana 83<br />
Psidium guajava L. Myrtaceae guava 43<br />
Citrus sinensis (L.) Osbeck Rutaceae sweet orange 38<br />
Casimora edulis La Llave & Lex. Rutaceae white sapota 29<br />
Ananas comosus (L.) Merr. Bromeliaceae m<br />
pine apple 24<br />
Prunus persica (L.) Batsch Rosaceae peach 15<br />
Carica papaya L. Caricaceae papaya 15<br />
Passiflora edulis Sims. Passifloraceae passion fruit 13<br />
Annona reticulata L. Annonaceae bullock’s heart 11<br />
Mangifera indica L. Anacardiaceae mango a<br />
8<br />
Cyphomandra betacea (Cav.) Sendt. Solanaceae tree tomato 8<br />
Fragaria vesca L. Rosaceae strawberry 5<br />
Citrus aurantifolia (Christm.) Swingle Rutaceae lime 4<br />
Punica granatum L.<br />
Stimulants<br />
Punicaceae pomegranate 1<br />
Coffea arabica L. Rubiaceae coffee 100<br />
Chata edulis (Vahl.) Forssk. ex Endl. Celastraceae khat 57<br />
Nicotiana tabacum L.<br />
Spices and condiments<br />
Solanaceae tobacco 8<br />
Capsicum frutescens L. Solanaceae hot pepper 43<br />
Ruta chalepensis L. Rutaceae rue 17<br />
Appendix 1 (contd.)
142<br />
TESFAYE T ABEBE ET AL.<br />
Capsicum annuum L. Solanaceae chilly 12<br />
Afromomum korarima (Braun) Jansen Zingiberaceae false cardamom 6<br />
Zingiber officinale L. Zingiberaceae ginger 3<br />
Rosmarinus offwinalis L. Lamiaceae rosemary 3<br />
Ocimum basilicum L. Lamiaceae sweet basil 3<br />
Lippia adonensis Hochst. ex Walp. Verbenaceae 3<br />
Piper nigrum L. Piperaceae black pepper 1<br />
Nigella sativa L.<br />
Oil crops<br />
Ranunculaceae black cumin 1<br />
Ricinus communis L. Euphorbiaceae castor 43<br />
Brassica carnata A. Br. Brassicaceae Ethiopian 19<br />
mustard<br />
Arachis hypogaea L. Fabaceae ground nut 9<br />
Carthamus tinctorius L. Asteraceae safflower 3<br />
Linum usitatissimum L.<br />
Medicinal plants<br />
Linaceae linseed 1<br />
Ocimum gratissimum L. Lamiaceae 13<br />
Foeniculum vulgare Mill. Apiaceae fennel 2<br />
Otostegia integrifolia Benth. Lamiaceae 1<br />
Artemisia absinthium L.<br />
Fragrance plants<br />
Asteraceae absinthe 1<br />
Ocimum gratissimum L. Lamiaceae 13<br />
Lippia adoensis Hochst. ex Walp Verbenaceae 12<br />
Cymbopogon citratus (DC.) Stapf.<br />
Other crops<br />
Poaceae lemon grass 1<br />
Rhamnus prinoides L’herit Rhamnaceae rhamnus 70<br />
Saccharum officinarum L. Poaceae sugarcane 54<br />
Lagenaria siceraria (Mol.) Stardl. Cucurbitaceae bottle gourd 5<br />
Agave sisalana Perr. Agavaceae sisal 4<br />
Gossypium herbaceum L. Malvaceae cotton 2<br />
Sorghum dochna (Forsk.) Snowden Poaceae sweet stalk<br />
sorghum<br />
1<br />
Pennisetum purpureum Schumach 1<br />
Poaceae elephant grass 12<br />
Chloris gayana Kunth 1<br />
Poaceae Rhodes grass 2<br />
Desmodium unicinatum (Jacq.) DC 1<br />
1<br />
introduced forage crop.<br />
Leguminoseae desmodium 1
CHAPTER 9<br />
HOMEGARDEN PLANT DIVERSITY IN<br />
RELATION TO REMOTENESS FROM<br />
URBAN CENTERS: A CASE STUDY FROM<br />
THE PERUVIAN AMAZON REGION<br />
A. WEZEL 1 AND J. OHL 2<br />
1 Institute of Landscape and Plant Ecology (320), University of Hohenheim, 70593<br />
Stuttgart, Germany; E-mail: . 2 School of Biological<br />
Sciences, University of East Anglia, Norwich NR4 7TJ, United Kingdom<br />
Note: Adapted from: Wezel A. and Ohl J. 2005. Does remoteness from urban centres<br />
influence plant diversity in homegardens and swidden fields: a case study from the<br />
Matsiguenka in the Amazonian rainforest of Peru. Agroforestry Systems 65: 241 – 251.<br />
Keywords: Floodplains, Indigenous people, Manu National Park, Shifting cultivation, Slash<br />
and burn, Swidden agriculture<br />
Abstract. Swidden cultivation is the traditional agricultural system in most parts of the Amazonian<br />
rainforest, and in many situations swiddens lead to the establishment of homegardens. In a remote<br />
area of the Manu National Park, Peru, such a system was investigated in two indigenous<br />
Matsiguenka communities for diversity of cultivated plants on swidden fields and in homegardens.<br />
The cultivated plants were identified from two to four plots per field in 46 fields in a total of 126<br />
survey plots and 19 homegardens. Altogether 71 species were found in the homegardens and 25 in<br />
the swidden fields. Cassava (Manihot ( esculenta)<br />
was dominant in the cultivated fields, whereas<br />
fruit trees such as peach palm (Bactris gasipaes), guava (Psidium guajava), and Inga edulis; and<br />
cotton (Gossypium barbadense) and a medicinal plant (Cyperus s sp.) predominated more than 75%<br />
of the homegardens. Species diversity increased steadily with age (length of cultivation) of the<br />
swidden fields. Diversity of species cultivated in the homegardens was low compared to other<br />
studies reported from the Amazon. This seemed to be due to remoteness from urban areas, relative<br />
isolation and consequently little interaction of the farmers with outside communities, and easy<br />
availability of plant products from nearby forests. Although these findings appear to contradict the<br />
premise that subsistence farming in such remote areas encourages farmers to produce a broad<br />
variety of species and, therefore, remoteness from urban centers increases species richness on<br />
farms; the extent to which the situation is impacted by easy availability of plant products from<br />
nearby forests, however, was not investigated in this study. In contrast to the homegardens,<br />
swidden fields in this study did not show any difference in species richness compared to other<br />
reported studies.<br />
143<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 143–158.<br />
© 2006 Springer. Printed in the Netherlands.
144<br />
A. WEZEL W AND J. OHL<br />
1. INTRODUCTION<br />
The indigenous agricultural system in most parts of the Amazonian rainforest is<br />
based on swidden cultivation (also known as slash-and-burn or shifting cultivation;<br />
Dufour, 1990). Besides cropping on swidden fields and cultivation of plants in<br />
homegardens, other activities of resource use such as hunting, fishing, and forest<br />
extraction are employed by rural peoples in the Amazonian floodplains and on the<br />
terra firme (elevated river terraces or hills).<br />
In a basic swidden cultivation cycle, the first step is to select a new field site in<br />
primary or secondary forest areas (Thrupp et al., 1997). The forests are then cleared<br />
and burnt, and different crops planted. Cropping is normally abandoned after 2 to 3<br />
years because of declining yields, partly due to increasing weed pressure. Finally,<br />
secondary forests develop on the fallowed fields and might be cleared again after<br />
several years. Analyses of different swidden systems in the Peruvian and Colombian<br />
Amazon including lists of plant species cultivated on swidden fields were reported<br />
by various authors previously (e.g., Johnson, 1983; Hiraoka, 1986; 1989; Eden and<br />
Andrade, 1987; Salick, 1989; Dufour, 1990; Coomes and Burt, 1997). In most such<br />
systems, cassava (Manihot esculenta) is the main crop during the first three years. In<br />
some cases, this is followed by another 2 to 3 years of plantain and banana (Musa<br />
spp.) production. Finally, the fields are fallowed (often called swidden fallows)<br />
when forest regrowth takes place, although fruit trees and other tree species still<br />
occupy the sites (e.g., Hiraoka, 1986; 1989).<br />
In systems with permanent or semi-permanent settlements, different plants are<br />
cultivated around the dwelling units. These homegardens are generally characterized<br />
by different vegetation strata (trees, shrubs and herbs) composed of annual and<br />
perennial agricultural crops and small livestock within the house compounds<br />
(Fernandez and Nair, 1986). Normally, the whole tree-crop-animal unit is<br />
intensively managed by family labor. Homegarden systems around the world have<br />
been comprehensively analyzed and summarized by Kumar and Nair (2004). In<br />
Peru, homegardens in the villages on the Amazonian floodplains have been<br />
investigated by Padoch and de Jong (1991) and Lamont et al. (1999), and those in<br />
the upland locations (upper Amazon) by Salick (1989). Works 1 described the<br />
homegardens of a small but steadily growing town in the Alto Mayo region of upper<br />
Amazon.<br />
Ohl (2004) analyzed the traditional economic system as well as the influences of<br />
new economic activities of two indigenous communities in a remote area in the<br />
Peruvian Amazon. Among other things, that study focused on the extent of changes<br />
currently taking place and its implications for sustainable resource use in the Manu<br />
National Park. The research of the broader project included an investigation on<br />
different aspects such as the socioeconomic situation of the households, land use<br />
practices, hunting and fishing activities as well as health care issues. This chapter<br />
focuses on the plant aspects of this broad study: plants cultivated on fields and in<br />
homegardens for meeting the basic food needs of the indigenous population, and the<br />
diversity as well as similarities among such plants in the two communities. The<br />
results were used to examine if remoteness from the urban centers increases plant<br />
diversity in homegardens and swidden fields, as often reported in the literature.
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
2. STUDY AREA<br />
145<br />
The study area is located in the lowland part of the Manu National Park in southeastern<br />
Peru (Fig. 1). The National Park comprises an area of about 1.72 million ha.<br />
The area studied within the National Park receives 2000 – 2500 mm annual precipitation<br />
2 . The average annual temperature is around 23°C. Two seasons are distinguished:<br />
a dry period from May to September and a rainy period from October to<br />
April. The vegetation is characterized by different types of tropical lowland<br />
rainforests on both periodically inundated alluvial plains (varzea) and on more<br />
elevated river terraces or hills (terra firme). Predominant soil types are Fluvisols and<br />
Gleysols (according to FAO classification) on the alluvial plains and Cambisols,<br />
Luvisols, and Acrisols on river terraces and on the hilly terrain 3 .<br />
This region is also the most sparsely populated area 4 of Peru with 0.1<br />
persons/km². Two Matsiguenka communities in the Fitzcarrald district of the Manu<br />
province were selected for the study following several visits to them during 2000 –<br />
2002; the community Tayakome (123 people) is located at 11°43.8’ S/71°38.8’ W<br />
(368 m altitude) and Yomibato (183 people) at 11°48’ S/71°54.4’ W (419 m<br />
altitude). Both are remote locations that can be reached only by boat on the river<br />
Manu, taking 1 to 4 days from Boca Manu, the nearest important settlement. Besides<br />
Tayakome and Yomibato, a third, smaller settlement of colonists from the Andes<br />
exists in the Manu National Park.<br />
2.1. Matsiguenka villages<br />
The two selected villages are relatively new communities founded in 1968 (Tayakome)<br />
and 1978 (Yomibato). A few decades ago, the Matsiguenka were mainly<br />
semi-nomadic hunter-gatherers, who used various sites along their treks to cultivate<br />
food crops (Johnson, 1989). After establishing permanent settlements, most people<br />
started to practice slash-and-burn, hunting, and fishing around their villages.<br />
Nevertheless, there are still some people, mainly in Yomibato, who wander around,<br />
moving between two or three distant huts spending a week or two in each, for<br />
successful hunting. In general, every household consists an average of 5.9 persons in<br />
Tayakome and 6.1 in Yomibato (range: 4 to 26 members per household in both<br />
villages: a household is defined as a group of people eating regularly together in one<br />
house), who actively cultivate 2.0 fields (a ‘field’ in this context is an area of land<br />
cleared in the primary or secondary forest for cropping) in Tayakome (0.73 ha) and<br />
1.8 fields in Yomibato (0.92 ha). The cropping period for a single field varies<br />
normally between 2 to 3 years during which cassava is mainly cultivated. In the<br />
third and fourth years, the swiddeners mostly harvest species such as plantains<br />
/bananas or papaya (Carica papaya), which were planted in the first year. After that,<br />
they return every now and then to the swidden fallows to collect mainly fruits from<br />
previously planted trees.<br />
The Matsiguenka often cultivate different crops in homegardens around their<br />
houses. Some chickens and ducks are also kept. Eggs are sometimes used, but their<br />
meat is only consumed in case of food shortage. Hunting trips are made only by
Figure 1. Location of the Manu National Park in Peru and the two villages studied.<br />
146<br />
A. WEZEL AND<br />
W J. OHL
.<br />
147<br />
men, mainly during the rainy season. Fishing is the main activity in the dry season<br />
when rivers are low. In addition, joint fishing is done in small rivers using fish<br />
poison.<br />
3. METHODS<br />
Cultivated plants were counted from 2 to 4 plots per field in a total of 46 fields in<br />
both communities; each plot was 5 x 10 m in size. There were a total of 126 survey<br />
plots in 1 to 2 year-old fields, implying that if the distribution of cultivated plants in<br />
the field appeared to be homogeneous, two plots were randomly selected and four if<br />
the distribution was non-homogeneous. Cultivated plants were counted in 19<br />
homegardens as well, and were organized into functional groups according to their<br />
preferred uses. Ornamental plants and timber species were not included in this study.<br />
Normally the homegardens are located within a zone of 5 to 25 m around the<br />
houses. The boundaries of most homegardens were quite evident because of the<br />
regular weeding that takes place within this zone. In few cases, however, it was<br />
difficult to distinguish between homegardens and the adjacent fields. Three age<br />
classes were recognized for the homegardens: young = up to 2 years since establishment;<br />
medium-old = 3 to 10 years; and old = more than 10 years.<br />
Plants which could not be directly identified were collected in a field herbarium.<br />
Local names provided by the owners were referenced to corresponding scientific<br />
names following Brack Egg (1999), and in few cases with the help of Shepard and<br />
Chicchón (2001) and Baer (1984). The collected plant samples were then verified<br />
with the help of botanists from the Universities of Cusco and Lima. Scientific plant<br />
names follow Brack Egg (1999).<br />
The similarity of species composition of fields and homegardens between the two<br />
communities was calculated using the Sørensen coefficient of similarity (Müller-<br />
Dombois and Ellenberg, 1974), according to the formula (2A/B+C) x 100 (where A =<br />
number of species common to two villages; B = total number of species in village 1<br />
and C = total number of species in village 2). For the comparison of species<br />
composition between fields and homegardens within each community also the same<br />
formula was used (where A = number of species common in fields and homegardens;<br />
B = total number of species in fields and C = total number of species in homegardens).<br />
4.1. Swidden fields<br />
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
4. RESULTS<br />
The dominant crop cultivated by the Matsiguenka on all fields is cassava (for details<br />
see Wezel and Ohl, 2005). It is consumed daily and used frequently to brew cassava<br />
beer. In Tayakome, 19 different cassava varieties were cultivated, and as many as 56<br />
in Yomibato. Other important plants included plantains/bananas and maize (Zea<br />
mays). Plants such as sugarcane (Saccharum officinarum), sweet potato (Ipomoea<br />
batatas), Lonchocarpus sp. (used as fish poison), papaya and cush-cush yam<br />
(Dioscorea trifida) were also cultivated, but less frequently; pineapple (Ananas (<br />
comosus) and guava (Psidium guajava) were grown only occasionally.
148<br />
Furthermore, some species such as plantains/bananas, sugarcane, Lonchocarpus<br />
sp. and cotton (Gossypium barbadense) were found much more frequently in the<br />
second year of cultivation than the first. By contrast, maize is only planted during<br />
the first year. Maize, sugarcane, sweet potato, and papaya are cultivated generally<br />
more often in Tayakome than in Yomibato. In total, 25 species of crops were found<br />
in the study villages, with 21 in each village; besides, 18 and 24 species were found<br />
in one- and two-year-old swidden fields respectively. Species composition of the<br />
fields was comparable between the study villages with 81% similarity (Sørensen’s<br />
coefficient).<br />
4.2. <strong>Homegardens</strong><br />
A. WEZEL W AND J. OHL<br />
In Matsiguenka homegardens, fruit trees such as peach palm (Bactris gasipaes),<br />
guava, and Inga edulis; cotton and a medicinal plant (Cyperus sp.) predominated<br />
more than 75% of the observations (Table 1). Other fruit-producing trees and shrubs<br />
such as cashew (Anacardium ( occidentale),<br />
Pouteria caimito, mango (Mangifera<br />
indica), papaya, plantains/bananas, lemon (Citrus limon) and orange (C. sinensis)<br />
were also frequent with an occurrence of over 50%. Tubers such as cassava and<br />
Xanthosoma poeppigii, as well as pineapple and sugarcane were also noted on 50%<br />
of the gardens. One homegarden had many medicinal plants (but only some could be<br />
identified). Guava, Inga edulis, and Pouteria caimito were the most frequent tree<br />
species in the homegardens.<br />
Papaya, cassava, and pineapple were most frequent in young homegardens and<br />
their frequency decreased with age (Table 1). By contrast, Inga edulis, Pouteria<br />
caimito, orange, Genipa americana, mandarin (Citrus reticulata) and Crescentia<br />
cujete were found more often in the older homegardens. Out of 71 species found in<br />
all homegardens studied, 25 species, not considering medicinal plants, were<br />
cultivated in young homegardens, 27 in medium-aged and 50 species in old<br />
homegardens. For medicinal plants, the numbers were 26, 29 and 66 species,<br />
respectively. Eighteen species were found only in old homegardens. The almost<br />
exclusive occurrence of medicinal plants in the category of ‘old homegardens’ might<br />
be related to the fact that they were mainly found in one homegarden whose owner<br />
had a broad knowledge of the medicinal uses of such plants. The average number of<br />
species per garden increased with age: 14 in young, 16 in medium, and 20 in old<br />
homegardens.<br />
On an average, 18 species of plants were found in the homegardens of the two<br />
villages (range: 7 to 31). In both communities, the homegardeners mostly cultivated<br />
the same species, although a few disparate species with low occurrences (e.g.,<br />
coconut palms, Cocos nucifera, with less than 15% occurrence) were noted. This is<br />
also reflected in the Sørensen coefficients of similarity, which showed that 75% of<br />
the species were similar in both villages when medicinal plants were excluded, and<br />
65% when they were included.<br />
Similarity of species between swidden fields and homegardens was 46% for both<br />
Yomibato (46% without medicinal plants) and Tayakome (54% without medicinal<br />
plants). Plants cultivated widely in swidden fields and homegardens were cassava,<br />
plantain/banana and, to a lesser extent, sugarcane.
Functional groups/species Occurrence (%) in different<br />
age classes of homegardens 1<br />
Matsiguenka name Local name English name Family<br />
Fruit trees 2<br />
Table 1. Cultivated plants in homegardens of the Matsiguenka in the Amazonian rainforest of Peru.<br />
Young Medium Old<br />
(n = 3) (n = 4) (n = 12)<br />
Bactris gasipaes 67 100 92 kuri pijuayo, chonta peach palm Arecaceae<br />
Psidium guajava 100 75 92 komashki guava guava Mirtaceae<br />
Inga edulis 67 75 92 intsipa guallaba, pakay Mimosaceae<br />
Anacardium occidentale 67 75 67 kasho marañon cashew Anacaridaceae<br />
Carica papaya 100 50 58 tinti papaya papaya Caricaceae<br />
Pouteria caimito 0 25 83 segorikashi caimito Sapotaceae<br />
Mangifera indica 0 75 67 manko mango mango Anacaridaceae<br />
Musa paradisiaca 3<br />
33 75 50 parianti platano plantain/banana Musaceae<br />
Citrus limon 33 75 58 irimoki limon lemon Rutaceae<br />
Citrus sinensis 0 50 67 naranka naranja orange Rutaceae<br />
Table 1 (cont.)<br />
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
149
Functional groups/species Occurrence (%) in different<br />
age classes of homegardens 1<br />
Matsiguenka name Local name English name Family<br />
Young Medium Old<br />
(n = 3) (n = 4) (n = 12)<br />
Persea americana 33 0 58 tsivi, inchatoki palta avocado Lauraceae<br />
Genipa americana 0 25 58 ana huito Rubiaceae<br />
Citrus reticulate 0 25 58 tasharina mandarina mandarin Rutaceae<br />
Annona chirimola 67 0 50 tsirimoito chirimoya cherimoya Anonaceae<br />
Mauritia flexuosa 33 50 25 koshiki, achoariki aguaje Arecaceae<br />
Crescentia cujete 0 25 50 pajo, pamoko calabaza tree gourd Bignoniaceae<br />
Citrus limetta 0 0 50 lima 4 lima sweet lime Rutaceae<br />
Artocarpus altilis 0 50 25 pan de arbol 4 pan de arbol breadfruit Moraceae<br />
Attalea phalerata 0 25 25 tsigaro chapaja Arecaceae<br />
Inga sp. 0 0 25 intsipa orompiano pacay “colombiano” Mimosaceae<br />
Citrus grandis 0 0 17 toronja 4 pomela, toronja pummelo Rutaceae<br />
Cocos nucifera 0 0 17 koko coco coconut palm Palmae<br />
Oenocarpus bataua 0 0 17 sega(ki) ungurahui Arecaceae<br />
Solanum sessiliflorum 0 0 17 kokona cokona Solanaceae<br />
Bactris sp. 0 0 8 manataroki chontilla Arecaceae<br />
150 A. WEZEL AND<br />
W J. OHL
Tubers<br />
Manihot esculenta 100 75 42 sekatsi yuca cassava Euphorbiaceae<br />
Xanthosoma poeppigii 33 25 67 tsanaro uncucha cocoyam Araceae<br />
Calathea allouia 33 0 25 shonaki dale-dale Guinea arrowroot Marantaceae<br />
Dioscorea trifida 33 0 17 magona sacha papa cush-cush yam Dioscoreaceae<br />
Pachyrhizus ahipa 0 0 8 poi ashipa yam bean Fabaceae<br />
Ischnosiphon killipii 0 0 8 shirina sachaoca Marantaceae<br />
Ipomea batatas 5<br />
0 0 8 koriti camote sweet potato Convolvulaceae<br />
Vegetables and pulses<br />
Capsicum pubescens 33 0 17 tsitikana aji Solanaceae<br />
Lycopersicon cf.<br />
tomate 4<br />
tomate tomato Solanaceae<br />
peruvianum 33 0 0<br />
Cyclanthera pedata 33 0 0 iritsima poreatsiri caihua wild cucumber Cucurbitaceae<br />
Cucurbita moschata 0 50 0 kemi zapallo seminole pumpkin Cucurbitaceae<br />
Solanum mommosum 0 25 8 ivoniaro nuña huaca Solanaceae<br />
Cajanus cajan 0 0 8 ivinkoki poroto de palo pigeonpea Fabaceae<br />
Capsicum annuum 0 0 8 masekagana aji chili Solanaceae<br />
Lablab niger 0 0 8 tsitstita<br />
poroto hyacinth bean Fabaceae<br />
Table 1 (cont.)<br />
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
151
Functional groups/species Occurrence (%) in different<br />
age classes of homegardens 1<br />
Matsiguenka name Local name English name Family<br />
Young Medium Old<br />
(n = 3) (n = 4) (n = 12)<br />
Others<br />
Gossypium barbadense 100 75 75 ampei algodon cotton Malvaceae<br />
Ananas comosus 100 75 50 tsirianti piña pineapple Bromeliaceae<br />
Saccharum officinarum 67 75 42 impongo caña azugar sugarcane Poaceae<br />
Bixa orellana 33 100 33 potsoti achiote annato Bixaceae<br />
Banisteriopsis sp. 33 75 25 kamarampi ayahuasca Malpighiaceae<br />
Lonchocarpus sp. 33 25 25 kogi, shimaaro, barbasco Fabaceae<br />
komo<br />
Brugmansia sp. 33 0 25 saaro, jayapa, toé Solanaceae<br />
kepigari<br />
Gynerium sagittatum 0 25 8 chakopi caña de flecha arrow cane Poaceae<br />
Nicotiana tabacum 33 0 0 seri tabaco tobacco Solanaceae<br />
Cedrela odorata 0 0 8 santaviri, santari cedro Meliaceae<br />
Curcuma longa 0 0 8 porikano palillo curcuma Zingiberaceae<br />
152 A. WEZEL AND<br />
W J. OHL
Hymenaea courbaril 0 0 8 koveni azucar huayo Caesalpinaceae<br />
Miconia sp. 0 0 8 savotaroki Melastomataceae<br />
Crescentia sp. 0 25 0 oeshinta, pamoko Bignoniaceae<br />
Medicinal plants 6<br />
Cyperus sp. 100 100 67 ivenkiki piri piri Cyperaceae<br />
Jatropha gossypiifolia 0 0 25 piñon piñon negro Euphorbiaceae<br />
Eryngium foetidum 0 0 8 sacha culantro sacha culantro Apiaceae<br />
Plukenetia volubilis 0 0 8 mani sachamani Euphorbiaceae<br />
Justicia pectoralis 0 0 8 viriorioshi Acanthaceae<br />
Cordia nodosa 0 0 8 matiagiroki Boraginaceae<br />
Martinella obovata 0 0 8 pocharo Bignoniaceae<br />
Eleutherine bulbosa 0 0 8 kapirokotapini yahuar piri-piri Iridaceae<br />
1 Young: 0 to 2 years, Medium: 3 to 10 years, Old: >10 years; 2 Inclusive of Musa paradisiaca and Carica papaya; 3 No distinction was between<br />
plantains and bananas. Anyhow, plantains were reported to be mainly cultivated; 4 The Matsiguenka use the common name as they have no own name<br />
for this species; 5 In few cases it might be a second Ipomoea species; 6 Additionally, nine different plants said to possess medicinal value were noted in<br />
the homegardens; but they could not be identified other than by their local names and are, therefore, not reported.<br />
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
153
154<br />
A. WEZEL W AND J. OHL<br />
5. DISCUSSION<br />
5.1. Remoteness of homegardens and richness of cultivated species<br />
One question that is discussed in homegarden studies is the relation between species<br />
richness and distance to urban markets (Fernandez and Nair, 1986; Padoch and<br />
de Jong, 1991; Lamont et al., 1999). Often it is mentioned that urban-market<br />
pressure results in decreased total species diversity in the homegardens, whereas<br />
subsistence farmers in remote areas are compelled to produce diverse products and,<br />
therefore, species diversity increases in remote areas (but see: Lamont et al., 1999).<br />
In the present study, differences in total species numbers between homegardens<br />
of the two study villages were small, ranging from 49 in Yomibato to 58 in<br />
Tayakome. These numbers, however, were lower than the species richness reported<br />
by Lamont et al. (1999) from north-east Peru, where they documented 104, 111, and<br />
125 different species in three villages located 3 to 10 hours away by boat ride from<br />
the nearest urban center. Padoch and de Jong (1991), however, recorded as many as<br />
168 species in the homegardens of another de-tribalised and market-influenced<br />
village in north-east Peru. It needs to be noted that Lamont et al. (1999) included<br />
species for construction in their analysis, whereas Padoch and de Jong (1991)<br />
included species for construction as well as ornamental plants; albeit t their numbers<br />
were relatively low. The remoteness of the villages and ethnical differences seem to<br />
be important in determining total species richness. For example, in the village with<br />
the highest species number, residents included former members and descendants of<br />
at least four tribal groups as well as a few families who trace their ancestry to<br />
Europe (Padoch and de Jong, 1991). Peoples of mixed European and Amazonian<br />
ancestry live in the village with 125 homegarden species (Lamont et al., 1999). The<br />
other two villages are considered native communities although peoples of mixed<br />
ancestry have migrated to one of these villages over the years. Similar results are<br />
mentioned by Works 1 with more than 120 different species in the homegardens of<br />
Moyobamba. This town is a steadily growing urban center in the upper Amazon area<br />
where many newcomers settled in recent decades. In contrast, the Mastiguenka<br />
communities are native, without mixture of different tribes and located most<br />
remotely from the urban centers. The Matsiguenka homegardens can thus be<br />
characterized as relatively “pristine” with fewer cultivated plants. This seems to be<br />
similar to the situation of the Andoke and Witoto Indians in the Colombian Amazon,<br />
who cultivate only 33 species in their homegardens (Eden and Andrade, 1987). The<br />
contact of the Andoke and Witoto Indians with the outside world is relatively<br />
limited and local production is largely subsistence-oriented. Although the<br />
Matsiguenka exchange cultivated plants with other Matsiguenka communities, they<br />
are presently not able to sell any plant products from homegardens in the urban<br />
market because of remoteness and transportation problems. This could be a<br />
disincentive for planting many commercial species. Although Matsiguenka<br />
communities rely on subsistence production, they do not cultivate a broad variety of<br />
different species. Furthermore, these communities are of relatively recent origin,
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
155<br />
having been founded only in 1968 (Tayakome) and 1978 (Yomibato). The<br />
Matsiguenka still collect many products from the forest including medicinal plants.<br />
This seems to be the reason why only few medicinal plants were found in most<br />
homegardens. About 55% of the medicinal plants that were noted in this study were<br />
found in one single homegarden whose owner is a traditional healer and he planted<br />
medicinal plants from the forest as well as from other places in his homegarden.<br />
In the present study, 25 species were found in the one-and two-year-old swidden<br />
fields, 21 in each village. Johnson (1983) reported 26 species in his random samples.<br />
For young swiddens in the Colombian Amazon, Eden and Andrade (1987) recorded<br />
a total of 38 cultivated species, with an average of 12 per field, and Dufour (1990)<br />
reported nine different crops per field – but that could be because only four plots<br />
were studied. Contrary to the situation in the homegardens, however, differences<br />
in species numbers in the swidden fields were relatively small and factors such as<br />
remoteness did not seem to have an influence. This might be due to the fact that<br />
in swidden fields the most common crops and fruit trees are cultivated, whereas in<br />
homegardens, factors such as remoteness and cultural difference play a much more<br />
important role in species selection.<br />
5.2. Frequently cultivated species in the homegardens and swidden fields<br />
Many plants found in the Matsiguenka homegardens with high frequency are also<br />
typical plants of homegardens throughout the tropics in the world, e.g., plantains/<br />
bananas, guava, mango, avocado (Persea americana), papaya, Citrus s spp., breadfruit<br />
(Artocarpus ( altilis),<br />
cassava, and sugarcane (Jensen, 1993; De Clerck and Negreros-<br />
Castillo, 2000; Méndez et al., 2001; Wezel and Bender, 2003). By contrast, coconut<br />
palms, which are planted very frequently in the homegardens worldwide, are rarely<br />
found in Matsiguenka homegardens, except for a few young trees in Tayakome.<br />
Instead, the peach palm is cultivated in 89% of the gardens analyzed. This species is<br />
a domesticated natural hybrid of different native Amazonian palms (Brack Egg,<br />
1999). Another frequently planted tree is Inga edulis, also a domesticated species in<br />
the tropical America. Both species have been reported from the homegardens of<br />
Latin America (Peru: Padoch and de Jong, 1991; Lamont et al., 1999; Colombia:<br />
Eden and Andrade, 1987; Brazil: Smith, 1996; Costa Rica: Zaldivar et al., 2002),<br />
although homegardens elsewhere seldom contain these species (Brazil, 1990). Other<br />
native plants which are most frequently cultivated in Mastiguenka homegardens and<br />
cultivated worldwide at present include cashew and guava, the latter having its<br />
origin in Peru itself (Brack Egg, 1999).<br />
Plant species found in the swidden fields of Matsiguenka are also reported from<br />
other areas of the Peruvian Amazon. For example, Johnson (1983) reported that<br />
cassava, maize, cocoyam (Xanthosoma sp.), pineapple, cotton, sugarcane, papaya<br />
and yam (Dioscorea sp.) are frequently planted crops of young Matsiguenka fields.<br />
In general, the most frequently planted crop that dominates the swiddens is cassava<br />
(Eden and Andrade, 1987; Dufour, 1990).
156<br />
5.3. Changes in species richness<br />
A. WEZEL W AND J. OHL<br />
The longer an area of land is used by the Matsiguenka, the more different will be the<br />
plant species cultivated. For instance, species diversity increased steadily from 18<br />
and 24 species on one-and two-year-old fields to 26, 29, and 66 species in young,<br />
medium-old, and old homegardens, respectively. Some species such as maize,<br />
Calathea allouia, Dioclea virgata, Citrullus lanatus, and tobacco (Nicotiana<br />
tabacum) are only cultivated in swidden fields, and not in the homegardens. In<br />
contrast, many tree species, e.g., Citrus spp., Pouteria caimito, mango and avocado,<br />
are solely observed in the homegardens. The tree species present in fields are<br />
Lonchocarpus sp., peach palm, guava, Inga edulis, and cashew. These species<br />
except Lonchocarpus sp. are the most common ones in homegardens too and they all<br />
are native to Amazonia.<br />
Typical plants reported by the Matsiguenka to be harvested on the old abandoned<br />
fields are peach palm, avocado, Lonchocarpus sp., sugarcane and plantains/bananas.<br />
On such abandoned sites in other parts of Amazon, often described as the agroforest<br />
stage, preference for harvested species is, however, different, except for peach palm<br />
and plantains/bananas. Hiraoka (1986; 1989) described that peach palms and<br />
plantains/bananas as well as Inga edulis, star apple (Chrysophyllum caimito), Brazil<br />
nut (Bertholletia excelsa), and Poraqueiba sericea are still used in the floodplains of<br />
Peruvian Amazon. In the Colombian Amazon, Inga edulis, Theobroma bicolor,<br />
breadfruit, Poraqueiba sericea, Pourouma cercropiifolia, and the West Indian locust<br />
(Hymenaea courbaril) are cultivated (Dufour, 1990).<br />
5.4. The swidden cultivation system in Manu<br />
Before the Matsiguenka settled in communities, they used to move their fields and<br />
huts around in the rainforest area of the Manu. The swidden process typically<br />
involved clearing a patch of rainforest for cultivation (2 to 4 years), building the huts<br />
in the center of the field, and cultivation of mainly cassava in the first two years.<br />
They also used to plant some trees in a very simple form of homegardens around the<br />
huts. Once the field is abandoned, they move to a new field, and build a new hut.<br />
However, every now and then, they return to the old fields to harvest plantains /bananas<br />
or papaya. Some of the homegardens investigated in this study also originated in this<br />
manner. Presently, however, most homegardens are created anew – around scattered<br />
huts of the villages. Although this swidden cultivation system is very similar to the<br />
traditional one, the difference is that the Matsiguenka now cultivate fields within a<br />
certain range of the village – without having to move through the forests. They also<br />
re-establish new fields on fallows within a short period of time. As calculated for<br />
Yomibato, 29% of the field areas cultivated in 2000 or 2001 are located within<br />
formerly cultivated fallow areas (Ohl, 2004). About 28% of these fields were<br />
established on 2 to 10 year fallows, 38% on 10 to 14 year fallows and 34% on 14 to<br />
21 year fallows. However, most fields (71% field area) were created by clearing<br />
primary forests or very old fallows of at least 26 years. On satellite images these<br />
differences could not be clearly seen; but it seems that mainly primary forests have<br />
been cleared for establishing the new fields.
DIVERSITY IN RELATION TO REMOTENESS FROM URBAN U CENTERS C<br />
6. CONCLUSIONS<br />
157<br />
The diversity of species cultivated by the Matsiguenka communities in homegardens<br />
is relatively low as compared to results of other studies reported from the rainforests<br />
of Amazon. Relative isolation from other communities and remoteness from urban<br />
areas seem to be the most important reasons for this low diversity. These findings<br />
are somewhat contrary to the often perceived notion that remoteness from urban<br />
centers increases species richness because subsistence production is based on a<br />
broad variety of species. Furthermore, these communities are still able to extract<br />
several plant products from the surrounding forests, and the impact of this factor on<br />
the observed low species diversity in homegardens was not investigated in this<br />
study.<br />
ACKNOWLEDGMENTS<br />
We thank E. Mannigel, H. Ohl and G. Shepard very much for their corrections and<br />
comments on the manuscript. The fieldwork of this study was financed by the<br />
<strong>Tropical</strong> Ecology Support Program of the GTZ, Germany.<br />
ENDNOTES<br />
1. Works M.A. 1990. Dooryard gardens in Moyobamba, Peru. Focus (Summer<br />
1990): 12 – 17.<br />
2. CTAR (Consejo Transitorio de Administracíon Regional, Madre de Dios),<br />
Parque Nacional del Manu and IMA (Instituto de Manejo de Agua y Medio Ambiente)<br />
1998. Propuesta de ordenamientio territorial de la provincia del Manu -<br />
Resumen ejectivo oficina de proramcíon y planeamiente ambiental – Unidad de<br />
estudios. Cusco, Peru, 231p.<br />
3. CTAR (Consejo Transitorio de Administracíon Regional, Madre de Dios) and<br />
IIAP (Instituto de Investigacíon de la Amazonia Peruana) 2000. Zonificacíon<br />
Ecologica Economica de la Region Madre de Dios, Medio Fisico II, Puerto<br />
Maldorado, Peru, 156p.<br />
4. Instituto Nacional de Estadistica e Informatica 2003. Censos – informacíon<br />
distrital. www.inei.gob.pe. (last accessed: December 2005).<br />
REFERENCES<br />
Baer G. 1984. Die Religion der Matsiguenka - Ost-Peru. Wepf & Co. AG Verlag, Basel,<br />
516p.<br />
Brack Egg A. 1999. Diccionario enciclopédico de plantas útiles del Perú. CBC, Cuzco, 550p.<br />
Brazil M.A. 1990. A list of herbaceous and woody plants grown in home gardens world-wide.<br />
In: Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 214 – 230. United<br />
Nations University Press, Tokyo.<br />
Coomes O.T. and Burt G.J. 1997. Indigenous market-oriented agroforestry: dissecting local<br />
diversity in western Amazonia. Agroforest Syst 37: 27 – 44.<br />
De Clerck F.A.J. and Negreros-Castillo P. 2000. Plant species of traditional Mayan homegardens of<br />
Mexico as analogs for multistrata agroforests. Agroforest Syst 48: 303 – 317.<br />
Dufour D.L.1990. Use of tropical rainforest by native Amazonians. BioScience 40: 652 – 659.
158<br />
A. WEZEL W AND J. OHL<br />
Eden M.J. and Andrade A. 1987. Ecological aspects of swidden cultivation among the<br />
Andoke and Witoto Indians of the Colombian Amazon. Hum Ecol 15: 339 – 359.<br />
Fernandes E.C.M. and Nair P.K.R. 1986. An evaluation of the structure and functions of<br />
tropical homegardens. Agric Syst 21: 279 – 310.<br />
Hiraoka M. 1986. Zonation of Mestizo riverine farming systems in northeast Peru. Natl Geogr<br />
Res 2: 354 – 371.<br />
Hiraoka M. 1989. Agricultural systems of the floodplains of the Peruvian Amazon. In:<br />
Browder J.G. (ed.), Fragile lands of Latin America: Strategies for sustainable<br />
development, pp 75 – 101. Westview Press, Boulder, CO.<br />
Jensen M. 1993. Soil conditions, vegetation structure and biomass of a Javanese homegarden.<br />
Agroforest Syst 24: 171 – 186.<br />
Johnson A. 1983. Machiguenka gardens. In: Hames R.B. and Vickers W.T. (eds), Adaptive<br />
responses of native Amazonians, pp 29 – 63. Academic Press, New York.<br />
Johnson A. 1989. How the Machiguenga manage resources: Conservation or exploitation of<br />
nature. In: Posey D.A. and Balee W. (eds), Resource management in Amazonia:<br />
Indigenous and folk strategies, pp 213 – 223. New York Botanical Society, New York.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Lamont S.R., Eshbaugh W.H. and Greenberg A.M. 1999. Species composition, diversity, and<br />
use of homegardens among three Amazonian villages. Econ Bot 53: 312 – 326.<br />
Méndez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socio-economic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Müller-Dombois D. and Ellenberg H. 1974. Aims and methods of vegetation ecology. John<br />
Wiley & Sons, New York, 547p.<br />
Ohl J. 2004. Die Ökonomie der Matsiguenka im Nationalpark Manu, Peru – Tourismus als<br />
Chance für eine nachhaltige Entwicklung? PhD thesis. University of Greifswald,<br />
Germany (on CD-ROM).<br />
Padoch C. and de Jong W. 1991. The house gardens of Santa Rosa: diversity and variability in<br />
an Amazonian agricultural system. Econ Bot 45: 166 – 175.<br />
Salick J. 1989. Ecological basis of Amuesha agriculture, Peruvian upper Amazon. Adv Econ<br />
Bot 7: 189 – 212.<br />
Shepard G.H.J. and Chicchón A. 2001. Resource use and ecology of the Matsiguenka of the<br />
eastern slopes of the Cordillera de Vilcabamba, Peru. In: Alonso L.E., Alonso A.,<br />
Schulenberg T.S., and Dallmeier F. (eds), Rapid assessment program, pp 163 – 174.<br />
Smithsonian Institution/Monitoring and Assessment of Biodiversity Program, Washington<br />
D.C.<br />
Smith N.J.H. 1996. Home gardens as a springboard for agroforestry development in<br />
Amazonia. Int Tree Crops J 9: 11 – 30.<br />
Thrupp L.A., Hecht S. and Browder J. 1997. The diversity and dynamics of shifting<br />
cultivation: myths, realities, and policy implications. World Resources Institute,<br />
Washington D.C. 49p.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
Wezel A. and Ohl J. 2005. Does remoteness from urban centres influence plant diversity in<br />
homegardens and swidden fields: a case study from the Matsiguenka in the Amazonian<br />
rainforest of Peru. Agroforest Syst 65: 241 – 251.<br />
Zaldivar M.E., Rocha O.J., Castro E. and Barrantes R. 2002. Species diversity of edible plants<br />
grown in homegardens of Chibchan Amerindian from Costa Rica. Hum Ecol 30:<br />
301 – 316.
CHAPTER 10<br />
GENDER AND SOCIAL DYNAMICS IN<br />
SWIDDEN AND HOMEGARDENS IN LATIN<br />
AMERICA<br />
P.L. HOWARD<br />
Department of Social Sciences, Wageningen University, Hollandseweg 1, 6706 KN<br />
Wageningen, the Netherlands; E-mail: <br />
Keywords: Gender relations, Social structures, Women’s status.<br />
Abstract. Structure, composition, and functions of homegardens are said to be closely related<br />
to the social structure of households, but this issue is not often researched. An analysis of the<br />
literature on swidden and homegardens in Latin America shows that such interrelationships<br />
become transparent when examining the gender division of labor, gendered access to garden<br />
resources including land, trees, and other plants, and gendered control over subsistence and<br />
cash crops and income derived from them. Social status related to gardening, gendered<br />
knowledge distribution and transmission, and social dynamics leading to change in gardening<br />
and gardens are also important parameters in this matrix. A review of 39 Latin American case<br />
studies dealing with swidden or homegardens revealed that women are by far the prominent<br />
garden managers across its sub-regions. Aside from the multiple material benefits provided by<br />
gardens, other drivers that tend to ensure that women will strive to maintain them include<br />
their emotional and spiritual values and the positive social status that productive and beautiful<br />
gardens confer. Homegardening is a ‘respectable’ way for women to contribute to subsistence<br />
production and manifest specialized knowledge and skills without competing with men.<br />
However, commercialization may be undermining both women’s control and the benefits they<br />
derive from homegardening as well as the complex structure and function of homegardens.<br />
1. INTRODUCTION<br />
Past research on homegardens shows that the composition, structure, and functions of<br />
gardens are interrelated with their economic, social, and cultural functions (see for<br />
example Wiersum, 2006). However, the social dimensions of homegardens have only<br />
rarely been researched in-depth. Social factors influencing swidden and homegardens<br />
159<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 159–182 .<br />
© 2006 Springer. Printed in the Netherlands.
160<br />
P.L. HOWARD<br />
have not been discussed in any depth in the agroforestry literature in Latin America.<br />
It is particularly by examining gender relations within swidden and homegardens<br />
that the complex interrelationships between social structures and gardens as land use<br />
systems become transparent. Examining gender relationships is also of great<br />
importance since, across most of Latin America, swidden and homegardening are<br />
largely women’s domains, and homegardens may help to mitigate the inequalities<br />
between the sexes that are evident across the region.<br />
This chapter is based on a review of the literature on swidden and homegardens<br />
in Latin America (that which is published in English as well as the little available to<br />
the author in Spanish) that reported sex-disaggregated information. Eight cases were<br />
found that focus on homegardens within Mayan production systems in<br />
Mesoamerica, whereas 12 cases refer to non-Mayan indigenous or mestizo (mixed<br />
Indian-Spanish descent) populations in the same region. In South America, 14 cases<br />
were found that deal with Amazonian Amerindian populations and swidden gardens,<br />
whereas only five cases were found that focus on homegardens among non-<br />
Amerindian South American populations, four of which are also in the Amazon<br />
basin. While swidden gardens and homegardens are distinct land use systems, they<br />
are both agroforestry systems that are rich in species diversity, possessing<br />
“sophisticated spatial structures and dynamics” and manifesting sustained yields<br />
(Michon, 1983). Further, while there has been very little study of homegardens<br />
among Amazonian Amerindians (for reasons for this see Heckler, 2001), there is a<br />
rich literature on swidden gardening.<br />
It must be recognized that a thorough comparative effort would require a<br />
substantially richer bibliographical underpinning. Further, the information provided<br />
in the 39 case studies that are reviewed here is very uneven and hence often difficult<br />
to compare. Thus, in analyzing this literature, the emphasis is on setting out certain<br />
similarities and identifying some of the potential explanatory factors in order to<br />
illustrate the nature and complexity of homegardens as social systems and of gender<br />
relations in swidden and homegardening, and to begin to relate these to the structure,<br />
composition, and functions of these gardens as agroecological systems. Finally, it is<br />
acknowledged that the 39 case studies analyzed herein do not cover the full<br />
spectrum of gardens across the sub-regions, or its ethnic, racial, and indigenous<br />
groups, and therefore the results can only be generalized within the limits of the<br />
study.<br />
2. THE GENDER DIVISION OF LABOR IN GARDENING IN LATIN<br />
AMERICA<br />
The gender division of labor not only provides many insights into how households<br />
organize homegarden production; it also highlights how contributions and<br />
responsibilities of individuals differ according to their positions within the<br />
household, which is very important for understanding the incentives, opportunities,<br />
and constraints that they confront when managing homegardens and how such<br />
individual factors influence homegarden structure, composition and functions. Many<br />
studies across the world seek to analyze the household division of labor in<br />
homegardens by sex and age, principally with the aim of understanding how
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA<br />
161<br />
production is organized. Some studies conceptualize and measure the division of<br />
labor in terms of tasks, where the breakdown may be gross or fine, e.g., land<br />
preparation, planting material procurement, varietal selection, planting, weeding,<br />
water management and irrigation, soil management, pest management, and<br />
harvesting. Others present the division of labor in relation to specific types of crops<br />
(e.g., medicinals, vegetables, spices, trees), to specific species (e.g., coffee [Coffea<br />
spp.], manioc [cassava, Manihot esculenta]), or to specific varieties (e.g., red maize<br />
[Zea mays] used for rituals, versus white maize used for daily consumption). Other<br />
relevant factors include the amount of time required and the timing and intensity of<br />
work, and the relation between homegarden work and individuals’ other labor<br />
obligations and physical mobility. Yet another way it is approached is in terms of<br />
the division of decision-making responsibilities (e.g., for location and design of the<br />
garden, selection and arrangement of species, cultural practices, destination of<br />
output). Divisions of labor are also sometimes discussed in relation to physical<br />
spaces such as zones within gardens or gardens in different locations that are<br />
considered to ‘belong’ to particular persons. Irrespective of how it is measured, the<br />
division of labor is based on cultural associations between sex, age, and kinship<br />
relations (e.g., senior male, wife, daughter-in-law) and obligations that people with<br />
such “social identities” (Boster, 1985a) have to provide particular resources for the<br />
household or for themselves, as well as the differential access to resources (land,<br />
labor, capital, markets, livestock, knowledge, and skills) that is related to these<br />
obligations. These cultural associations are rooted in cosmologies (‘world views’;<br />
understandings of the universe and human beings’ place in it) and related concepts<br />
of what is appropriate behavior for people of different social identities, which at<br />
least for the past generation have been undergoing rapid change nearly across the<br />
region.<br />
In general, homegardening studies in Latin American still do not mention the<br />
gender division of labor, and those that do often present only one of the possible<br />
indicators without specifying why that particular indicator was chosen. Who<br />
provides information about the gender division of labor is also an issue. Lerch<br />
(1999) pointed out that men and women gave very different answers when asked<br />
who was mainly responsible for the homegarden: men said that both were<br />
responsible whereas women said that they themselves were responsible. In fact,<br />
women were most observed working in homegardens. Dufour (1981) also reported<br />
that Tukanoan men in her study site in the Colombian Amazon insisted on<br />
representing their households to outsiders and socially it was not acknowledged that<br />
women are knowledgeable about plants; however, she found that the plant<br />
knowledge of men and women of the same social status did not differ significantly.<br />
Such problems affect much more than only data on the gender division of labor –<br />
getting the informants ‘right’ is necessary to avoid all sorts of research bias, but<br />
particularly bias about women’s work and knowledge around plants (Howard,<br />
2003).<br />
Table 1 presents the reported sex of the ‘main gardener’ (exclusively women,<br />
mainly women, or both men and women together) across the 39 cases, disaggregated<br />
by sub-region. Reporting the sex of the ‘main gardener’ does not mean that the other<br />
sex is not involved – even where it is indicated that “only women” are responsible,
162 P.L. HOWARD<br />
men may occasionally “help out” and, where “mainly women” garden, men often<br />
take on certain tasks or manage certain crops. In those case studies where men were<br />
reported to be the “only” or “main” gardeners, this was in relation to a minority of<br />
the households studied.<br />
2.1. Mayan Mesoamerica<br />
Geographically, Mesoamerica includes the seven countries of Central America as<br />
well as Mexico. Culturally it “joins present day middle and south Mexico, Belize,<br />
Guatemala and parts of Honduras and El Salvador,” much of which was historically<br />
dominated by the Mayan civilization (see Montagnini, 2006). As Montagnini<br />
indicates, homegardens are a complex and much-studied feature of the traditional<br />
land use system among the Mayan people, which have evolved in conjunction with a<br />
particular system of shifting cultivation and bush fallow (milpa) agricultural<br />
production that is organized around the ‘milpa triad’: maize, beans (Phaseolus<br />
coccineus, P. polyanthus, P. vulgaris) and squash (Curcurbita moschata,<br />
C. argyrosperma, C. pepo). In all but one (Murray, 2001) of the Mayan homegarden<br />
studies reviewed, it was found that women were the exclusive or main homegarden<br />
managers, although children and other household members might provide labor. For<br />
Mayan populations, the milpa, which provides the bulk of subsistence staples and<br />
cash crops, is “symbolically the male domain and is the source of male prestige”<br />
(Stavrakis, 1979; Greenberg, 1996; Murray, 2001; Lope Alzina, 2006). Women<br />
often provide ‘additional’ or seasonal labor for milpa production, but it is considered<br />
improper for women to be seen in the milpa without the company of males, a<br />
proscription which is enforced by social sanctions, gossip, and even the threat of<br />
witchcraft (e.g., Murray, 2001). On the other hand, homegardens (solares, huertos,<br />
patxokon na) are perceived as female domains or spaces where a great diversity of<br />
vegetables, condiments, ornamentals, medicinals, and other utilitarian or ritualistic<br />
plants are maintained along with most useful trees, and where women are primary<br />
decision-makers (Benjamin, 2000; Patterson, 2000). When men are involved, this is<br />
either related to particular tasks such as land clearing, tree pruning and thinning,<br />
construction of structures and fences and chopping undesirable growth (Benjamin,<br />
2000; Patterson, 2000), or to specific species or crops. Men also use homegardens as<br />
experimental stations and in situ gene banks for crop diversity - for example, in a<br />
case study site in the Yucatan, Mexico, men use homegardens to test new maize<br />
varieties and preserve traditional varieties that they do not wish to plant in their<br />
fields (Lope Alzina, 2006). Trees and tree crops may be of particular concern to<br />
Mayan men especially when these have commercial value (Patterson, 2000), but<br />
they are usually principally women’s responsibility (Gillespie et al., 2004) because<br />
they fall within the physical space of the homegarden.<br />
In one case, in Belize among the Kekchi Maya, the interrelationship between the<br />
gender division of labor, production spaces, crops, and conceptions about what is<br />
appropriate behavior for women was shown to have an effect on the species<br />
diversity and size of different types of gardens. Women manage homegardens and<br />
‘milpa gardens’. The latter are established after milpa fields are left fallow and are<br />
generally much larger than homegardens (Patterson, 2000). Women maintain
Sex of the<br />
“main<br />
gardener”<br />
Women only i 4 50<br />
i<br />
Table 1. Sex of the “Main Gardener” in the 39 homegardens across Latin America.<br />
Number and percentage of cases, countries, and literature citations forr different sub-region/ethnic groups<br />
Mayan Mesoamerica Non-Mayan Mesoamerica Amazonian Amerindian Non-Amerindian S.A<br />
# % Country (literature # % Country (literature # % Country (literature # % Country<br />
reference)<br />
reference)<br />
reference)<br />
Belize (35),<br />
Guatemala (18),<br />
Mexico (12, 14)<br />
Mainly women 3 37.5 Belize (29),<br />
Mexico (5, 22)<br />
3 25 Belize (28); Mexico (6, 13) 5 35.7 Brazil (30);<br />
Colombia (17, 38);<br />
Peru (1);<br />
Venezuela (16)<br />
3 25 Honduras (9), Mexico (3,<br />
24)<br />
6 42.9 Colombia (10),<br />
Ecuador (8, 36),<br />
Peru (7),<br />
Venezuela (15, 37)<br />
2 3<br />
0<br />
3 7<br />
0<br />
(literature<br />
reference)<br />
Brazil (34);<br />
Ecuador (11)<br />
Brazil (23,<br />
39), Peru (20)<br />
Table 1 (cont.)<br />
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 163
Men and<br />
women<br />
1 12.5 Mexico (26) 6 50 Costa Rica (27); Guatemala<br />
(31); Honduras (21);<br />
Mexico (19); Nicaragua<br />
(25); Panama (33)<br />
3 21.5 Brazil (4), Peru (2,<br />
32).<br />
Total 8 12 14 5<br />
i Men may ‘help’ or not, but their labor is otherwise not systematically involved.<br />
Literature citations and countries of the case studies.<br />
1. Aikman, 1999 (Peru); 2. Alexiades, 1999 (Peru); 3. Angel Perez et al., 2004 (Mexico); 4. Baleé, 1994 (Brazil); 5. Benjamin, 2000 (Mexico); 6.<br />
Blanckeart et al., 2004 (Mexico); 7. Boster, 1985a (Peru); 8. Descola, 1994 (Ecuador); 9. Doxon, 1988 (Honduras); 10. Dufour, 1981 (Colombia); 11.<br />
Finerman and Sackett, 2003 (Ecuador); 12. Gillespie, et al., 2004 (Mexico); 13. Govers, 1997 (Mexico); 14. Greenberg, 1996 (Mexico); 15. Heckler,<br />
2004 (Venezuela); 16. Hoffmann, 1993 (Venezuela); 17. Irvine, 1987 (Colombia); 18. Keys, 1999 (Guatemala); 19. Lazos Chavero and Alvarez Buylla,<br />
1988 (Mexico); 20. Lerch, 1999 (Peru); 21. Lok (endnote 1; Honduras); 22. Lope Alzina, 2006 (Mexico); 23. Madaleno, 2000 (Brazil); 24. Martin, 1996<br />
(Mexico); 25. Mendez et al., 2001 (Nicaragua); 26. Murray, 2001 (Mexico); 27. Ochoa et al. (endnote 2; Costa Rica); 28. Palacio, 1980 (Belize); 29.<br />
Patterson, 2000 (Belize); 30. Posey, 1984 (Brazil); 31. Ruonavaara, 1996 (Guatemala); 32. Salick, 1997 (Peru); 33. Samaniego and Lok (endnote 3;<br />
Panama); 34. Sereni Murrieta and Winklerprins, 2003 (Brazil); 35. Stavrakis, 1979 (Belize); 36. Uzendoski, 2004 (Ecuador); 37.Veth and Reinders<br />
(endnote 4; Venezuela); 38. Wilson, 1997 (Colombia); 39. Winklerprins, 2002 (Brazil).<br />
0 0<br />
164 P.L. HOWARD
milpa gardens because of constraints in terms of soil quality, animal predation, and<br />
lack of space in homegardens. It was found that women who cultivate a large<br />
number of edible crops and herbs in their homegardens are those whose husbands<br />
are frequently away for considerable periods, during which time women rarely if<br />
ever travel to milpa gardens. Women whose husbands do not leave the village<br />
cultivate larger milpa gardens and maintain fewer species in their homegardens. In<br />
another case, in a Mayan community in the Yucatan (Lope Alzina, 2006) where<br />
communal land has become available within city limits, people use it as a second,<br />
non-traditional gardening space. Men and women share labor and decision-making<br />
to a much greater extent and women are allowed to work in these gardens<br />
unaccompanied by men, even though these gardens are organized as a kind of<br />
“miniature milpa,” containing the traditional milpa staple crop triad. Thus, gendered<br />
norms appear to be more flexible when people work outside of the traditional<br />
production system, and such flexibility also affects the structure, composition, and<br />
functions of homegardens.<br />
2.2. Non-Mayan Mesoamerica<br />
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 165<br />
In the division of labor among non-Mayan Mesoamerican populations, men are<br />
typically responsible for field crop and cattle production and women for<br />
homegardening and small livestock (usually pigs and chickens). The staple crops<br />
produced across much of Mesoamerica are similar to those produced in the Mayan<br />
milpa: especially maize and beans are prominent. The exception is presented by the<br />
one case study on the Garifuna (Palacio, 1980) which is a Black Carib population in<br />
which women are the main crop producers. Eleven of the studies presented<br />
information on the “main gardeners” – in six of these cases, women are reported to<br />
be exclusive or principal gardeners, whereas in the other five cases homegardens are<br />
managed by both sexes. The main differences with the Mayan division of labor in<br />
terms of men’s participation appears to be that men are involved in homegardening<br />
principally in relation to crops with high commercial value, especially tree crops<br />
[e.g., citrus (Citrus spp.) and coffee], and there are more cases where men use<br />
homegardens to test exotic crops that they wish to introduce into agricultural<br />
production (Angel Peréz and Mendoza, 2004; endnotes 2 and 3). <strong>Homegardens</strong> may<br />
also be considered in general as women’s spaces in non-Mayan Mesoamerica and<br />
the restrictions on women’s work in milpas appear to be strict (Govers, 1997;<br />
Roquas, 2002).<br />
2.3. Amazonian Amerindians and swidden gardening<br />
Nearly all Amazonian Amerindian societies have traditionally depended for their<br />
livelihoods on a combination of hunting, fishing, gathering, and gardening activities,<br />
where men hunt and fish and women are responsible for gardening, although these<br />
relations are changing mainly due to commercialization (Knauft, 1997; Heckler,<br />
2004). Amazonian Amerindians often have highly complex land use systems that<br />
combine multiple types of swidden gardens (including fallow field gardening) and
166 P.L. HOWARD<br />
homegardening to provision themselves with starchy staples, particularly manioc<br />
(both bitter and sweet varieties), which are complemented especially by plantain and<br />
banana (Musa spp.), yam and sweet potato (Dioscorea spp.), taro (Colocasia<br />
esculenta), vegetables, fruits and medicinals. The complexity of their agroforestry<br />
systems and social organization has made them the subject of much in-depth<br />
research.<br />
In all but three of the 14 case studies on Amazonian swidden gardens that were<br />
reviewed, women were the exclusive or principal gardeners. In one case, among the<br />
Ese Eja in Peru (Alexiades, 1999), there is also an age division of labor that gives a<br />
more prominent role to older men in gardening than in other Amazonian Amerindian<br />
cases, mainly due to the fact that certain cultivars are associated with malevolent<br />
spirits that may harm fetuses and infants, which effectively prohibits women of<br />
childbearing age from cultivating or consuming them. In another case, among the<br />
Ka’apor in the Eastern Amazon of Brazil (Baleé, 1994), men invest a slightly greater<br />
amount of time in swidden gardening than women, and neither sex invests much<br />
time in homegardening. However, men’s involvement in swidden gardening over<br />
much of Amerindian Amazonia is often restricted mainly to clearing undergrowth<br />
and felling trees for new gardens, whereas all other tasks are left to women (Posey,<br />
1984; Hoffman, 1993; Descola, 1994). In some cases it is reported that men assist in<br />
garden maintenance (Hoffman, 1993; Uzendoski, 2004), particularly among<br />
Guyanese groups where the gender division of labor is less rigid – for example,<br />
among the Piaroa of Venezuela, men are reported to help in weeding, harvesting and<br />
carrying crops from swidden gardens (Heckler, 2004).<br />
The gender division of labor is not only reflected in tasks associated with<br />
gardening – it is also often strongly related to crops as well as to physical spaces,<br />
associations which are embedded within cosmology and concepts of masculinity and<br />
femininity that are in turn related to prestige and to complementarities and conflicts<br />
between the sexes. Manioc is by far the most important crop across Amazonian<br />
Amerindian cultures, and it is strongly culturally associated with women – in only<br />
two cases (Baleé, 1994; Salick, 1997) was it found that men had a substantial role in<br />
manioc cultivation and in one of these (Salick, 1997) it was reported that this<br />
probably represented a deviation from the traditional division of labor due to labor<br />
shortages. Manioc and manioc beer figure importantly not only in the diet, but as<br />
well in ritual and exchange. The highly complex cosmology associated with women,<br />
manioc, and gardening is discussed in relation to the Achuar (Descola, 1994) and the<br />
Warua 4 .<br />
Apart from tubers, other crop-sex associations are also quite evident. Among the<br />
Ka‘apor (Baleé, 1994), both men and women plant manioc, but women are<br />
exclusively responsible for planting cotton (Gossypium spp.), Indian shot (Canna<br />
indica), job’s tears (Coix lacryma jobi) and pipiriwa (Cyperus corymbosus), which<br />
are used only by women for textiles or for body ornamentation. Only men plant<br />
maize. Among the Piaroa, it is also men who plant maize, and they exclusively plant<br />
tobacco (Nicotiana tabacum) (Heckler, 2004). In fact, Amazonian Amerindian men<br />
are often strongly linked to particular species and have exclusive power to manage<br />
these species – among the Achuar, only men may plant botanical fish poisons since<br />
“if these were to be handled by women, they would lose their effectiveness”
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 167<br />
(Descola, 1994). Achuar men are also predominantly associated with hallucinogens,<br />
tobacco, maize, and bananas, which are only planted outside the main swidden<br />
garden around the edges of the house yard. This reveals yet another aspect of the<br />
gender division of labor. Women’s swidden gardens are out-of-bounds to men since<br />
they are “the only absolutely female space in the Achuar social topography, the only<br />
place where women truly exercise a material and symbolic hegemony” (Descola,<br />
1994). Uzendoski (2004) also noted the very strong association between physical<br />
production spaces and gender among the Napo Runa of the Ecuadorian Amazon,<br />
who see the forest (sacha) as masculine while gardens (chagra) are seen as mainly<br />
feminine.<br />
2.4. Non-Amerindian South America<br />
Few case studies were found that focus on homegardening in non-Amerindian South<br />
American societies and that discuss the gender division of labor. Four out of the five<br />
studies reviewed deal with the Amazonian region but not with Amerindian<br />
populations, whereas only one was found that relates to the Andes (Finerman and<br />
Sackett, 2003).<br />
All five cases reported that women are the exclusive or principal gardeners in the<br />
majority of the households that were investigated. However, in three of these cases,<br />
a number of households were found where men were main gardeners, although those<br />
households were in the minority. In the case of three villages in rural Amazonian<br />
Peru (Lerch, 1999), in those households where men were the main gardeners, it was<br />
clear that women were also involved in the work. In the two urban cases in Para<br />
State, Brazil, 70% of the urban growers in Belém were women, whereas in<br />
Santarém, 67% of the homegardens were maintained by women (Madaleno, 2000;<br />
Winklerprins, 2002). On Ituqi Island, also in Para State, among a Caboclo<br />
population (mixed Brazilian Amerindian and European or African ancestry),<br />
homegardens are said to be the “unquestionable domain of women” (Sereni Murrieta<br />
and Winklerprins, 2003). In the Ecuadorian Andes, Finerman and Sackett (2003)<br />
also found that women are unquestionably the heads of gardens. These gardens are<br />
“medicine cabinets” (one contained 194 species of which 132 were medicinals and<br />
on average nearly 70% of species in homegardens were used for medicine), where<br />
lay medicine is clearly defined as a female domain.<br />
The case studies reviewed above demonstrate that the gender division of labor<br />
can be viewed in multiple ways – as a division of tasks or responsibilities, of crops,<br />
or of resources or physical domains, and typically as a combination of these.<br />
Regardless of how it is viewed, it is related to culturally established norms of<br />
behavior that often have their roots in cosmology and that clearly differ according to<br />
ethnicity and tribal affiliation. What is considered appropriate behavior for women<br />
differs strongly between Amerindian and non-Amerindian populations insofar as<br />
women have primary responsibility for staple crop production in many Amerindian<br />
societies. Yet they are excluded from such production over much of Latin America,<br />
where instead they are responsible for a myriad of so-called ‘minor’ crops,<br />
particularly those with cultural, culinary, and medicinal values that are typically<br />
produced in homegardens. In both cases, the responsibility for swidden and
168 P.L. HOWARD<br />
homegardens, and ipso facto for the skills, resources, knowledge, and biological<br />
diversity that are entailed, falls mainly to women.<br />
3. GENDER AND ACCESS TO GARDEN RESOURCES<br />
From the foregoing discussion, it appears that the gender division of labor is closely<br />
related to men and women’s differential access to homegarden resources, especially<br />
land, trees, and other plants. Terms of access and rights to these resources are more<br />
complex and significant than most case studies suggest and have significant<br />
consequences for garden structure, composition and functions, for the investments<br />
made and benefits derived from gardening, and for the distribution of such benefits<br />
between households and among household members.<br />
As is the case with property rights of all types, rights to swidden and<br />
homegarden resources are also differentiated by sex. Variations in homegarden<br />
resource access according to sex that are found in the case studies are summarized in<br />
Table 2. This table includes only a subset of the total number of case studies<br />
reviewed since the majority did not provide sufficient sex-disaggregated information<br />
about resource access.<br />
Table 2. Patterns of gendered access to garden resources in the 39 case studies across Latin<br />
America.<br />
Agricultural<br />
land<br />
owner-<br />
ship ii<br />
Agricultural<br />
land<br />
usufruct<br />
Garden<br />
land<br />
ownership<br />
ii<br />
Garden<br />
land<br />
usufruct<br />
Garden<br />
tree<br />
ownership<br />
iii<br />
i<br />
Garden<br />
plant<br />
ownership<br />
iii<br />
Countries (literature<br />
references: see<br />
Table 1 for author<br />
details)<br />
MF M MF F F F Ecuador (11)<br />
M M M MF MF MF Belize (29)<br />
Mexico (5, 12, 22)<br />
M M M MF M F Costa Rica (27)<br />
Mexico (3, 19)<br />
Panama (21, 33)<br />
- - M MF MF MF Ecuador (8)<br />
Note: signifies physically separate; M = male; F = female.<br />
i Includes only cases where gendered resource access and control were discussed.<br />
ii Where there is no private ownership, this means customary control over land allocation.<br />
iii May be explicit, or may be inferred from the case studies.<br />
Although homegardens in Latin America are nearly by definition small<br />
(generally considerably less than 1 ha and at times only a dozen square meters), land<br />
is obviously still a crucial production factor, and homegarden land can be even more<br />
productive on a per hectare basis than agricultural land 3 . Irrespective of the sex of<br />
the main gardener, it is reported that poorer households have greater difficulty<br />
obtaining access to land for homegardening and greater difficulties meeting
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 169<br />
household needs from gardens when they do have access. For these people, lack of<br />
tenure security represents the greatest threat since many occupy land illegally,<br />
especially in urban or urbanizing areas (Madaleno, 2000; Finerman and Sackett,<br />
2003). Lower land access is reported to prevent cultivation of species that require<br />
substantial amounts of space (Doxon, 1988). As well, tree planting and security of<br />
land tenure are often interrelated since tree planting often creates rights to land or,<br />
conversely, only landowners may plant trees (Bruce and Fortmann, 1988). Thus, tree<br />
and land tenure may affect composition and structure through the number and type<br />
of trees planted in homegardens and through garden size.<br />
It is normally presumed that whoever owns or formally controls homegarden<br />
land will control homegarden production, but this is certainly not so in the case<br />
studies reviewed here. Deere and Leon (2001) show that male land ownership and<br />
control predominate over most of Mesoamerica and non-Amerindian South<br />
America, which is confirmed by the homegarden studies that report such<br />
information: in only one case was it said that men and women jointly own land<br />
(Finerman and Sackett, 2003). Generally, it is men who have the ultimate right to<br />
dispose of land, although decision-making may be joint as Patterson (2000) showed<br />
for Mayan homegardens in Belize. In all homegardening cases, women appear to<br />
obtain informal usufruct rights to homegarden land from their husbands. Among<br />
populations where there is no clear concept of land ownership, such as among many<br />
swidden horticulturalists, ‘spaces’ are frequently gendered and gardens may be<br />
considered strongly or weakly as ‘women’s spaces’. Generally, men formally<br />
control swidden garden land and allocate it to women (Descola, 1994; Goldman,<br />
1963; Posey, 1984). However, neither of these phenomenon is pervasive – for<br />
example, “The Cubeo always speaks of a particular manioc plot as belonging to a<br />
woman, the only instance of individual possession of land in Cubeo society”<br />
(Thompson, 1977).<br />
Only two studies were found that elucidate why or how it is that women gain<br />
which type of usufruct rights to homegarden land that men control (Descola, 1994;<br />
Lope Alzina, 2006), and these discuss how such rights are influenced by<br />
negotiations between men and women. Among the polygamous Achuar of<br />
Amazonian Ecuador (Descola, 1994), each co-wife must cultivate her own garden<br />
plot. Men divide plots that they have cleared and assign them to each co-wife by<br />
planting rows of banana. The size of the garden is negotiated: while both men and<br />
women wish to have large gardens, women consider their access to labor for<br />
weeding and negotiate with their husbands considering this constraint. It is also<br />
notable that once each co-wife’s patch “has been materially marked out under male<br />
authority, the garden finally becomes the closed area of a purely female praxis.”<br />
Still, it is clear that women have strong obligations to produce manioc beer for the<br />
men who provide them with land.<br />
Access to homegarden land must be very important for those Latin American<br />
women who lack access to other land to cultivate, particularly when women are able<br />
to use this land in a way that is highly productive and to make most decisions<br />
regarding its use and management. The fact that women obtain land through their<br />
husbands implies that divorce or separation may deprive them of access altogether,
170 P.L. HOWARD<br />
and men can also ultimately decide to use such land in other ways, which is<br />
discussed further below.<br />
Land is not the only resource that is crucial to homegardening. Agrobiodiversity<br />
is also a major resource and rights to trees and other plants cannot simply be<br />
assumed to pertain to those who control the land. What appears to be most common<br />
in the cases reviewed is that men may grow specific homegarden crops of their own,<br />
but most homegarden species belong to women. Within this, a relatively consistent<br />
pattern is discernable in relation to trees, since trees are often related to male<br />
ownership (Bruce and Fortmann, 1988) and tree tenure also differs from “plant<br />
tenure” (Howard and Nabanoga, 2006). In the Mayan cases, tree ownership does not<br />
appear to be clearly related to either men or women and women generally plant and<br />
manage trees in homegardens (Benjamin, 2000; Gillespie et al., 2004). Nevertheless,<br />
in the Mayan case that reported that trees provide cash crops, men also participate in<br />
their management and use (Patterson, 2000). In many non-Mayan Mesoamerican<br />
communities, however, it appears that men make decisions about trees and control<br />
income from them, particularly when they have high commercial value (Lazos<br />
Chavero and Alvarez Buylla, 1988; endnotes 2 and 3).<br />
One case provides insights into the relationship between land tenure, tree tenure,<br />
and cosmology. Samaniego and Lok 3 report that, among the Ngöbe in Panama,<br />
women attribute greatest importance to homegardening while men value tree<br />
(especially coffee) production most highly. Land is generally communally owned,<br />
but the person that plants a tree becomes the owner of the land upon which the tree<br />
is planted. The head of a household will bury the placenta pertaining to every<br />
newborn and plant a tree on that site, and only the head knows the tree species and<br />
the site. The well-being of the tree and of the person whose birth the tree marked are<br />
directly interrelated, so household heads should always be consulted regarding the<br />
management of trees in homegardens.<br />
Aside from trees, women may have exclusive rights to plants growing in<br />
homegardens and their husbands or other household members may have no right to<br />
harvest or otherwise destroy these plants without their permission. Finerman and<br />
Sackett (2003) reported that, in their study village in the Ecuadorian Andes, men and<br />
women jointly own land and animals. Men make most decisions concerning<br />
farmland and cattle, while households and homegardens are women’s domains, both<br />
in terms of management and of rights to the plants growing therein. Anyone wishing<br />
to have access to a plant in a woman’s garden must ask her permission. Dufour<br />
(1981) observed that, among the Tukanoan Indians of the Colombian Northwest<br />
Amazon, women often plant manioc in a section of another woman’s garden. This<br />
highlights the fact that there are myriad social relations and subtle social norms<br />
about property in land and in plants that are related to factors other than<br />
“ownership”: other homegarden research shows that even a person who “owns” or<br />
manages trees or plants in a homegarden might not have exclusive rights to them<br />
(Howard and Nabanoga, 2006).<br />
Lok 1 provided an example that demonstrates how rights to plants and trees may<br />
be circumscribed depending upon the species and upon who controls the zone in<br />
which they are planted, as well as how gendered rights to homegarden resources<br />
affect their structure and composition. She researched homegardens in a Mestizo
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 171<br />
(mixed Spanish-Indian) community in north central Honduras and found 253 useful<br />
plant species in a sample of 10 gardens, with an average 60 species per garden<br />
distributed in nine management zones that could be discriminated by examining<br />
vertical strata, geophysical characteristics and “gender access or responsibility.”<br />
Women have responsibility for particular zones, such as the residential zone where<br />
ornamentals, vegetables, and medicinal plants are produced, as well as tree and plant<br />
nurseries. Men are in charge of the coffee zone, which provides much shade for<br />
vegetables and medicinal plants that women plant within it, but men simply<br />
‘tolerate’ these plants and eliminate them without their wives’ permission if they see<br />
fit in order to plant more coffee. Lok 1 concluded that the study of management zones<br />
“makes it possible to relate agroecological variables to social and economic ones,<br />
which is of great importance in homegarden analysis.” It is clear that one factor in<br />
this analysis is gendered rights to homegarden trees, plants and zones.<br />
4. GENDER AND COMMODITY PRODUCTION IN HOMEGARDENING<br />
Both the gender division of labor in homegardening and gendered rights to<br />
homegarden resources appear to be related to the control over income generated<br />
through cash crops. In the author’s experience with rural homegardens in Honduras,<br />
Nicaragua and El Salvador from 1982 – 1990, women use such income to pay for<br />
school fees, pharmaceuticals and medical services, and as “pocket money” for<br />
making daily purchases of food and other goods to meet household needs.<br />
Homegarden produce is available in small quantities year-round, so it is unsurprising<br />
that women are responsible both for such small daily purchases and for the<br />
production that provides the income for these purchases. The cases reviewed suggest<br />
that, when women market homegarden produce, they do so nearly exclusively in<br />
local markets, and the amount of income generated is generally quite small in<br />
relation to total household income. However, the amount generated can certainly be<br />
more substantial, as several homegarden studies across the globe attest. Finerman<br />
and Sackett (2003) reported that, in the Andes of Ecuador, women sell sufficient<br />
surplus from their homegardens to contribute to household income and improve<br />
their own status.<br />
Table 3 presents an overview of the gender division of labor in relation to the<br />
production of subsistence crops, of crops that are marketed on a small-scale, and of<br />
high-value crops or crops marketed on a larger scale for those case studies that<br />
provided information (15 of the 39 reviewed). It shows that women are more likely<br />
to manage crops destined principally for subsistence (in 80% of the cases) or for sale<br />
in small quantities in local markets (in 88%). As cash cropping occurs on a larger<br />
scale or high value crops are produced, men’s involvement and control are much<br />
more evident (86%).<br />
The associations between women and subsistence production are quite strong, as<br />
are the associations between women and medicinals, spices, condiments, and<br />
ornamentals. A typical example is found in Angel Peréz and Mendoza (2004) in<br />
relation to a traditional Totonac community in Veracruz, Mexico. They reported that<br />
women manage culturally important plants (for subsistence, ritualistic, and<br />
medicinal purposes) and are responsible for backyard gardens and orchards, whereas
172 P.L. HOWARD<br />
men use homegardens to test and adapt exotic plants that they later introduce into<br />
commercial field crop production. Patterson (2000) found a similar pattern among<br />
the Kekchi Maya in Belize where homegarden cash crops have recently increased in<br />
number and are mainly introduced by male heads.<br />
Table 3. Responsibility for subsistence and cash crop production in homegardens by sex i in<br />
15 case studies across Latin America.<br />
Subsistence Small<br />
scale<br />
marketing<br />
M = male; F = female.<br />
Major or<br />
high value<br />
cash crops<br />
Countries (literature references: see<br />
Table 1 for author details)<br />
F - M Belize (29), Honduras (21), Mexico (3 ii ,<br />
26), Panama (33)<br />
MF M Mexico (Alvarez Buylla et al., 1989) iii<br />
F F -<br />
Guatemala (18), Ecuador (11), Mexico<br />
(6, 13, 14, 22), Venezuela (16)<br />
MF - M Costa Rica 27)<br />
MF - MF Nicaragua (25)<br />
12 80.0% 7 87.5% 0 0.0% Total cases and percent of women in<br />
them<br />
3 20.0% 0 0.0% 1 14.3%<br />
Total cases and percent of both men and<br />
women<br />
0 0.0% 1 12.5% 6 85.7% Total cases and percent of men<br />
15 8 7 Total cases<br />
i<br />
Includes only cases where some garden produce is reported to be sold.<br />
ii<br />
Men experiment with cash crops destined eventually for agricultural fields.<br />
iii<br />
Implicit. Men manage fruit trees, and only citrus is sold in small quantities.<br />
Given the strong influence of women’s decision-making in homegardening<br />
across most of the cases in Latin America, it is interesting to examine whether<br />
commodity production plays a role in the gender division of labor in homegardens<br />
that are managed by both men and women. Table 4 presents the cross-tabulation of<br />
the 10 cases where data on the sex of the main gardener and the production of major<br />
market crops were both reported. In five of the six cases where both men and<br />
women share responsibility for homegardening, men produce major or high value<br />
cash crops in homegardens and, in one case, both men and women produce them. In<br />
only one case was it reported that, while men and women are main gardeners, no<br />
major or high value cash crops are produced; in another two cases, women are the<br />
main homegardeners but men manage high value crops. It is important to note that<br />
all of these cases refer to the Mesoamerican context: the South American cases<br />
presented insufficient data.<br />
A few studies have discussed what occurs in terms of shifting responsibilities<br />
for, and benefits from, homegardening when it begins to generate substantial<br />
amounts of cash income or cash crops are introduced, even in contexts where<br />
homegardening is culturally strongly associated with women. Murray (2001)
eported that, among the highland Maya of Chiapas, Mexico, people are quite<br />
dependent on the cash economy. Men emigrate and secure paid jobs, and much cash<br />
crop production occurs in homegardens where chemical inputs are also used.<br />
Commercial flower production is one of the activities that men have integrated into<br />
traditional homegardens. The strong integration into the market economy has<br />
undermined women’s economic and decision-making power in these households and<br />
gardens. As homegarden production becomes more lucrative or more marketoriented,<br />
women’s roles in them as managers, sellers, and earners of cash income<br />
appear to shift. Other studies report that commercialization may have negative<br />
effects with respect to agrobiodiversity and household food security. For example,<br />
Baleé (1994) reported that, in the eastern Amazon of Brazil, agricultural<br />
extensionists encouraged the production of rice as a cash crop. The result was that<br />
the space for traditional crops such as foods, spices, and other utilitarian plants was<br />
reduced to the point that these crops are no longer found in swiddens in these<br />
villages, which would obviously have a substantial impact on women.<br />
Table 4. Cross-tabulation of sex of the main gardener and responsibility for major or high<br />
i<br />
value cash crops in 10 case studies across Latin America .<br />
Sex of main<br />
gardener<br />
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 173<br />
Subsistence Major or high<br />
value cash<br />
crops<br />
Countries (literature references:<br />
see Table 1 for author details)<br />
Men and women Men and<br />
women<br />
- Mexico (19)<br />
Women Women Men Belize (29), Mexico (3) ii<br />
Men and women Men Costa Rica (27), Honduras (21),<br />
Mexico (26), Panama (33)<br />
Men and women Men and<br />
women<br />
Nicaragua (25)<br />
i<br />
Ruonavaara (1996 – Guatemala) reported that both women and men manage homegardens<br />
and also reported small-scale marketing, but did not report who was responsible.<br />
ii<br />
Men only experiment with cash crops destined eventually for agricultural fields.<br />
Commercialization may leave women’s gardening responsibilities intact but may<br />
create other shifts that affect the composition and structure of homegardens and<br />
therefore agrobiodiversity and dietary composition. In Ecuador, dependency on<br />
global markets caused an economic crisis when the nation’s economy collapsed in<br />
the late 1990s. Residents of the village that Finerman and Sackett (2003) studied in<br />
the Andes have lost property and been forced to emigrate, so that “increasing<br />
number of homegardens lie abandoned by absentee landowners, or are plowed under<br />
to make way for cash crops that have done little, thus far, to ease the financial<br />
burdens of the owners.”<br />
Women are not necessarily marginalized when homegardens increase in<br />
economic importance; rather, both women and homegardens may provide a buffer<br />
against the worst effects of economic or environmental crisis affecting men’s<br />
agricultural production. Greenberg (1996) reported that, due to decreasing viability
174 P.L. HOWARD<br />
of men’s agricultural production in rural areas of the Yucatan Peninsula, families<br />
have migrated to the tourist resort of Quintana Roo in search of wage labor. Men no<br />
longer engage in agricultural production, but women maintain traditional homegardens<br />
and agrobiodiversity in this urban setting, and homegardens generate cash<br />
for these families in many ways. Still, there may be other negative implications of<br />
shifting gender roles: this change in gender domains may partly account for social<br />
problems and men’s excessive drinking.<br />
Yet other trade-offs for women and their households must be considered, since<br />
production for subsistence and for cash income generation are certainly not the only<br />
measures of the value that homegardens provide. As many authors point out,<br />
homegardens are often sources of non-monetary exchange values through giftgiving<br />
and reciprocal exchange. These are very important especially to women<br />
gardeners as sources both of material goods and of status and social autonomy.<br />
5. SOCIAL STATUS, GENDER, AND GARDENS<br />
Much of the research that has been done on homegardens has emphasized the<br />
economic and ecological functions and benefits of homegardening and has stressed<br />
these as principal reasons for their creation and maintenance, without examining in<br />
any depth other ways in which homegardens provide social or material advantages<br />
for their owners. Even so, several of the articles reviewed acknowledge the social<br />
status that is associated with homegardening, especially with having a particularly<br />
large, beautiful or genetically diverse garden. As Sereni Murrieta and Winklerprins<br />
(2003) noted, a homegarden “says much about its keeper.”<br />
The same also appears to be true of swidden gardening. Descola’s (1994) work<br />
highlighted how researchers often mistakenly assume that the diversity that swidden<br />
gardeners create or maintain is due to ecological or economic motivations rather<br />
than to status-seeking behavior. At the same time, he showed how gardening may<br />
increase women’s status in the eyes of men. For the Achuar, it is a “point of honor”<br />
for women to cultivate large swidden gardens. The garden diversity evident,<br />
particularly in tubers, cannot be attributed to nutritional or culinary needs since<br />
“men - whose attitude openly encourages their wives’ agronomic capacities -<br />
recognize by taste alone only a very low proportion of the varieties of manioc, yams,<br />
or sweet potatoes.” Nor can it be attributed to the need to reduce species-specific<br />
diseases since only one serious manioc disease is recognized, and only a few plants<br />
are usually affected. Rather, “a woman who successfully grows a rich pallet of<br />
plants thereby demonstrates her competence as a gardener and fully assumes the<br />
main social role ascribed to women by proving her agronomic virtuosity” (Descola,<br />
1994).<br />
Finerman and Sackett (2003) found in the Ecuadorian Andes that people observe<br />
each other’s homegardens and deduce information about the owners’ wealth status,<br />
occupation and market orientation, as well as health status. The abundance and<br />
diversity of a garden is an important source of status for women who develop<br />
reputations as skilled gardeners whom people continually approach for planting<br />
materials, for advice and to exchange produce. Women boast about their<br />
homegardens and about the independence these afford. Yet the implications of
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 175<br />
homegardening for women’s status are not only related to their production<br />
capacities; they are also clearly linked to the roles women are expected to perform as<br />
family caretakers and as representatives of their households. <strong>Homegardens</strong> reveal:<br />
the extent of the owner’s commitment to family well-being . . The presence of a garden<br />
rich in . . [medicinal plants] epitomizes her exertions on behalf of kin, and her<br />
proficiency as primary health provider; a spacious and productive garden filled with<br />
medicinal plants suggests that the family, too, is prosperous and fit . . Gardens<br />
themselves [are] a manifestation of the community’s most deeply held values:<br />
autonomy, status, religious piety, and personal investment in family. . A garden<br />
demonstrates a woman’s freedom from dependence on products from neighbors and<br />
commercial vendors; her fiscal standing evidenced by her ability to expend valuable<br />
land on a garden; her faith displayed by a sacrifice of resources to adorn the church; and<br />
her industriousness and devotion to family exhibited by her investment in plant<br />
cultivation (Finerman and Sackett, 2003).<br />
It is clear that the status provided through gardening is not confined to gardens’<br />
visible characteristics or the skills of their owners. Many studies show that garden<br />
produce that is not consumed is much more commonly given as gifts or exchanged<br />
with others rather than sold in markets, and most homegarden studies also report that<br />
the vast majority of garden planting materials that are not self-provisioned are<br />
acquired through gifts and exchange (Blanckaert et al., 2004), predominantly<br />
between women (Boster, 1985b; Alvarez Buylla et al., 1989; Hoffman, 1993;<br />
Greenberg, 1996; Lerch, 1999; Patterson, 2000; Ruonavaara, 1996; Finerman and<br />
Sackett, 2003; Sereni Murrieta and Winklerprins, 2003). Such exchanges are not<br />
only important in terms of the garden products or planting materials that gardeners<br />
access – they are just as important as a means to create and maintain social<br />
networks. Gift giving and exchange of planting materials often help Mayan women<br />
maintain kinship and neighborly ties with people in distant places (Greenberg, 1996)<br />
and provide additional opportunities to accumulate knowledge (Patterson, 2000).<br />
Likewise, Finerman and Sackett (2003) referred to women’s plant “borrowing” in<br />
the Andes as an important basis for household exchanges, which are most common<br />
among female relatives and close friends. Lerch (1999) researched homegarden<br />
plant diversity and exchange in the Amazon where networks for exchange of<br />
indigenous planting material have been strong historically. In the villages she<br />
studied, reciprocal exchange among neighbors (who might also be kin) was the most<br />
important source of plant material acquisition, and households with high plant<br />
diversity exchanged plants at a higher rate.<br />
Among Amazonia Amerindians, male prestige is often related to ceremonial<br />
exchange of food products such as manioc beer (Descola, 1994; Heckler, 2004;<br />
Thompson, 1977). Women may also gain prestige as producers of the crops that men<br />
exchange as occurs among the Cubeo (Goldman, 1963), the Achuar (Descola, 1994)<br />
and the Piaroa (Heckler, 2004). Among Piaroa groups, women manioc cultivators<br />
can assert themselves as agronomic experts, which is evident in the great diversity of<br />
manioc cultivars they produce. They create alliances by exchanging this diversity, as<br />
well as by processing manioc in “processing parties” which are events of<br />
“communality and congeniality” in which women gain prestige as hard workers and<br />
food providers (Heckler, 2004).
176 P.L. HOWARD<br />
To the degree that women’s status is positively affected by their homegardening<br />
activities, their status may erode as homegardening itself declines. Stavrakis (1979)<br />
noted in the villages that she studied in Belize how kitchen gardens lost prestige as<br />
people began to reject local fruits and vegetables in favor of imported varieties, and<br />
gardens became obsolete. “As the garden loses its social value, so naturally do<br />
women’s gardening activities.” Aikman (1999) and Hoffman (1993) argue that<br />
women’s traditional knowledge and management of local crop diversity that they<br />
maintain in home or swidden gardens may become valueless, and their high social<br />
status turn to social stigma, as such knowledge and production become increasingly<br />
associated with poverty and backwardness.<br />
6. KNOWLEDGE AND GENDER IN HOMEGARDENING<br />
The status derived from gardening is in part due to the knowledge and skills that are<br />
necessary to create and maintain them. Depending on the degree to which gardening<br />
knowledge is specialized, it will be unevenly distributed and this distribution will<br />
reflect factors such as age, sex roles, and differential ‘opportunities to learn’ (Boster,<br />
1985a). That gardening knowledge is specialized is widely reported in the literature<br />
reviewed. To the degree that the species diversity in homegardening is greater than<br />
in agricultural fields, this implies greater breadth of ethnobotanical and agronomic<br />
knowledge than what is common in agricultural production. Further, because so<br />
many species and varieties are intercropped in homegardens, knowledge of plant<br />
associations is also likely to be greater. Such associations are also very likely to be<br />
related to microclimates that are created within homegardens and that do not exist<br />
elsewhere, which “enables: (i) the growing of varieties with different climatic<br />
requirements . . . (ii) the elaboration of a management calendar independent of the<br />
climatic functions, and (iii) the experimentation with new varieties” (Alvarez Buylla<br />
et al., 1989; endnote 1).<br />
When Benjamin (2000) examined Mayan cultural homegarden practices in<br />
depth, she found that women homegardeners’ knowledge is based on “principles”<br />
that maximize micro-environmental conditions for successful plant propagation,<br />
which are passed on across generations. Similarly, Gillespie et al. (2004) found that<br />
Mayan women’s management of Ramón trees (Brosimum alicastrum), a dry-season<br />
forage source found in all homegardens in their study area in the Yucatan, is based<br />
upon an intimate knowledge of environmental factors that are taken into account<br />
when propagating the species, where their management techniques were found to<br />
increase growth very substantially.<br />
Gardening knowledge is not confined to agroecology and agronomy. Garden<br />
planning for subsistence purposes must combine an understanding of vegetative<br />
cycles, of perishability and processing and storage characteristics, and of timing and<br />
quantity of demand, including the needs for ingredients for specific dishes and/or<br />
medicines and substitutability of those ingredients (Lope Alzina, 2006), and of<br />
the need to meet nutritional and medicinal requirements of households whose<br />
composition also changes over time. Finerman and Sackett (2003) showed that<br />
the composition of homegardens in their study area closely reflects the stage in the<br />
life cycle, where the medicinals produced reflect in part the specific needs of young
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 177<br />
families or elderly household members. Several other researchers confirm that<br />
homegardens provide the basis for acquiring much environmental, agronomic,<br />
cultural and other knowledge related to plants and plant uses (Alvarez Buylla et al.,<br />
1989; Angel Peréz and Mendoza, 2004; Greenberg, 1996).<br />
Indigenous knowledge associated with gardening is also related to plants as<br />
cultural capital, where individual plants take on social meaning. For example, Sereni<br />
Murrieta and Winklerprins (2003) found that women were able to relate the history<br />
of many individual plants, their origins, utility, and their status as a gift, a symbol of<br />
someone’s affection or a commemoration of an event (see also Finerman and<br />
Sackett, 2003). Much ritualistic knowledge may also be entailed in gardening as<br />
Descola’s (1994) work on the Achuar amply testifies.<br />
It can therefore be presumed that knowledge entailed in managing complex<br />
gardens takes a considerable part of a lifetime to accrue, involves considerable<br />
hands-on experience and trial and error (experimentation), and entails continual<br />
exchange of information. It is clear that, across the region, women are more often<br />
homegardening specialists; as principle knowledge holders, it can be hypothesized<br />
that it is also women who are primarily responsible for the transmission of<br />
homegardening knowledge. In other words, homegardening knowledge and<br />
knowledge transmission are largely gender-related.<br />
There is ample testimony to gendered gardening knowledge in the literature<br />
reviewed in this chapter. In one case where women are nearly exclusively<br />
responsible for homegardening, it was reported that “men generally disavow any<br />
knowledge of homegardening, deferring to their wives for even basic information<br />
about gardens and their products” (Finerman and Sackett, 2003). In another such<br />
case, Descola (1994) came to an even more dramatic conclusion: Achuar men “are<br />
. . . totally incapable of replacing their wives if the need arises, and moreover have no<br />
desire to do so. When a man no longer has any woman (mother, wife, sister, or<br />
daughter) to cultivate his garden and prepare his food, he has no choice but to kill<br />
himself.”<br />
However, it is more common that both men and women have homegardening<br />
knowledge and that the division of such knowledge reflects the nature of their<br />
involvement. Such a conclusion is born out by an unusual study 2 that researched<br />
gendered species knowledge in 23 households in the Nicoya Peninsula of Costa<br />
Rica, where both women and their husbands participate in homegardening. The 13<br />
homegarden species most frequently used were selected: four exclusively for<br />
medicinal use, five for medicinal and food use, and four exclusively for food. The<br />
results showed that women’s knowledge of medicinal plants was always higher than<br />
men’s. Regarding food plants, only for Musa spp. (plantains and bananas) did the<br />
knowledge between men and women differ significantly, where men had greater<br />
knowledge than women. The authors related these findings to the gender division of<br />
labor where women were responsible for health care and food preparation and men<br />
for cash crop production, and six out of nine of the food items studied had<br />
commercial values.<br />
It is also important to stress that gardening knowledge, like ethnobotanical or<br />
ethnobiological knowledge in general, varies not only between men and women, but<br />
as well according to factors such as kinship, age, social class, ethnicity,
178 P.L. HOWARD<br />
specialization, and personal propensity (Howard, 2003). As Greenberg (1996)<br />
reported among the Maya of Quintana Roo, Mexico, “There are individual<br />
differences in the intensity of peoples’ interests in plants and their cultivation.”<br />
When examining knowledge transmission networks and processes of knowledge<br />
erosion, the influence of kinship and age also comes to the fore. Knowledge<br />
transmission is a dynamic and continual process since household circumstances and<br />
ecological and economic conditions change continually, and homegardens must be<br />
adapted to such changes. In this, women and their social networks play a<br />
predominant role. Several authors show that homegardening knowledge is transmitted<br />
largely among women and then principally among closely related kin (Boster,<br />
1985a; Descola, 1994; Greenberg, 1996; Keys, 1999; Patterson, 2000). Children’s<br />
labor in homegardening is so common that it is not surprising that much general<br />
knowledge is transmitted to them as they work under the supervision of their<br />
mothers. Keys (1999), whose research specifically focused on homegarden<br />
knowledge transmission among the Kaqchikel Maya of Guatemala, showed how<br />
homegardens act as veritable classrooms for both girls and boys where women teach<br />
children how to use farm tools and to cultivate and manage crops. What boys learn<br />
is not only applicable to the homegarden. Keys observed that boys have already<br />
learned the basic concepts of cultivation before they accompany their fathers to the<br />
milpa. Not only cultivation techniques, but as well knowledge about the use of<br />
plants for food, medicine and handicrafts, are transmitted from mother to child<br />
through homegardens. Patterson (2000), working within Mayan communities in<br />
Belize, showed that it was often not only mothers, but also other female relatives,<br />
who formed key knowledge transmission networks. All gardeners interviewed stated<br />
that they acquired environmental and homegarden management knowledge from<br />
older female family members, whereas 94% said they also acquired environmental<br />
knowledge from “other” female family members including younger sisters and more<br />
distant relations. The process of knowledge transmission begins at age five or six<br />
when girls accompany their female relatives to gardens where they learn to identify,<br />
water, and harvest or collect plants and to tend small animals. Hoffman (1993)<br />
found that not only was gardening knowledge transmitted between mothers and<br />
daughters: plant material as well as knowledge, material, skills and practices were<br />
often part of a “package” of cultural and physical capital that flows among women<br />
and between women and their offspring. Boster (1985b) also reported this for<br />
women Aguaruna manioc cultivators in northern Peru.<br />
Some homegarden researchers remark that homegarden knowledge is eroding or<br />
is likely to erode in the near future. They provide several reasons for this, some of<br />
which are gender-specific. One is cultural erosion: as young people assimilate into a<br />
dominant culture through education and migration, they learn less about plants and<br />
homegardening (Angel Peréz and Mendoza, 2004). Benjamin (2000) cites<br />
emigration among Mayan youth as the main risk. Keys (1999) pointed out that<br />
particularly young Guatemalan women are affected by off-farm employment in<br />
textile factories, which leaves them no time for homegardening, and Hoffman<br />
(1993) stressed not only off-farm employment, but also migration and participation<br />
in formal educational systems that denigrate women’s traditional gardening<br />
practices, which leads to loss of knowledge.
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 179<br />
7. CONCLUSIONS<br />
Many women in Latin America contribute to subsistence and to meeting the cash needs<br />
of their families but usually do so in ways that are not permitted to be predominant or<br />
very visible. Swidden gardening and other work that Amazonian Amerindian women<br />
perform accord them social status and prestige. Although men mainly clear and allocate<br />
swidden garden land to women, women have strong if not exclusive claims to most<br />
swidden garden resources. Many Mayan and mestizo women are far less likely than men<br />
to own property, and are generally not permitted to engage in agricultural production or<br />
to generate substantial amounts of income. Still, as ‘acceptable’ social and environmental<br />
spaces where domesticity is centered and esteemed, homegardens offer these women<br />
sources of authority, autonomy, status, social networks and visible ‘public’ spaces of<br />
recognition without challenging male dominance. <strong>Homegardens</strong> are clearly essential to<br />
women: they fit in well with their domestic duties, labor patterns, productive decisionmaking<br />
spheres, aesthetic sensibilities, and cultural roles. Through gardening, women<br />
develop great knowledge and proficiency in relation to the plant world and to the<br />
environment, which permits them to shape and manage these to meet the needs of their<br />
households. In addition to utilitarian or monetary values, homegarden species have<br />
deeper, spiritual emotional, and symbolic meaning for women whose spaces and<br />
relations are circumscribed by historically and culturally-specific phenomena that<br />
relegate them to subordinate positions; they are also assertions (and continual<br />
reassertions) of women’s importance, contributions and the continuity of traditions and<br />
identity that they bring to their societies, families and communities. In this, homegardens<br />
serve men as much as they serve women. They permit women to contribute to family<br />
subsistence, status, and identity in ways that are ‘respectable’.<br />
Women can more readily enter markets where they do not compete with men or<br />
when they do not earn so much income that they challenge men’s economic<br />
predominance. Beyond this, they also appear to be able to negotiate change with<br />
husbands and other family members based upon their authority as garden managers.<br />
The terms of such negotiations may be restrictive, but they may also afford women<br />
the ability to meet their own particularistic needs, and may contribute positively to their<br />
status and increase their ability to “have a greater say” in the management of their<br />
households and communities. On the other hand, this does not negate the fact that<br />
women’s command over homegarden resources is tenuous and likely to shift as<br />
commercialization increases, and they may also lose access altogether in the case of<br />
separation or divorce. Homegardening may also be seen as a source, or a continuing<br />
reminder, of women’s subordinate status, and change processes may leave them<br />
bereft of control over or access to homegarden resources.<br />
Threats to homegardening are many – as are the driving forces to maintain them<br />
that are mentioned above. Commoditization, the decreasing status of local<br />
agrobiodiversity in human consumption and health due to acculturation, urbanization<br />
which draws youth away from primary production, and formal education that denigrates<br />
‘traditional’, ‘peasant’ or ‘indigenous’ ways of life, can all be major threats since they<br />
subvert many of the dynamics that have maintained the value of homegardens –<br />
diversity, independence and autonomy, cultural identity, local adaptability, home- and<br />
needs-centeredness, and multi-value production. Many of these threats at the same time
180 P.L. HOWARD<br />
may offer women greater formal equality and autonomy. But it is likely that women will<br />
attempt to negotiate the trade-offs between such potential gains and losses – as ample<br />
literature on homegardens among urban migrant populations attests, women are very<br />
likely to continue to exert every effort to create, maintain and manage the most socially<br />
and agroecologically complex systems known to the region.<br />
ENDNOTES<br />
1. Lok R. 2001. A better understanding of traditional homegardens through the use<br />
of locally defined management zones. Indigenous Knowledge and Development<br />
Monitor 9: 14 – 18.<br />
2. Ochoa L., Fassaert C., Somarriba E. and Schlonvoight A. 1998. Conocimiento<br />
de mujeres y hombres sobre las especies de uso medicinal y alimenticio en<br />
huertos caseros de Nicoya, Costa Rica. Agroforestería en las Américas 5:<br />
17 – 18, 7 – 11.<br />
3. Samaniego G. and Lok R. 1998. Valor de la percepción y del conocimiento<br />
local de indígenas Ngöbe, en Chiriqui, Panamá. Agroforestería en las Américas<br />
5: 17 – 18, 12 – 16.<br />
4. Veth B. and Reinders M. 1995. Planten zijn als mensen: genezen met<br />
medicinale planten bij de Warao en Wayana in de Guyana's. Indigo 3(5): 4 – 7.<br />
REFERENCES<br />
Aikman S. 1999. Schooling and development: Eroding Amazon women’s knowledge and<br />
diversity. In: Heward C. and Bunwaree P. (eds), Gender, education and development:<br />
Beyond access to empowerment, pp. 99 – 153. Zed Press, London.<br />
Alexiades M.N. 1999. Ethnobotany of the Ese Eja: Plants, health and change in an<br />
Amazonian Society (Plant Medicinals). PhD dissertation, City University of New York.<br />
University Microfilms International, Ann Arbor, Michigan.<br />
Alvarez Buylla M., Lazos Chavero M.A. and García Barrios J.R. 1989. <strong>Homegardens</strong> of a<br />
humid tropical region in South East Mexico: An example of an agroforestry cropping<br />
system in a recently established community. Agroforest Syst 8: 133 – 156.<br />
Angel Peréz, A. del and Mendoza B., M.A. 2004. Totonac homegardens and natural resources<br />
in Veracruz, Mexico. Agric Hum Val 21: 329 – 346.<br />
Baleé W.L. 1994. Footprints of the forest: Ka’apor ethnobotany–The historical ecology of plant<br />
utilization by an Amazonian people. Columbia University Press, New York pp. 50 – 60; 158.<br />
Benjamin T.J. 2000. Maya cultural practices in Yucatecan homegardens: An ecophysiological<br />
perspective. PhD dissertation, Purdue University. University Microfilms International,<br />
Ann Arbor, Michigan.<br />
Blanckaert I., Swennen R.L., Paredes Flores M., Rosas Lopez R. and Lira Saade R. 2004.<br />
Floristic composition, plant uses and management practices in homegardens of San Rafael<br />
Coxcatlán, Valley of Tehuacán-Cuicatlán, Mexico. J. Arid Env 57: 39 – 62.<br />
Boster J.S. 1985a. Requiem for the omniscient informant: there’s life in the old girl yet. In:<br />
Dougherty J. (ed.), Directions in cognitive anthropology, pp. 177 – 198. University of<br />
Illinois Press, Champaign, Illinois.<br />
Boster J.S. 1985b. Selection for perceptual distinctiveness: Evidence from Aguaruna cultivars<br />
of Manihot esculenta. Econ Bot 39: 310 –325.
GENDER G AND SOCIAL DYNAMICS IN LATIN L AMERICA 181<br />
Bruce J. and Fortmann L. 1988. Why land tenure and tree tenure matter: Some fuel for<br />
thought. In: Fortmann L. and Bruce J.W. (eds), Whose trees? Proprietary dimensions of<br />
forestry, pp 1 – 9. Westview Press, Boulder and London.<br />
Deere C.D. and Leon M. 2001. Empowering women: Land and property rights in Latin<br />
America. University of Pittsburgh Press, Pittsburgh pp. 264 – 291.<br />
Descola P. 1994. In the society of nature: A native ecology in Amazonia, pp 93 – 212.<br />
Cambridge University Press, Cambridge.<br />
Doxon L.E. 1988. Diversity, distribution and use of edible and ornamental plants in<br />
homegardens in Honduras. PhD Dissertation, Kansas State University. University<br />
Microfilms International, Ann Arbor, Michigan.<br />
Dufour D.L. 1981. Household variation in energy flow in a population of tropical forest<br />
horticulturalists. PhD dissertation. State University of New York at Binghamton.<br />
University Microfilms International, Ann Arbor, Michigan.<br />
Finerman R. and Sackett R. 2003. Using homegardens to decipher health and healing in the<br />
Andes. Med Anth Quart 17: 459 – 482.<br />
Gillespie A.R., Bocanegra Ferguson D.M. and Jiménez Osornio J.J. 2004. The propagation of<br />
Ramón (Brosimum alicastrum Sw.; Moraceae) in Mayan homegardens of the Yucatan<br />
peninsula of Mexico. New Forests 27: 25 – 38.<br />
Goldman I. 1963. The Cubeo: Indians of the North-west Amazon. University of Illinois Press,<br />
Champaign, Illinois.<br />
Govers C. 1997. Agriculture, cosmology and the gender division of labour in a Totonac<br />
highland village (Mexico). In: de Bruijn M. and van den Hombergh H. (eds), Gender and<br />
land use: Diversity in environmental practices, pp. 27 – 47. Thela Publishers, Amsterdam.<br />
Greenberg L.S. 1996. You are What you eat: ethnicity and change in Yucatec immigrant<br />
house lots, Quintana Roo, Mexico. PhD dissertation, University of Wisconsin-Madison.<br />
University Microfilms International, Ann Arbor, Michigan.<br />
Heckler S.L. 2001. The ethnobotany of the Piaroa: Analysis of an Amazonian people in<br />
transition. PhD dissertation, Cornell University. University Microfilms International, Ann<br />
Arbor, Michigan.<br />
Heckler S.L. 2004. Tedium and creativity: The valorization of manioc cultivation and Piaroa<br />
women. J Roy Anth Inst 10: 241 – 259.<br />
Hoffman S.D. 1993. Subsistence in transition: Indigenous Agriculture in Amazonas,<br />
Venezuela. PhD Dissertation, University of f California Berkeley. University Microfilms<br />
International, Ann Arbor, Michigan.<br />
Howard P. 2003. Women and plants: An exploration. In: Howard P. (ed.), Women and plants:<br />
Gender relations in biodiversity management and conservation, pp. 1 – 47. Zed Press and<br />
Palgrave Macmillan, London and New York.<br />
Howard P. and Nabanoga G. 2006. Are there customary rights to plants? An inquiry among<br />
the Baganda (Uganda), with special attention to gender. World Devel (in press).<br />
Irvine D. 1987. Resource Management by the Runa Indians of the Ecuadorian Amazon. PhD<br />
dissertation, Stanford University. University Microfilms International, Ann Arbor, Michigan.<br />
Keys E. 1999. Kaqchikel gardens: Women, children, and multiple roles of gardens among the<br />
Maya of Highland Guatemala. Yearb Conf Lat Am Geogr 25: 89 – 10.<br />
Knauft B.M. 1997. Gender identity, political economy and modernity in Melanesia and<br />
Amazonia. J Roy Anth Inst 3: 233 – 259.<br />
Lazos Chavero E. and Alvarez Buylla M. 1988. Ethnobotany in a tropical-humid region: The<br />
homegardens of Balzapote, Veracruz, Mexico. J Ethnobio 8: 45 – 79.<br />
Lerch N.C. 1999. <strong>Homegardens</strong>, cultivated plant diversity, and exchange of planting material<br />
in the Pacaya-Samiria National reserve area, Northeastern Peruvian Amazon. Masters<br />
thesis, McGill University. University Microfilms International, Ann Arbor, Michigan.
182 P.L. HOWARD<br />
Lope Alzina D. 2006. Gendered production spaces and crop varietal selection: a case study in<br />
Yucatan, Mexico. Sing. J. Trop. Geog (in press).<br />
Madaleno I. 2000. Urban agriculture in Belém, Brazil. Cities 17: 73 – 77.<br />
Martin G.J. 1996. Comparative ethnobotany of the Chinantec and Mixe of the Sierra Norte,<br />
Oaxaca, Mexico. PhD dissertation, University of California, Berkeley. University<br />
Microfilms International, Ann Arbor, Michigan.<br />
Mendez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in Nicaragua:<br />
Micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51: 85 – 96.<br />
Michon G. 1983. Village forest gardens in West Java. In: Huxley P.A. (ed.), Plant research and<br />
agroforestry, pp. 13 – 24. International Centre for Research in Agroforestry, Nairobi, Kenya.<br />
Montagnini F. 2006. Status of homegardens in Mesoamerica In: Kumar B.M. and Nair P.K.R.<br />
(eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp. 61–<br />
84. Springer Science, Dordrecht..<br />
Murray S.J. 2001. Plants in the ‘Patxokon Na’: Tzotzil Maya homegardens in the highlands of<br />
Southeastern Mexico. PhD dissertation, Wayne State University. University Microfilms<br />
International, Ann Arbor, Michigan.<br />
Palacio J. 1980. Food and social relations in a Garifuna village (Belize). PhD dissertation,<br />
University of California at Berkeley. University Microfilms International, Ann Arbor,<br />
Michigan.<br />
Patterson M.L. 2000. Agroforestry in Belize: Maya homegardens in San Lucas. Masters<br />
thesis, University of Alberta. University Microfilms International, Ann Arbor, Michigan.<br />
Posey D.A. 1984. A preliminary report on diversified management of tropical forest by the<br />
Kayapó Indians of the Brazilian Amazon. In: Prance G.T. and Kallunki J.A. (eds),<br />
Ethnobotany in the neotropics: Proceedings. Ethnobotany in the Neotropics Symposium,<br />
Society for Economic Botany (13-14 June 1983), Oxford, Ohio. Adv Econ Bot 1: 112 –<br />
126. New York Botanical Garden, New York.<br />
Roquas E. 2002. Stacked law. Land, property and conflict in Honduras. PhD dissertation,<br />
Wageningen University. Rozenberg Publishers. Amsterdam.<br />
Ruonavaara D.L. 1996. Traditional household gardens of the Petén, Guatemala. Masters thesis,<br />
Michigan State University. University Microfilms International, Ann Arbor, Michigan.<br />
Salick J. 1997. Subsistence and the single woman among the Amuesha of the Upper Amazon,<br />
Peru. In: Sachs C. (ed.), Women working the environment, pp. 139 – 153. Taylor and<br />
Francis, Washington DC.<br />
Sereni Murrieta R.S. and Winklerprins A. 2003. Flowers of water: <strong>Homegardens</strong> and gender roles<br />
in a riverine Caboclo community in the lower Amazon, Brazil. Cult Agri 25: 35 – 47.<br />
Stavrakis O. 1979. The effects of agricultural change upon social relations in a village in<br />
Northern Belize, Central America. PhD dissertation, University of Minnesota. University<br />
Microfilms International, Ann Arbor, Michigan.<br />
Thompson S. 1977. Women, horticulture, and society in tropical America. Am Anthrop 79:<br />
908 – 910.<br />
Uzendoski M.A. 2004. Manioc beer and meat: Value, reproduction and cosmic substance<br />
among the Napo Runa of the Ecuadorian Amazon. J Roy Anth Inst 10: 883 – 902.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp. 13 – 24. Springer Science, Dordrecht.<br />
Wilson W.M. 1997. Why bitter cassava (Manihot esculenta Crantz)? Productivity and<br />
perception of cassava in a Tukanoan Indian settlement in the Northwest Amazon<br />
(Colombia). PhD dissertation, University of Colorado at Boulder. University Microfilms<br />
International, Ann Arbor, Michigan.<br />
Winklerprins A. 2002. House-lot gardens in Santarém, Pará, Brazil: linking rural with urban.<br />
Urb Ecosyst 6: 43 – 65.
SECTION 3<br />
SOME NEW THRUST AREAS
CHAPTER 11<br />
CARBON SEQUESTRATION POTENTIAL<br />
OF TROPICAL HOMEGARDENS<br />
B.M. KUMAR<br />
Department of Silviculture and Agroforestry, College of Forestry, Kerala<br />
Agricultural University, Thrissur 680656, Kerala, India; E-mail:<br />
<br />
Keywords: Carbon stocks, Multistrata systems, Net primary productivity, Soil organic<br />
matter.<br />
Abstract. This chapter examines the premise that tropical homegardens have a special role in<br />
carbon (C) sequestration because of their ability for carbon storage in the standing biomass,<br />
soil, and the wood products. In doing so, it analyzes the potential for C storage in<br />
homegardens and the role of homegardens in reducing CO 2 concentration in the atmosphere.<br />
Lack of reliable inventories/estimates and uncertainties in the estimation of C sequestration<br />
potential of homegardens present formidable difficulties in the analysis. Nevertheless,<br />
available information indicates that homegardening has a higher potential to sequester C<br />
compared to monospecific production systems, and the costs are lower than emission<br />
reduction or sequestration by other means. Indeed, the C sequestration potential of<br />
homegardens that mimic the structure and diversity of mature evergreen forest formations is<br />
comparable to that of such forest stands. Although experimental evidence suggests that<br />
species diversity does not necessarily mean high C sequestration, complementary or<br />
compensatory gains in resource acquisition, possibility of biological N 2 fixation and the<br />
relatively low herbivory pressure, may explain this high C sequestration ability of<br />
homegardens. Extension of homegardens into more lands and adaptive management of the<br />
existing gardens offer scope for enhanced C sequestration and economic gains.<br />
1. INTRODUCTION<br />
The Intergovernmental Panel on Climate Change (IPCC) Special Report on Land<br />
Use, Land Use Change and Forestry (LULUCF) suggests that the average annual<br />
accounted carbon stock changes in the first commitment period (2008–2012),<br />
resulting from afforestation and reforestation, would be between 197 and 584 Tg C<br />
year –1 (Watson et al., 2000). Agroforestry, including the homegarden, plays a<br />
cardinal role in this respect—net changes in global C stocks are estimated to be 26<br />
185<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 185–204.<br />
© 2006 Springer. Printed in the Netherlands.
186 B.M. KUMAR<br />
Tg C year –1 for better agroforest management and 390 Tg (million tons) C year –1<br />
r for<br />
agroforestry-related land use changes in 2010 (Watson et al., 2000). Information<br />
relating to this substantial, but under-exploited potential of agroforestry as a carbon<br />
sequestration strategy has been recently reviewed by Albrecht and Kandji (2003)<br />
and Montagnini and Nair (2004). This chapter is a follow up to these reports, with<br />
the objective of examining the role of tropical homegardens as a mechanism for<br />
carbon sequestration, on which presently little or no concrete information exists.<br />
Furthermore, an attempt is made to examine carbon restitution (above- and<br />
belowground) as a function of species richness in homegardens, using biomass<br />
productivity as a ‘proxy’ of carbon sequestration by comparing several woody<br />
perennial-based polycultures.<br />
2. EXTENT OF HOMEGARDENING<br />
Although homegardens are an age-old practice in many parts of the tropics and even<br />
other parts of the world (Kumar and Nair, 2004; Nair and Kumar, 2006), only<br />
limited data are available on their extent and distribution. The available information<br />
suggests that Indonesian homegardens or pekarangan cover about 5.13 million ha of<br />
land, of which 1.74 million ha are in Java 1 . Homesteads cover about 0.54 million ha<br />
in Bangladesh 2 and 1.05 million ha in Sri Lanka 3 (which constitutes about 60% of<br />
the land holdings
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
187<br />
than 2000 g C m –2<br />
m yr –1 , is imperative; Kaye et al., 2000), carbon conservation<br />
(easing of anthropogenic pressure on existing stocks of C in forests through<br />
conservation and management efforts), and carbon substitution (substitution of<br />
energy demand materials by renewable natural resources, fuelwood production,<br />
increased conversion of biomass into durable wood products for use in place of<br />
energy-intensive materials; Kürsten, 2000).<br />
While most agroforestry systems are important in respect to one or the other<br />
mechanisms mentioned above (Ruark et al., 2003), the homegardens perhaps are<br />
unique in that all three mechanisms are relevant; i.e., they sequester C in biomass<br />
and soil, reduce fossil-fuel burning by promoting woodfuel production, help in the<br />
conservation of C stocks in existing forests by alleviating the pressure on natural<br />
forests and ensure greater synergy with the Convention on Biological Diversity<br />
(CBD). Moreover, there is no complete removal of biomass from the homegardens<br />
(Gajaseni and Gajaseni, 1999), signifying the permanence of the system. While lack<br />
of stability or permanence of the C sequestered being a major concern in LULUCF<br />
C sequestration projects (UNFCCC, 2002), the homegarden system is remarkably<br />
resilient. Additionally, C storage can last for decades if boles, stems or branches are<br />
processed in any form of long-lasting products (Roy, 1999) and the homegarden<br />
system has reasonable prospects in that respect too (Kumar et al., 1994).<br />
Available reports and case studies on biomass production/carbon sequestration<br />
potential of tropical homegardens are summarized in Table 1, and are compared<br />
with other tropical land use systems. One particular problem, however, is the<br />
profound age-related variations in the C stocks of different land use activities.<br />
Although Tomich et al. (2002) suggested that time-averaged C stocks (e.g., half the<br />
system’s C stock at its maximum age or rotation length) will be appropriate to<br />
compare C stocks of different land use systems on a scale that adjusts C stocks of<br />
the systems to their ages, adequate information on this aspect is not available in<br />
many case studies. Notwithstanding such intrinsic variability and assuming that<br />
homegardens are “steady-state systems” (Kumar and Nair, 2004), Table 1 is an<br />
attempt to compare different systems. As expected, the multi-layered woody<br />
perennial dominated systems have higher C sequestration potentials than other<br />
comparable systems. For example, the Javanese and Sumatran homegardens<br />
accumulated C in the range of 55.8 to 162.7 Mg ha –1 [19 Sumatran homegardens of<br />
12 to 17 years age (Roshetko et al., 2002) and a Javanese garden of undefined age<br />
(Jensen, 1993)], which is considerably greater than monocultures of annual crops,<br />
most woodlots and simple agroforests (with one dominant species such as oil palm,<br />
cacao or coffee). Likewise, the data show that a shift from single-crop production<br />
systems to multistrata systems increased the C sequestration potential. For example,<br />
conversion of all “sun-coffee” to “shade coffee” systems in Sumatra increased<br />
average landscape level C stocks by an estimated 10 Mg C ha −1 during a 20-year<br />
period (van Noordwijk et al., 2002). Monospecific woodlots also accumulate<br />
substantial C in their biomass – which is, however, dependent on the species, site,<br />
and management (Table 1).<br />
In certain cases, the aboveground C in homegardens is on par with the C stocks<br />
reported for similar-aged secondary forests (e.g., Jensen, 1993); but lower than that
Land use practice Duration<br />
[years]<br />
Table 1. Carbon uptake rates and carbon stocks of prominent land use systems in the tropics 1 .<br />
C uptake [Mg<br />
C ha –1 yr –1 ]<br />
C stocks [Mg<br />
C ha 1 ] 1<br />
Remarks and source<br />
Primary and logged forest Unknown 0 192 to 276 Sum of above-and below-ground C; summary of<br />
116 sites within different land uses before and<br />
after slash and burn from Brazil, Cameroon,<br />
Indonesia and Peru (time averaged system C<br />
stocks; Sanchez, 2000)<br />
Cropping after slash and burn 2 –76 to –112 39 to 52 As above<br />
Crops/bush fallow 4 2 to 4 32 to 36 As above<br />
Tall secondary forest fallows 23 5 to 9 95 to 142 As above<br />
Complex agroforests 25 to 40 2 to 4 65 to 118 As above<br />
Simple agroforests 15 5 to 9 65 to 92 As above<br />
Pastures, Imperata grasslands 4 to 12 –0.2 to –0.6 27 to 31 As above<br />
Indonesian homegardens, Sumatra 13.4 - 107.2 ± 37.2<br />
[range: 55.8 to<br />
162.7]<br />
Sum of aboveground, litter, herbs, soil and roots<br />
(35.3, 2.0, 0.3, 60.8 and 8.8 Mg C ha –1<br />
respectively; Roshetko et al., 2002)<br />
Javanese homegarden, Legokole - - 63 Total biomass (including 4.4 Mg ha –1 of ground<br />
litter; Jensen 1993) were converted to carbon<br />
stocks by multiplying with 0.5<br />
–<br />
188<br />
B.M. KUMAR
Forest remnants, Lampung, Indonesia - - 262 Time averaged total C stock above –0.3m in the<br />
soil (van Noordwijk et al., 2002)<br />
Shade-coffee, Lampung, Indonesia - - 82 As above<br />
Sun-coffee (monoculture), Lampung, - - 52 As above<br />
Indonesia<br />
Monospecific stands of Casuarina<br />
equisetifolia, Eucalyptus robusta and<br />
Luecaena leucocephala, Puerto Rico<br />
Woodlots off Acacia auriculiformis,<br />
Ailanthus triphysa, Artocarpus<br />
heterophyllus, Artocarpus hirsutus,<br />
Casuarina equisetifolia, Emblica<br />
officinalis, Leucaena leucocephala,<br />
Paraserianthes falcataria and<br />
Pterocarpus marsupium, Kerala,<br />
India<br />
Amazonian forests: Terra firme, Tall<br />
Caatinga and Tall Bana forests<br />
4 - 128.3, 115.7<br />
and 116.9<br />
respectively<br />
Sum of aboveground, litter, herbs, soil and root<br />
C; unplanted control had 83.2 Mg C ha –1<br />
(Parrotta, 1999)<br />
8.8 - 26.3 to 178.4 Sum of bole, branch, foliage, roots (Kumar<br />
et al., 1998) and detrital C (Jamaludheen and<br />
Kumar, 1999): converted to carbon stocks by<br />
multiplying with 0.5<br />
Unknown - 152, 178 and<br />
155 respectively<br />
Above + belowground C (Cuevas and Medina<br />
1986)<br />
Table 1 (cont.)<br />
CARBON<br />
C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
189
Land use practice Duration<br />
[years]<br />
C uptake [Mg<br />
C ha –1 y –1 ]<br />
C stocks [Mg<br />
C ha –1 ]<br />
Remarks and source<br />
Natural forests, Jambi, Indonesia 120 - 500 Aboveground C: (cited from Roshetko et al.,<br />
2002)<br />
Central American lowland forests Unknown 146 Average for six forest types s (above +<br />
belowground: 114 + 32 Mg ha –1 ); Sanford and<br />
Cuevas (1996)<br />
Mature agroforests, Sumatra 30 - 101 Aboveground C: Roshetko et al. (2002)<br />
Secondary forests, Sumatra 30 - 86 As above<br />
Young agroforests, Sumatra 9 - 14 As above<br />
Cassava, Sumatra 0.3 - 0.5 As above<br />
Cordia alliodora + cacao, Turrialba,<br />
Costa Rica<br />
initial<br />
5<br />
10<br />
98 (soil) 156.6<br />
213.8<br />
Perennial C stock [i.e., soil + tree + cacao (cacao<br />
branches and stems), tree stems, estimated 85%<br />
of roots (coarse root proportion), and Cordia<br />
branches)]; Beer et al. (1990) cited in<br />
Montagnini and Nair (2004)<br />
190 B.M. KUMAR
Erythrina poeppigiana + cacao,<br />
Turrialba, Costa Rica<br />
initial<br />
5<br />
10<br />
220.8<br />
Mature forests, Mekoe, Cameroon Unknown 270 Biomass (sum of tree, understorey, litter and<br />
roots; Duguma et al., 2001) converted to carbon<br />
stocks by multiplying with 0.5<br />
Cacao agroforests, Mekoe, Cameroon 26 152 As above<br />
115<br />
159.8<br />
As above; except for Cordia branches<br />
Tectona grandis plantation, Panama 20 120.2 Sum of above (104.5 Mg ha –1 ) and belowground<br />
C (15.7 Mg ha –1 ); Kraenzel et al. (2003)<br />
Food crop fields, Mekoe, Cameroon - 43 Biomass (sum of tree 2 , understorey, and roots;<br />
Duguma et al., 2001) converted to carbon stocks<br />
by multiplying with 0.5<br />
1 Also, see Schroeder (1994) who estimated the average carbon storage by agroforestry practices as 9, 21, 50, and 63 Mg C ha −1 for semiarid, subhumid,<br />
humid, and temperate regions and Albrecht and Kandji (2003) who reported a value between 12 and 228 Mg C ha −1 with a median value of 95 Mg ha −1 .<br />
2 When land was cleared, indigenous fruit, medicinal and timber trees (e.g., Ricinodendron heudelotii, Cola nitida, Voacanga africana, Triplochiton<br />
sclerozylon etc.) were deliberately retained.<br />
CARBON<br />
C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
191
192<br />
accumulated by the mature forests in the region (114 to 500 Mg aboveground C ha –1<br />
Table 1). Indeed, the homegardens resemble young secondary forests in structure<br />
and biomass accumulation and may be considered as a human-made forest kept in a<br />
permanent early successional state with considerable productive potential. Consistent<br />
with this, in a study on Kerala homegardens, Kumar et al. (1994) showed that the<br />
average standing stock of commercial timber ranged from 6.6 to 50.8 m 3<br />
ha –1 . Overall, the data presented shows that the homegardens that mimic the<br />
structure and diversity of mature evergreen forest formations (Fig. 1) rank very close<br />
to mature forests in their biomass C storage potential (Table 2). This observation is<br />
based, however, on a few datasets, and should be followed up with more rigorous<br />
studies.<br />
Figure 1. Diversity, multistrata canopy structure, and various functional groups of food, fuel,<br />
fruit and nut yielding plants in a Kerala homegarden [coconut palms (Cocos nucifera), areca<br />
or betel nut palms (Areca catechu), jackfruit tree (Artocarpus heterophyllus), black pepper<br />
vines (Piper nigrum), plantains (Musa a spp.) and the like].<br />
3.1. Uncertainties in estimating homegarden C stocks<br />
Since net ecosystem productivity generally reflects the overall gain or loss of<br />
terrestrial C pools (Nair and Nair, 2003), larger C sinks are probable when croplands<br />
(input-intensive production systems) are converted into homegardens (sensu<br />
.<br />
B.M. KUMAR
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
193<br />
Houghton and Goodale, 2004) than simple agroforests/plantations. Information on<br />
the actual rate of change in homegarden coverage and the spatial and temporal<br />
heterogeneity in C stocks are, however, not available. Lack of such data at the<br />
landscape-level particularly hampers our understanding of the potential of<br />
homegarden systems to sequester C and its eventual use in C sink projects, which is<br />
a situation that is common to most agroforestry systems (Montagnini and Nair,<br />
2004).<br />
Yet another challenge is the difficulty in estimating tree biomass itself. Despite<br />
the fact that most trees accumulate C in their wood, precise estimates on the C<br />
sequestration potential of several tropical trees are not available (Roshetko et al.,<br />
2002). Aboveground biomass is usually estimated with general regression equations<br />
developed for trees in the natural forests. However, the size of individual tree<br />
canopies in a forest and in an open agroforestry setting could be variable, as the trees<br />
in some agroforestry systems have more space and access to light. In addition, the<br />
crown and root architecture and tree management practices are different; the<br />
resultant variations in structure could probably result in erroneous estimates.<br />
A more important technical issue is the definition of a standard set of methods<br />
and procedures for the inventory and monitoring of C stocks in current and potential<br />
land use and management approaches (FAO, 2004). Differing interpretations of<br />
source and sink category or other definitions, use of simplified representations with<br />
“averaged” values and uncertainties in the basic processes leading to emissions<br />
and/or removals further complicate the matter (de Jong, 2001). In addition, to<br />
estimate the effects of harvest on homegarden C stocks, accurate information on<br />
three items is required: pre-harvest biomass, the fraction of this biomass harvested<br />
or damaged, and the fraction of the harvested biomass removed; much of these are<br />
not available, making estimation of the C sequestration potential of homegardens at<br />
the landscape-level a difficult issue.<br />
4. PLANT DIVERSITY IN HOMEGARDENS AND C SEQUESTRATION<br />
High biodiversity is an intrinsic property of the homegardens (Kumar and Nair,<br />
2004), which presumably favors greater NPP (Vandermeer, 1989) and higher C<br />
sequestration potential than monospecific production systems. This could be because<br />
diverse assemblages (Fig. 2) have a greater likelihood of containing species with<br />
strong responses to resources compared to species-poor assemblages (Tilman et al.,<br />
1997). The inference that diversity leads to greater NPP and thus stability of<br />
ecosystems, however, is the subject of an ongoing debate in ecology (McCann,<br />
2000). That is, although homegardens and other multistrata systems are assumed to<br />
promote NPP and improve the soil and biomass C sequestration (Table 2), often<br />
doubts are expressed concerning the productive capacities of species mixtures<br />
(FAO, 1992; Wedin and Tilman, 1993). In particular, asymmetric competition<br />
(resource acquisition at differential rates; Wedin and Tilman, 1993) and thereby<br />
resource pre-emption by the dominant component of a competing mixture may<br />
retard their productive potential.
Attributes Types of land use system<br />
C stocks 1<br />
Aboveground<br />
Soil<br />
Fossil fuel inputs/subsidies (C costs) 2<br />
Table 2. Summary of the relative attributes of a land use continuum in the tropics. o<br />
Intensive monoculture Polyculture<br />
Annual crops Perennial Simple <strong>Homegardens</strong><br />
crops/<br />
and complex<br />
plantations<br />
multistrata<br />
systems<br />
agroforests 9<br />
Secondary<br />
forests<br />
Mature<br />
forests<br />
low<br />
low-high medium-high medium-high very high<br />
low<br />
low-high low-high medium-high medium-high very high<br />
high medium-high low- low zero zero<br />
medium<br />
Ecosystem services 3 low low-medium low-high medium-high high very high<br />
Diversity 3<br />
low low medium high very high very high<br />
Herbivory pressure 4<br />
high high medium low low very low<br />
Loss rate of soil C (decomposition) 5 high-very high medium medium low-medium low-medium low<br />
Nutrient outputs 6 (leaching/other high medium medium low low very low<br />
losses)<br />
Soil biota 7 low low medium high high very high<br />
low-high 8<br />
194 B.M. KUMAR
1 Low (121 Mg C ha –1 ); upper limits represent the midpoints<br />
of the range of values reported by Sanchez (2000) for pastures, simple agroforests and complex agroforests rounded to the nearest multiple of 10 (see<br />
Table 1).<br />
2 Annual/perennial crops are usually fertilized, irrigated, and managed with heavy doses of plant protection chemicals; agroforestry in<br />
general is less input intensive and for homegardens, in particular, little or no chemical inputs are used, while the natural systems are selfnourished.<br />
3 Agricultural intensification (e.g., large-scale use of agricultural chemicals) reduces diversity and abundance of biota; for example, the<br />
bees, which render pollination services (Kremen et al., 2002).<br />
4 The natural enemy complex of crop pests/pathogens is generally low in intensive monospecific production systems than in polycul<br />
n tures;<br />
consequently, the herbivory pressure is much lower in the natural and woody perennial-based mixtures (Keenan et al., 1995; Ball et al.,<br />
1995; Jactel et al., 2005).<br />
5 Soil organic matter (SOM), a keystone component of the ecosystem (sensu Swift et al., 2004), is related to the quantity and variability of<br />
plant litter inputs. Higher floristic diversity generally ensures greater litter heterogeneity (Hättenschwiler et al., 2005) and the “speciesrich”<br />
systems generally have a greater chance of maintaining soil organic matter relations than the “species-poor” ones (Russell et al.,<br />
2004).<br />
6 Loss of perennial vegetation leads to erosion, reduced soil quality, and low productivity (Singh et al., 1992; Vinod et al., 2003).<br />
7 Greater organic matter fluxes in woody perennial-based systems favor soil biota (Vohland and Schroth, 1999; Kumar and Nair, 2004).<br />
8 Wherever ranges are mentioned, it denotes variations because of stand age, species, management and/stage of succession.<br />
9 with one dominant species such as oil palm, cacao, coffee and the like.<br />
CARBON<br />
C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
195
196<br />
The implicit assumption in studies reporting the positive “mixture effect,”<br />
however, is that one or more of the components improve the environment<br />
(facilitative production principle; Vandermeer, 1989) and/or share site resources<br />
harmoniously. The contribution of biologically fixed N2 to the associated non-N2<br />
fixing component is particularly relevant in this respect. Legumes in general are<br />
thought to be soil improvers—and may promote the growth and productivity of<br />
components in such systems (Kaye et al., 2000); yet there is no agreement on the<br />
role of woody legumes in promoting growth and NPP of associated woody nonlegume<br />
components. Lack of consistent impacts of the legume components in<br />
experimental mixtures (Parrotta, 1999; Gathumbi et al., 2004), can be explained<br />
based on species, site attributes—especially soil N content and soil management,<br />
ensuring the availability of appropriate rhizobial strains and maintenance of<br />
conditions suitable for their multiplication.<br />
In certain cases, productivity has been linked to site quality; for example, higher<br />
productivity for mixtures on nutrient-poor sites (Montagnini et al., 1995).<br />
Furthermore, there are considerable variations in the C sequestration potential of<br />
individual gardens and species, implying both within- and between- garden<br />
variations (Table 1). Yet, no comparative accounts on homegarden productivity as a<br />
function of its floristic attributes could be found. Issues such as what contributes to<br />
the superior performance of multistrata systems and homegardens also have not<br />
been adequately addressed. Such an analysis, however, is relevant to the CBD to<br />
which land use change, agriculture and forestry activities recognized by the Kyoto<br />
Protocol are closely linked. Aside from the ecological benefits of biological<br />
diversity conservation and improved site fertility, species mixtures offer greater<br />
resistance to insect infestation or disease outbreak (Table 2). A recent review, based<br />
on a meta-analysis of more than 50 field experiments, which contrasted pure stand<br />
vs. mixed stand of the same tree species, demonstrated a significant increase in<br />
insect pest damage in single-species stands (Jactel et al., 2005).<br />
It is probable that the relative superiority is dependent on species/circumstances,<br />
and is not amenable to sweeping generalizations; i.e., the effect may be positive,<br />
negative, or neutral. Ideally, in a mixture, the components should exploit different<br />
vertical layers—both above- and belowground—which signifies greater resource<br />
utilization efficiency. This idea, however, pre-supposes that species with divergent<br />
growth characteristics, be mixed for optimizing resource capture (Kumar et al.,<br />
2001; Gathumbi et al., 2002). An interesting aspect of belowground resource use,<br />
however, is that the proximity of species/individuals often favors competitive<br />
downward displacement of tree roots (Kumar and Divakara, 2001). That is, in<br />
certain cases, species may develop vertically stratified root systems, and this spatial<br />
segregation of the roots of associated plants may abate possible inter specific<br />
competition in species-mixtures (Divakara et al., 2001). By extension, in homegardens,<br />
depending upon the nature of associated tree components, a greater<br />
potential to capture the lower leaching nutrients and accomplishing on-site nutrient<br />
conservation is probable (safety-net mechanism). Therefore, if planned with<br />
consideration for each species’ growth characteristics, mixed stands and homegardens<br />
could, theoretically, be more productive than single species stands and<br />
would probably sequester more C.<br />
.<br />
B.M. KUMAR
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
5. PRODUCTIVITY UNDER RISING ATMOSPHERIC CO2 LEVELS<br />
197<br />
Although it is now clear that high CO2 emission levels (Houghton, 1995) will have<br />
several adverse fallouts, indications are that the elevated CO2 may increase plant<br />
photosynthesis and NPP to some extent (Mingkui and Woodard, 1998). Given that<br />
the capacity of the photosynthetic machinery of C3 plants remains unsaturated at<br />
current concentrations of close to 370 ppm of CO2 (Körner, 2003), this seems<br />
reasonable too (but see Luo et al., 2004). Some experimental evidences also suggest<br />
that plant diversity and composition influence the enhancement of biomass and C<br />
acquisition in ecosystems subjected to elevated atmospheric CO2 concentrations. For<br />
instance, Reich et al. (2001) reported that biomass accumulation was greater in<br />
species-rich than in species-poor experimental populations under conditions of CO2<br />
and N fertilization. By extension, homegardens, which are inherently species-rich,<br />
may trap progressively greater quantities of atmospheric CO2 under rising levels of<br />
this gas. In view of the limited nature and range of the experimental studies reported<br />
(mostly from temperate regions and none on tropical homegardens), however, it is<br />
difficult to draw firm generalizations on the effects of enriched CO2 levels on C<br />
sequestration, especially in the tropics.<br />
6. SOIL CARBON SEQUESTRATION<br />
More than half of the C assimilated by woody perennials is eventually transported<br />
belowground via root growth and organic matter turnover processes (e.g., fine root<br />
dynamics, rhizodeposition, and litter dynamics), making soil organic carbon (SOC)<br />
a significant pool of terrestrial C (~2500 Pg C globally; Lal, 2004). In view of the<br />
great diversity and abundance of woody perennial components, it is perhaps<br />
reasonable to assume that the magnitude of such processes will be greater in<br />
homegardens compared to other systems (Gajaseni and Gajaseni, 1999; Kumar and<br />
Nair, 2004). Judicious management of plant residues as it is often practiced in<br />
homegardens also can contribute to increases in soil organic matter content<br />
(Montagnini, 2006). There is, however, great variation among homegardens in this<br />
respect. For instance, Roshetko et al. (2002) found that SOC of Indonesian<br />
homegardens ranged between 10.4 to 103.7 Mg C ha –1 .<br />
The C stored within the soil may increase and under certain conditions biomass<br />
production also increases, augmenting C inputs (root biomass, litter and prunings)<br />
into the soil (sensu Lal et al., 1998). Consistent with this, Russell (2002) noted that<br />
total SOC may increase directly with basal area of the trees included in the system.<br />
Both inputs and decomposition rates are, however, strongly affected by a host of<br />
factors (Lal et al., 1998) including climate change (Schimel et al., 2000). Warmer<br />
temperatures generally accelerate litter decomposition. However, in view of the<br />
possible stimulatory effects of rising atmospheric CO2 levels on photosynthetic<br />
production and the associated greater litterfall production rates, the effects are<br />
seemingly more complex (Kumar et al., 2005).<br />
Soil organisms such as microflora (bacteria, fungi, actinomycetes and algae),<br />
mesofauna (mites, collembola, micro-arthropods and enchytraeid worms), microfauna<br />
(protozoa, nematodes and mites) and macrofauna (earthworms, spiders, slaters,
198<br />
B.M. KUMAR<br />
centipedes, larvae, molluscs, etc.) fulfill a wide range of ecosystem services that<br />
underpin C sequestration and eventually the sustainability of the homegarden system<br />
(Table 2). However, as on many other aspects of belowground diversity, few data<br />
are available on the composition of soil biota or its determinants in the<br />
homegardens. This is partly because soil research in multistrata agroforestry systems<br />
poses methodological difficulties. Owing to variations in soil microenvironment,<br />
profound intra-garden variations in soil biotic activity are also probable.<br />
7. CARBON SEQUESTRATION PROGRAMS AND LIVELIHOOD<br />
SECURITY OF RURAL PEOPLE<br />
The Kyoto Protocol, the main instrument of the United Nations Framework<br />
Convention on Climate Change (UNFCCC), has set up the Clean Development<br />
Mechanism (CDM) concept as a cost-effective process to reduce rural poverty by<br />
extending payments to low-income farmers who provide carbon storage through<br />
land use systems 5 . Projects under the CDMs usually have the dual mandate of<br />
reducing greenhouse gas emissions and contributing to sustainable development.<br />
Implicit in this are, trade-offs between carbon sequestration, local social development,<br />
economic well-being and access to resources, and other aspects of<br />
environmental changes. Moreover, C storage through agroforestry is less costly<br />
(range $1–69/Mg C, median $13/Mg C) than through other CO2 mitigating options<br />
such as pure tree-based systems, carbon dioxide capture and storage or emission<br />
reduction (Albrecht and Kandji, 2003). It allows investors in developed countries to<br />
receive carbon credits in exchange for greenhouse gas emission reductions, whilst<br />
the developing countries where such investments are made, receive investments.<br />
There are many examples of how payment for environmental services to farmers can<br />
be made, while implementing mitigation projects (Brown et al., 2004; Montagnini<br />
and Nair, 2004). Carbon finance projects, thus, could transcend the existing barriers<br />
in resource mobilization for sustainable development of the developing countries.<br />
Although a number of such projects have been initiated as pilot activities around<br />
the globe, in alliance with non-governmental or development agencies, none of these<br />
as of date, are on tropical homegardens, implying that the potential of homegardens<br />
as a strategy for carbon sequestration has not yet been fully recognized, let alone<br />
exploited. Yet, the homegarden system offers considerable scope to improve biomass<br />
accumulation, and overcome “excess problems” (i.e., ameliorating “soil sickness”<br />
through mechanisms such as phytoremediation). Three pathways could be explored<br />
to promote externalities in agroforestry, in general, and homegarden systems, in<br />
particular. These are:<br />
• “Bringing more land under homegardens”: More land should be brought under<br />
agroforestry, resulting in more C sequestered in the landscape. There are already<br />
plenty of degraded lands available in most developing countries. For example, an<br />
estimated 1900 million ha of land is affected by soil degradation worldwide; of<br />
these, the largest area (around 747 million ha) is in the Asian region (van Lynden<br />
and Oldeman, 1997); India alone has an estimated 130 million ha of degraded<br />
lands 6 . The bottom line is that degraded sites could be molded into reasonably
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
199<br />
productive systems by appropriate policy and/or management interventions. For<br />
example, the indigenous Mayan groups have survived the extreme conditions by<br />
developing the multistrata homegardens over the karst topography, formed by<br />
limestone bedrock, and limited amounts of precipitation (Benjamin et al., 2001).<br />
• Intensification: More C can be sequestered per unit of land by improving<br />
efficiency of production through the choice of optimal species combinations and/or<br />
appropriate stand management practices, on which little scientific information<br />
exists, however. Moreover, restoring soil C triggers soil quality improvements<br />
(Lal, 2004). Multistrata stands and polycultures such as homegardens not only<br />
increase C sinks in soil and vegetation but also improve agricultural productivity<br />
and livelihood security, and are thought to be one step closer in the<br />
transformation of barren landscapes to “perpetually natural looking forests”—<br />
clearly a “win-win” situation (FAO, 2004).<br />
• Conservation: Ensuring long-term stability and sustainability—if such<br />
polycultures at least partially alleviate the anthropogenic pressure on natural<br />
forests— improves biodiversity conservation and reduce fossil fuel consumption.<br />
Many such traditional land use systems are, however, experiencing severe strains<br />
(Kumar and Nair, 2004), especially in the backdrop of technological changes;<br />
and to preserve them, appropriate land use policies/managerial interventions are<br />
needed.<br />
8. CONCLUSIONS<br />
Under the Kyoto Protocol, one clear strategy for mitigating the increase in<br />
atmospheric CO2 is to expand the size of the terrestrial C sink, using trees on<br />
agricultural lands as “biological scrubbers.” The magnitude of such C sequestration<br />
may, however, be dependent on the nature and extent of agroforestry system<br />
involved, and its structure and function, which in turn, are dependent on species<br />
composition and system management. Apparently, the homegardens have a special<br />
role in such abatement processes. Overall, they occupy the penultimate position in a<br />
tropical land use continuum ranging from annual crops to mature forests (Table 2).<br />
In particular, aspects such as higher biomass production potential and the return of a<br />
greater proportion of plant materials to the soil to increase its C stock compared to<br />
other agroforestry systems have been adequately demonstrated. In addition, they<br />
ensure “carbon permanence,” which the “carbon contracts” require, farmers to<br />
adopt; maintain sustainability and exploit the synergies between CBD and the Kyoto<br />
Protocol.<br />
One of the major constraints in employing homegardens to provide<br />
environmental benefits, however, is the lack of quantitative data on such potential<br />
advantages. Nevertheless, in view of the substantial coverage of homegardens in<br />
some geographical regions (e.g., south and southeast Asia) and especially if<br />
effective policies to promote such land use systems are implemented especially for<br />
degraded lands, they could become large carbon sinks; and the mitigation costs are<br />
probably lower than what is required for emission source controls. Indeed, the<br />
traditional knowledge has shown that the homegarden system is ideally suited for
200<br />
B.M. KUMAR<br />
regions characterized by highly weathered soils with relatively lower nutrient<br />
endowments as in the lateritic soils of Kerala and the karst deposits of Yucatán<br />
Peninsula. On a final note, science and natural resource policy should recognize the<br />
work of local people who still maintain agroecosystems with high agrobiodiversity<br />
as part of their culture, lifestyle, or practice.<br />
ENDNOTES<br />
1. Badan Pusat Statistik (BPS) 2000. Statistical yearbook of Indonesia 2000,<br />
Jakarta, 590p.<br />
2. Bangladesh Bureau of Statistics (BBS) 2001. Statistical yearbook 2000.<br />
Ministry of Planning, Government of People’s Republic of Bangladesh, Dhaka,<br />
452p.<br />
3. Department of Census and Statistics (DCS) 2003. Census of agriculture – Sri<br />
Lanka 2002. Agricultural holdings, extent under major crops and livestock<br />
statistics by district and DS/AGA division: based on operator’s residence—<br />
small holding sector. Preliminary Release– No. 2, P.O. Box 563, Colombo, 62p<br />
(www.statistics.gov.lk). Last accessed: November 2005.<br />
4. Kerala State Land Use Board (KSLUB) 1995. Land resources of Kerala State.<br />
Thiruvananthapuram, Kerala, 209p.<br />
5. Smith J. and Scherr S.J. 2002. Forest Carbon and local livelihoods: assessment<br />
of opportunities and policy recommendations. CIFOR Occasional Paper 37,<br />
Centre for International Forestry Research, Jakarta, 45p.<br />
6. Indian Council of Forestry Research and Education (ICFRE) 2000. Forestry<br />
statistics—2000. Directorate of Statistics, Indian Council of Forestry Research<br />
and Education, Dehra Dun, pp 55.<br />
REFERENCES<br />
Albrecht A. and Kandji S.T. 2003. Carbon sequestration in tropical agroforestry systems.<br />
Agric Ecosyst Environ 99: 15 – 27.<br />
Ball J.B., Wormald T.J. and Russo L. 1995. Experience with mixed and single species<br />
plantations. Commonwealth For Rev 74: 301 – 305.<br />
Beer J., Bonnemann A., Chavez W., Fassbender H.W., Imbach A.C. and Martel I. 1990.<br />
Modelling agroforestry systems of cacao (Theobroma cacao) with laurel (Cordia<br />
alliodora) or poro (Erythrina poeppigiana) in Costa Rica. V. Productivity indices, organic<br />
material models and sustainability over ten years. Agroforest Syst 12: 229 – 249.<br />
Benjamin T.J., Montanez P.I. Jimenex J.J.M. and Gillespie A.R. 2001. Carbon, water and<br />
nutrient flux in Maya homegardens in the Yucatan peninsula of Mexico. Agroforest Syst<br />
53: 103 – 111.<br />
Brown K., Adger W. N., Boyd E., Corbera-Elizalde E. and Shackley S. 2004. How do CDM<br />
projects contribute to sustainable development? Tyndall Centre Technical Report No. 16.<br />
University of East Anglia, Norwich, UK, 54p. http://www.tyndall.ac.uk/publications/<br />
tech_reports/tech_reports.shtml (last accessed: December 2005).<br />
Christanty L. 1990. <strong>Homegardens</strong> in tropical Asia with special reference to Indonesia. In<br />
Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp. 9 – 20. United Nations<br />
University Press, Tokyo.
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
201<br />
Cuevas E. and Medina E. 1986. Nutrient dynamics within Amazonian forests. 1. Nutrient flux<br />
in fine litterfall and efficiency of nutrient utilization. Oecologia 68: 466 – 472.<br />
de Jong B.H.J. 2001. Uncertainties in estimating the potential for carbon mitigation of forest<br />
management. For Ecol Manag 154: 85 – 104.<br />
Divakara B.N., Kumar B.M., Balachandran P.V. and Kamalamm N.V. 2001. Bamboo hedgerow<br />
systems in Kerala, India: root distribution and competition with trees for phosphorus.<br />
Agroforest Syst 51: 189 – 200.<br />
Duguma B., Gockowski J. and Bakala J. 2001. Smallholder cacao (Theobroma cacao Linn.)<br />
cultivation in agroforestry systems of West and Central Africa: challenges and<br />
opportunities. Agroforest Syst 51: 177 – 188.<br />
FAO. 2004. Assessing carbon stocks and modelling win–win scenarios of carbon<br />
sequestration through land use changes. Food and Agriculture Organization of the United<br />
Nations, Rome, 156p.<br />
FAO. 1992. Mixed and pure forest plantations in the tropics and sub-tropics. FAO Forestry<br />
Paper 103, Food and Agriculture Organization of the United Nations, Rome, 152p.<br />
Gajaseni J. and Gajaseni N. 1999. Ecological rationalities of the traditional homegarden<br />
system in the Chao Phraya Basin, Thailand. Agroforest Syst 46: 3 – 23.<br />
Gathumbi S.M., Cadisch G. and Giller K.E. 2004. Improved fallows: effects of species<br />
interaction on growth and productivity in monoculture and mixed stands. For Ecol Manag<br />
187: 267 – 280.<br />
Gathumbi S.M., Ndufa J.K., Giller K.E. and Cadisch G. 2002. Do species mixtures increase<br />
above- and belowground resource capturing in woody and herbaceous tropical legumes?<br />
Agron J 94: 518 – 526.<br />
Hättenschwiler S., Tiunov A.V. and Scheu S. 2005. Biodiversity and litter decomposition in<br />
terrestrial ecosystems. Ann Rev Ecol Evol Syst 36: 191 – 218.<br />
Houghton J.T. 1995. Determining emissions of carbon from land: a global strategy. In: Murai<br />
S. (ed.), Toward global planning of sustainable use of the earth, pp 59 – 76. Elsevier,<br />
Amsterdam.<br />
Houghton R.A. and Goodale C.L. 2004. Effects of land use change on the carbon balance of<br />
terrestrial ecosystems. Ecosystems and land use change. Geophys Monogr Ser 153:<br />
85 – 98.<br />
Jactel H., Brockerhoff E. and Duelli P. 2005. A test of the biodiversity–stability theory: metaanalysis<br />
of tree species diversity effects on insect pest infestations, and re-examination of<br />
responsible factors. In: Scherer-Lorenzen M., Körner Ch., and Schulze E.-D. (eds), Forest<br />
diversity and function: Temperate and Boreal Systems. Ecological Studies, Vol. 176. pp<br />
235 – 262. Springer-Verlag, Berlin, Heidelberg.<br />
Jamaludheen V. and Kumar B.M. 1999. Litter of nine multipurpose trees in Kerala, Indiavariations<br />
in the amount, quality, decay rates and release of nutrients. For Ecol Manag<br />
115: 1 – 11.<br />
Jensen M. 1993. Soil conditions, vegetation structure and biomass of a Javanese homegarden.<br />
Agroforest Syst 24: 171 – 186.<br />
Kaye J.P., Resh C.S., Kaye M.W. and Chimner R.A. 2000. Nutrient and carbon dynamics in a<br />
replacement series of Eucalyptus and Albizia trees. Ecology 81: 3267 – 3273.<br />
Keenan R., Lamb D. and Sexton G. 1995. Experience with mixed species rainforest<br />
plantations in North Queensland. Commonwealth For Rev 74: 315 – 321.<br />
Körner C. 2003. Carbon limitation in trees. J Ecol 91: 4 – 17.<br />
Kraenzel M., Castillo A., Moore, T. and Potvin C. 2003. Carbon storage of harvest –age teak<br />
(Tectona grandis) plantations, Panama. For Ecol Manag 173: 213 – 225.<br />
Kremen C., Williams N.M. and Thorp R.W. 2002. Crop pollination from native bees at risk<br />
from agricultural intensification. Proc Natl Acad Sci 99: 16812 – 16816.
202<br />
Kumar B.M. and Divakara B.N. 2001. Proximity, clump size and root distribution pattern in<br />
bamboo: A case study of Bambusa arundinacea (Retz.) Willd., Poaceae, in the Ultisols of<br />
Kerala, India. J Bamboo Rattan 1: 43 – 58.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Kumar B.M, George S.J. and Chinnamani S. 1994. Diversity, structure, and standing stock of<br />
wood in the homegardens of Kerala in peninsular India. Agroforest Syst 25: 243 – 262.<br />
Kumar B.M., George S.J., Jamaludheen V. and Suresh T.K. 1998. Comparison of biomass<br />
production, tree allometry and nutrient use efficiency of multipurpose trees grown in<br />
wood lot and silvopastoral experiments in Kerala, India. For Ecol Manag 112: 145 – 163.<br />
Kumar B.M., Haibara K. and Toda H. 2005. Does plant litter become more recalcitrant under<br />
elevated atmospheric CO2 levels? Global Environ Res 9: 83-91.<br />
Kumar B.M., Thomas J. and Fisher R.F. 2001. Ailanthus triphysa at different density and<br />
fertilizer levels in Kerala, India: tree growth, light transmittance, and understorey ginger<br />
yield. Agroforest Syst 52: 133 – 144.<br />
Kürsten E. 2000. Fuelwood production in agroforestry systems for sustainable land use and<br />
CO2 mitigation. Ecol Eng 16: 69 – 72.<br />
Lal R. 2004. Soil carbon sequestration impacts on global change and food security. Science<br />
304: 1623 – 1627.<br />
Lal R., Kimble J.M., Follett R.F. and Stewart B.A. 1998. Soil Processes and the Carbon<br />
Cycle. CRC Press LLC, MA, 609p.<br />
Luo Y., Su Bo, Currie W.S., Dukes J.S., Finzi A., Hartwig U., Hungate B., McMurtrie R.E.,<br />
Oren R., Parton W.J., Pataki D.E., Shaw, M.R., Zak D.R. and Field C.B. 2004.<br />
Progressive nitrogen limitation of ecosystem responses to rising atmospheric carbon<br />
dioxide. BioScience 54: 731 – 739.<br />
McCann K.S. 2000. The diversity-stability debate. Nature 405: 228 – 233.<br />
Mingkui C. and Woodard F.I. 1998. Dynamic responses of terrestrial ecosystem carbon<br />
cycling to global climatic change. Nature 393: 249 – 252.<br />
Montagnini F. 2006. Status of homegardens in Mesoamerica. In: Kumar B.M. and Nair<br />
P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry,<br />
pp 61 – 84. Springer Science, Dordrecht.<br />
B.M. KUMAR<br />
Montagnini F. and Nair P.K.R. 2004 Carbon sequestration: an underexploited environmental<br />
benefit of agroforestry systems. Agroforest Syst 61: 281 – 295.<br />
Montagnini F., Gonzalez E., Porras C. and Rheingans R. 1995. Mixed and pure forest<br />
plantations in the humid neotropics: a comparison of early growth, pest damage, and<br />
establishment costs. Commonwealth For Rev 74: 306 – 314.<br />
Nair P.K.R. and Kumar B.M. 2006. Introduction. In: Kumar B.M. and Nair P.K.R. (eds),<br />
<strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp 1 – 10.<br />
Springer Science, Dordrecht.<br />
Nair P.K.R. and Nair V.D. 2003. Carbon storage in North American agroforestry systems. In:<br />
Kimble J., Heath L.S., Birdsey R.A., and Lal R. (eds), The potential of U.S. forest soils to<br />
sequester carbon and mitigate the greenhouse effect, pp 333 – 346. CRC Press, Boca<br />
Raton, FL.<br />
Parrotta J.A. 1999. Productivity, nutrient cycling and succession in single- and mixed-species<br />
stands of Casuarina equisetifolia, Eucalyptus robusta and Leucaena leucocephala in<br />
Puerto Rico. For Ecol Manag 124: 45 – 77.<br />
Reich P.B., Knops J., Tilman D., Craine J., Ellsworth D. Tjoelker M., Lee T., Wedin D.,<br />
Naem S., Bahauddin D., Hendrey G., Jose S., Wrage K., Goth J. and Bengston W. 2001.<br />
Plant diversity enhances ecosystem responses to elevated CO2 and nitrogen deposition.<br />
Nature 410: 809 – 812.
CARBON C SEQUESTRATION<br />
S POTENTIAL OF HOMEGARDENS<br />
203<br />
Roshetko M., Delaney M., Hairiah K. and Purnomosidhi P. 2002. Carbon stocks in<br />
Indonesian homegarden systems: Can smallholder systems be targeted for increased<br />
carbon storage? Am J Alt Agr 17: 125 – 137.<br />
Roy C. 1999. Options techniques et socio-économiques des émissions de CO 2<br />
et d’augmentation des stocks de carbone. CR Acad Agric. France 85: 311 – 320.<br />
Ruark G.A., Schoeneberger M.M. and Nair P.K.R. 2003. Agroforestry–Helping to Achieve<br />
Sustainable Forest Management. UNFF (United Nations Forum for Forests) Intersessional<br />
Experts Meeting on the Role of Planted Forests in Sustainable Forest Management, New<br />
Zealand (24 – 30 March 2003). www.maf.govt.nz/unff-planted-forestry-meeting (last<br />
accessed: July 2005).<br />
Russell A.E. 2002. Relationships between crop-species diversity and soil characteristics in<br />
southwest Indian agroecosystems Agric Ecosyst Environ 92: 235 – 249.<br />
Russell A.E., Cambardella C.A., Ewel J.J. and Parkin T.B. 2004. Species, rotation, and life<br />
form diversity effects on soil carbon in experimental tropical ecosystems. Ecol Appl 14:<br />
47 – 60.<br />
Sanchez P.A. 2000. Linking climate change research with food security and poverty reduction<br />
in the tropics. Agric Ecosyst Environ 82: 371 – 383.<br />
Sanford R.L. and Cuevas E. 1996. Root growth and rhizosphere interactions in tropical<br />
forests. In: Mulkey S., Chazdon R.L., and Smith A.P. (eds), <strong>Tropical</strong> forest plant<br />
ecophysiology, pp 268 – 300. Chapman and Hall, New York.<br />
Schimel D., Melillo J., Tian H., McGuire A.D., Kicklighter D., Kittel T., Rosenbloom N.,<br />
Running S., Thornton P., Ojima D., Parton W., Kelly R., Sykes M., Neilson R. and Rizzo<br />
B. 2000. Contribution of increasing CO 2 and climate to carbon storage by ecosystems in<br />
the United States. Science 287: 2004 – 2006.<br />
Schroeder P. 1994. Carbon storage benefits of agroforestry systems. Agroforest Syst 27:<br />
89 – 97.<br />
Singh G., Babu R., Narain P., Bhushan L.S. and Abrol I.P. 1992. Soil erosion rates in India.<br />
J Soil Water Conserv 47 (1): 97 – 99.<br />
Swift M.J., Izac A.-M.N. and van Noordwijk M. 2004. Biodiversity and ecosystem services in<br />
agricultural landscapes—are we asking the right questions? Agric Ecosyst Environ 104:<br />
113 – 134.<br />
Tilman D., Lehman C.L. and Thomson K.T. 1997. Plant diversity and ecosystem<br />
productivity: theoretical considerations. Proc Natl Acad Sci USA 94: 1857 – 1861.<br />
Tomich T.P., de Foresta H., Dennis R., Ketterings Q., Murdiyarso D., Palm C., Stolle F., and<br />
van Noordwijk M. 2002. Carbon offsets for conservation and development in Indonesia?<br />
Am Alt Agr 17: 125 – 137.<br />
UNFCCC 2002. Activities implemented jointly under the pilot phase. Sixth synthesis report.<br />
Subsidiary Body for Scientific and Technological Advice. FCCC/SBSTA/2002/8. United<br />
Nations Framework Convention on Climate Change, Bonn 5p (http://unfccc.int/resource/<br />
docs/2002/sbsta/08. pdf; last accessed: December 2005).<br />
van Lynden G.W.J. and Oldeman L.R. 1997. The Assessment of the Status of Human-<br />
Induced Soil Degradation in South and Southeast Asia. International Soil Reference and<br />
Information Centre, Wageningen, 35p.<br />
van Noordwijk M., Rahayu S., Hairiah K., Wulan Y.C., Farida A. and Verbist B. 2002.<br />
Carbon stock assessment for a forest-to- coffee conversion landscape in Sumber-Jaya<br />
(Lampung, Indonesia): from allometric equations to land use change analysis. Science in<br />
China Series C-Life Sciences 45: 75 – 86 Suppl. S Oct 2002. Science in China Press,<br />
Beijing.<br />
Vandermeer J. 1989. The Ecology of Intercropping. Cambridge University Press, Cambridge,<br />
249p.
204<br />
B.M. KUMAR<br />
Vinod V.R., Syed Anwarulla M. and Vishwanth D.P. 2003. Run off and soil loss under<br />
different land use systems in the Western Ghats of Karnataka. Indian J Soil Conserv 31:<br />
131 – 138.<br />
Vohland K. and Schroth G. 1999. Distribution patterns of the litter macrofauna in agroforestry<br />
and monoculture plantations in central Amazonia as affected by plant species and<br />
management. Applied Soil Ecol 13: 57 – 68.<br />
Watson R.T., Noble I.R., Bolin B., Ravindranath N.H., Verardo D.J. and Dokken D.J. (eds).<br />
2000. Intergovernmental Panel on Climate Change (IPCC). Land use, land use change,<br />
and forestry. A special report of the Intergovernmental Panel on Climate Change.<br />
Cambridge University Press, 377p.<br />
Wedin D. and Tilman D. 1993. Competition among grasses along a nitrogen gradient: initial<br />
conditions and mechanisms of competition. Ecol Monogr 63: 199 – 229.
CHAPTER 12<br />
MEDICINAL PLANTS IN TROPICAL<br />
HOMEGARDENS<br />
M.R. RAO 1 AND B.R. RAJESWARA RAO 2<br />
1 Former ICRAF Scientist; Current address: Plot No. 11, ICRISAT Colony (Phase-<br />
I), Brig. Syed Road, Manovikasnagar (P.O.), Secunderabad 500 009, India; E-mail:<br />
. 2 Central Institute of Medicinal and Aromatic Plants<br />
(CIMAP) Resource Centre, Boduppal, Uppal P.O., Hyderabad 500 039, India<br />
Keywords: Aromatic plants, Bioprospecting, Indigenous knowledge, Phytochemicals,<br />
Traditional medicine, Value addition.<br />
Abstract. Nearly 80% of the people living in developing countries depend on medicinal<br />
plants (MPs) for primary healthcare, and homegardens are an important source of production<br />
of these plants. <strong>Homegardens</strong> can fulfill the dual role of production and in situ conservation of<br />
MPs to overcome their dwindling supplies and threat of extinction from natural sources. MPs<br />
in homegardens are either deliberately cultivated or they come up spontaneously. They are an<br />
important constituent of homegardens, next only to food crops and fruit trees; yet their<br />
economic value is not fully recognized, let alone exploited. <strong>Homegardens</strong> offer an<br />
economically and socially viable option for large-scale production of phytochemicals from<br />
important MPs under organic cultivation. Promoting organic production of selected<br />
commercially valuable species of MPs through homegardening can, thus, augment the<br />
farmers’ income, enhance rural employment opportunities, and help reduce migration of rural<br />
youth to urban centers in search of jobs. Research is needed to improve the existing<br />
germplasm, introduce suitable commercial MPs in different agroecosystems, and develop<br />
cultivation and processing techniques to increase yield and improve product quality, and<br />
exploit indigenous knowledge and market opportunities.<br />
1. INTRODUCTION<br />
Humans depended on certain plants for healthcare since time immemorial. Centuries<br />
of experimentation on the use of plants or products derived from them has led to the<br />
development of indigenous systems of medicine that are still respected and used in<br />
many societies. Plants have been a source of medicines for humans and livestock<br />
and pesticides to protect crops from certain pests and diseases. In India, over 200<br />
types of vegetable drugs were in use during the Vedic period (3700 – 2000 BC).<br />
Charak Samhita (600 BC) mentioned 1270 medicinal plants (MPs), while Sushruta<br />
205<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 205–232.<br />
© 2006 Springer. Printed in the Netherlands.
206 M.R. RRAO AND B.R. RRAJESWARA RAO R<br />
Samhita (450 BC) and Vagbhatta’s Astangahridaya (342 BC) mention about 1100<br />
and 1150 MPs, respectively (Chadha and Gupta, 1995). America, Arabia, China,<br />
Egypt, Greece, Mexico, and many other countries in Europe and Asia too recorded<br />
the use of MPs (Principe, 1991). Furthermore, about 1800 species of MPs are<br />
reported to be used in the traditional Indian medical system of Ayurveda, 750<br />
species in Unani or Tib, 500 species in Siddha, 400 species in the Tibetan medicine<br />
and 5000 species in the Chinese medicine. Traditional medical systems in Japan,<br />
Korea (Kampo system), Indonesia (Jamu system), South Africa (Julu system),<br />
Bhutan (Gso-ba-rig-pa), Sri Lanka (Deshiya Chikitsa), and Malaysia (Malay herbal<br />
medicine) also recorded a number of MPs and their uses (Principe, 1991).<br />
An estimated 14 to 28% of the 422 000 plants occurring on earth had been used<br />
by human cultures for medicinal purposes at one time or another (Farnsworth and<br />
Soejarto, 1991). Approximately 80% of the people in developing countries rely even<br />
today mainly on traditional medicines for humans (FAO, 1996) as well as domestic<br />
animals, a major portion of which are extracts of medicinal plants or their active<br />
principles. More than 6500 species of such medicinal plants have been identified in<br />
Asia, 1900 species in tropical America and 1300 species in north-west Amazon<br />
(Farnsworth and Soejarto, 1991). Global trade in plant-based drugs was estimated at<br />
US$ 100 billion, of which traditional medicines using medicinal plants accounted<br />
for 60 billion (WHO, 2004). In addition, trade 1 in herbal teas, drug adjuncts, dietary<br />
foods etc. (sold over the counter) was estimated at US$ 5 billion in 1997. India has<br />
approximately 150 000 practitioners of traditional systems of medicine, 10 000<br />
licensed pharmacies manufacturing plant-based drugs. The trade in medicinal herbs<br />
in India was estimated at US$ 1 billion (EXIM Bank, 2003) and the country exports<br />
medicinal herbs worth US$ 287 million annually 2 .<br />
Most of the medicinal plants (70 to 90%) have traditionally been collected from<br />
forests and natural habitats. Indiscriminate extraction over years not only reduced<br />
their supplies but also endangered some of these valuable species. The growing<br />
demand for plant-derived drugs both in modern and traditional systems of medicine 3<br />
further exacerbated the problem in many natural habitats. This has led to the<br />
extinction of about 75 species between 1600 and 1900 and a similar number in a<br />
short span between 1900 and 1970 (Principe, 1991; Rao, 1999). It is feared that if<br />
this trend continues, about 60 000 species will become extinct in the next century<br />
(Principe, 1991). Considering the economic importance of medicinal plants, there is<br />
an urgent need to systematically cultivate them to exploit their full potential and to<br />
save them from extinction. MPs can be cultivated like any other crop(s) in different<br />
systems including agroforestry – in forest plantations, homegardens, as intercrops<br />
between trees, and as components of multistrata systems (Rao et al., 2004). This<br />
chapter reviews the status of medicinal plants in tropical homegardens and examines<br />
the scope for improving their relative contribution to the economy of rural families.<br />
2. MEDICINAL PLANTS IN HOMEGARDENS<br />
<strong>Homegardens</strong> being one of the earliest forms of agroforestry practiced in the tropics<br />
(Kumar and Nair, 2004), it is only logical to be expected that MPs have been an<br />
essential component of these production systems (Tables 1 and 2). Indeed, the
MEDICINAL M PLANTS IN HOMEGARDENS<br />
207<br />
homegardens make a substantial contribution to the supply of MPs, which may be<br />
traded or consumed locally by the family or community (Albuquerique and<br />
Andrade, 2002). There is, however, no reliable data on the extent of homegardens in<br />
different countries (see Nair and Kumar, 2006), yields of medicinal plants, or<br />
products extracted and sold at national and international levels. Majority of MPs in<br />
homegardens are herbs/vines/climbers and they together with vegetables and spices<br />
generally constitute the lower layer (0 – 1 m), unless they are vines and climbers.<br />
Additionally, a number of homegarden shrub and tree species also have medicinal<br />
value and they constitute the second (1 – 3 m) and upper (>10 m) layers respectively<br />
(Wezel and Bender, 2003). Some species that grow spontaneously in homegardens<br />
may possess medicinal value which may or may not be recognized and used. For<br />
example, in Chiriqui, Panama, the Ngöbe community utilizes the land fallowed for<br />
soil fertility replenishment as a source of MPs (Samaniego and Lok, 1998). Nearly<br />
half of the 41 weed species found in the homegardens of Central Sulawesi,<br />
Indonesia, possess medicinal value (Kehlenbeck and Maass, 2005). In India,<br />
seasonal weeds such as Phyllanthus amarus, Boerhaavia diffusa, Achyranthus<br />
aspera, Tribulus terrestris, Sida cordifolia, and Aerva lanata that occur both in<br />
cultivated fields (including homegardens) and wild are collected for medicinal<br />
purposes (Rao et al., 1999).<br />
2.1. Relative importance of MPs in homegardens<br />
While some components in the homegardens have exclusive medicinal value, others<br />
are multipurpose species combining medicinal value with food, ornamental, fiber,<br />
and spice values. For example, in the Kandyan homegardens of Sri Lanka, 30% of<br />
the total 125 species found were exclusively mentioned for medicinal uses and 12%<br />
combined medicinal with other uses. Among the medicinal species, trees constituted<br />
7%, shrubs 5%, herbs 15%, and creepers 3% of the total species (Perera and<br />
Rajapakse, 1991). <strong>Homegardens</strong> in Bukoba district in northwestern Tanzania<br />
contained species that were said to be used exclusively for medicine (Baphiopsis<br />
spp., Cyperus dives, Leonotis nepetifolia, Vernonia amygdalina, and Solanum<br />
incanum), those that combined medicine and fuelwood (Senecio multicorymbosa<br />
tree for medicines to cattle), medicine, fruit, and fuelwood (Psidium guajava and<br />
Citrus limon), and propping poles and medicine (Ricinus communis; Rugalema<br />
et al., 1994). The Chagga homegardens on Mt. Kilimanjaro in Tanzania were<br />
dominated by woody components; nearly 50% of the 111 species found in the region<br />
were trees, of which 30% were mentioned as medicines for humans and livestock<br />
(O’Kting’ati et al., 1984). Of the 77 useful plants (shrubs, vines, and forbs) found<br />
across 80 traditional Mayan homegardens in Quitana Roo, Mexico, nine were<br />
reported to have exclusive medicinal value and 26 species combined medicine, food,<br />
spice, and ornamental values (De Clerck and Negreros-Castillo, 2000). About 70%<br />
of 301 species in the forest and homegardens in the Yucatan, Mexico were classified<br />
for medicinal purpose; however, only 16 species were exclusively used for medicine<br />
and the rest had multiple uses (Rico-Gray et al., 1991).
Table 1. Spices, condiments, and aromatic plants possessing medicinal value grown in tropical homegardens.<br />
Species Family Part(s) used Uses (for treatment of diseases/other<br />
applications mentioned)<br />
Trees<br />
Cinnamomum Lauraceae bark diarrhea, gastric debility, flatulence, nausea,<br />
zeylanicum (cinnamom)<br />
vomiting, herbal tea<br />
Citrus aurantifolia Rutaceae fruit source of vitamin C, cataract, bleeding gum,<br />
(lime)<br />
herbal tea, smallpox<br />
Syzygium aromaticum Myrtacae flower buds carminative, antispasmodic, galacto purifier,<br />
(clove)<br />
antibacterial, appetizer, rubifacient<br />
Myristica fragrans Myristicaceae seeds, aril dyspepsia, diarrhea, hepatopathy, impotency,<br />
(nutmeg)<br />
insomnia, cardiac disorders<br />
Murraya koenigii Rutaceae leaves carminative, skin diseases, anorexia,<br />
(curry leaf)<br />
dyspepsia, flatulence, hair tonic, stomach<br />
ache<br />
Tamarindus indica Caesalpiniaceae root, leaves, fruit jaundice, scabies, smallpox, alcoholic<br />
(tamarind)<br />
pulp, seed intoxication, carminative, refrigerant<br />
Shrubs/herbs/grasses<br />
Allium sativum (garlic) Liliaceae bulb antiperiodic, antibacterial, diuretic, skin<br />
diseases<br />
Where grown?<br />
Sri Lanka, Indonesia,<br />
Madagascar, Brazil,<br />
Seychelles<br />
Many countries in<br />
tropics<br />
Southeast Asia , Sri<br />
Lanka, Tanzania, Brazil<br />
Southeast Asia, Sri<br />
Lanka, West Indies<br />
India<br />
South Asia, East, and<br />
West Africa<br />
throughout tropics<br />
208 M.R. RRAO AND B.R. RR RAJESWARA RAO R
Allium cepa (onion) Liliaceae bulb pulmonary phthisis, whooping cough, colic<br />
dyspepsia, reduces cholesterol<br />
Capsicum annuum Solanaceae fruits gout, arthritis, dyspepsia, hoarseness,<br />
flatulence<br />
Cymbopogon flexuosus, Poaceae leaves source of vitamin A, leprosy, epilepsy,<br />
C. citratus (lemongrass)<br />
mosquito repellent, herbal tea<br />
Coriandrum sativum<br />
(coriander)<br />
Cuminum cyminum<br />
(cumin)<br />
Curcuma longa (C.<br />
domestica)<br />
(turmeric)<br />
Elettaria cardamomum<br />
(small cardamom)<br />
Kaempferia galanga<br />
(candramula)<br />
South and Southeast Asia,<br />
Spain, Brazil, Egypt<br />
throughout tropics<br />
Mexico, Brazil, China,<br />
Haiti, South and<br />
Southeast Asia, Africa<br />
South and Southeast<br />
Asia<br />
Apiaceae fruit, leaf colic, laxative, blood purifier, indigestion,<br />
sour throat<br />
Apiaceae seed dyspepsia, flatulence, diarrhea, skin diseases East Asia, India<br />
Zingiberaceae rhizome antiseptic, skin allergies, viral hepatitis, antibacterial,<br />
wounds, anti-inflammatory, soar<br />
throat<br />
Zingiberaceae fruit nausea, indigestion, abdominal pains,<br />
Zingiberaceae rhizomes, rootstock.<br />
leaves<br />
bronchitis, respiratory infections<br />
digestive, vulnerary, anthelminthic,<br />
dyspepsia, leprosy, skin diseases,<br />
rheumatism, asthma, bronchitis, malaria,<br />
urolithiasis<br />
South, Southeast, and<br />
East Asia<br />
South and Southeast<br />
Asia<br />
India<br />
Table 1 (cont.)<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
209
Species Family Part(s) used Uses(for treatment of diseases/other Where grown?<br />
applications mentioned)<br />
Mentha arvensis (mint) Labiatae leaves cough syrups, flavoring agent, expectorant, Mexico<br />
pain reliever<br />
Mentha piperita (mint) Labiatae leaves flavoring agent Mexico<br />
Piper betle (betel vine) Piperaceae leaves antiseptic, aphrodisiac, expectorant, South and Southeast<br />
bronchitis, rheumatism, stimulant,<br />
Asia<br />
carminative, wounds<br />
Piper nigrum<br />
Piperaceae dried berries indigestion, chronic rheumatism, asthma, Asia, Africa, Brazil<br />
(black pepper)<br />
cough, throat complaints<br />
Trigonella foenum- Fabaceae seeds anti-diabetic, flatulence, carminative, India, Middle East,<br />
graecum (fenugreek)<br />
emollient, galactagogue<br />
Egypt, Morocco<br />
Zingiber officinale Zingiberaceae rhizome asthma, skin diseases, de-worming, nausea, South and Southeast<br />
(ginger)<br />
carminative, common colds<br />
Asia, China, Nigeria<br />
Source: Padoch and De Jong (1991), Perera and Rajapakse (1991), Rugalema et al. (1994), Lamont et al. (1999), Rao et al. . (1999), De e Clerck<br />
and Negreros-Castillo (2000), Millat-e-Mustafa et al. (2002), and Wezel and Bender (2003).<br />
210 M.R. RRAO AND B.R. RR RAJESWARA RAO R
MEDICINAL M PLANTS IN HOMEGARDENS<br />
211<br />
Many of the economic species grown in homegardens possess complementary<br />
medicinal values. Such species may or may not be exploited commercially for their<br />
medicinal properties but are used locally within the family and community. For<br />
example, people in southeastern Nigeria uses a number of species that they grow in<br />
their compound farms—for purposes other than healthcare, for medicinal purposes<br />
(Okafor and Fernandes, 1987). Such species include Cajanus cajan (leaves for<br />
treating measles), Carica papaya (leaves for treating malaria), Cola lipidota/<br />
C. nitida/C. pachycarpa (stimulant), Kigelia africana (bark for treating sores),<br />
Jatropha curcas (leaves for ringworm treatment), Neubouldia laevis (stem and roots<br />
medicinal), and Invingia gabonensis var. gabonensis (leaves and bark medicinal).<br />
Similarly, many plants are collected for medicinal uses from multistoried<br />
agroforestry systems in west Sumatra (Indonesia), although none was grown in the<br />
system consciously for that purpose (Michon et al., 1986). Majority of spices, a<br />
number of vegetables and ornamentals grown in homegardens also have medicinal<br />
uses (Table 1). The homegardens in Java and Sumatra were reported to contain 26<br />
medicinal species and a similar number of spices (Kubota et al., 1992).<br />
Agelet et al. (2000) made a detailed analysis of medicinal plants found in 155<br />
homegardens in the mountain zones of Catalonia (north-eastern Iberian Peninsula,<br />
Spain). The gardens contained nine distinct categories of species: plants exclusively<br />
cultivated for medicinal purpose (23) mostly close to the house, the medicinal wild<br />
plants favored by homegarden structure and care (105), and seven kinds of<br />
horticultural plants with complementary medicinal values (117). There was,<br />
however, loss of about 56 taxa or 23% of the total over the years.<br />
Despite the presence of many medicinal species in homegardens, only a few<br />
species stand out as economically important in any given region. The most<br />
frequently found species in 31 homegardens in three villages in Cuba, were<br />
Jatropha gossypiifolia, Senna occidentalis, Xanthoxylum pistacifolium, Pluchea<br />
odorata, and Rhoeo spathacea (Wezel and Bender, 2003). Common among species<br />
expressly cultivated for medicinal purpose in Catalan homegardens were Tanacetum<br />
parthenium – a plant used for intestinal antiseptic – and Liliun candidum for<br />
vulnerary use (Agelet et al., 2000). In the state of Kerala (India), Kaempferia<br />
galanga – which has been traditionally collected from forests, is now being<br />
commercially cultivated in the homegardens (Kumar et al., 2005) and as intercrop in<br />
orchard crops (Maheswarappa et al., 1998). Tribals living in the Eastern Ghats of<br />
Andhra Pradesh (India) have been growing Piper longum and Curcuma angustifolia<br />
extensively for medicinal purposes along with turmeric (Curcuma longa) using<br />
Jatropha curcas as a bio-fence in homegardens (K.P. Sastry, CIMAP Resource<br />
Centre, Hyderabad, pers. comm., July 2005). In the ‘Dai homegardens’ of<br />
Xishuangbanna province in China, the prominent medicinal species found were<br />
Acanthopanax trifoliatus, Toona sinensis, Sapindus rarak, Tamarindus indica,<br />
Bryophyllum pinnatum, Euphorbia antiquorum, and Prunus persica (Saint-Pierre,<br />
1991). Ammomum villosum, which requires about 70% shade, is planted under forest<br />
cover after clearing the undergrowth and it yields 30 to 150 kg rhizomes ha –1 year –1<br />
depending on water resource availability. <strong>Homegardens</strong> even in an isolated Soqotra<br />
island in the Republic of Yemen despite containing on average 3.9 to 8.4 species per<br />
garden included medicinal plants such as Aloe perryi, Jatropha unicostata, and
212<br />
Commiphora ornifolia (Ceccolini, 2002). This should indicate the importance given<br />
to MPs by rural people in the tropics.<br />
Table 2. Relative importance of medicinal species in relation to total species in tropical<br />
homegardens.<br />
Region/location <strong>Homegardens</strong><br />
examined<br />
(no.)<br />
M.R. RRAO AND B.R. RRAJESWARA RRAO<br />
Total and<br />
medicinal a<br />
species<br />
across<br />
gardens<br />
Total and<br />
medicinal a<br />
species per<br />
garden<br />
Reference<br />
Santa Rosa, Peruvian 21 168 (46) 18 to 74 Padoch and de Jong<br />
Amazon<br />
(9.7) (1991)<br />
Bukoba, North- 72 57 (10) N/A Rugalema et al.<br />
western Tanzania<br />
(1994)<br />
Amazon,<br />
Northeastern Peru<br />
51 161 (56) N/A (9.5) Lamont et al. (1999)<br />
Catalonia, Iberian 145 N/A (250) N/A (30 to Agelet et al. (2000)<br />
Peninsula, Spain<br />
60)<br />
Congo (Zaire) N/A 273 (74) N/A Mpoyi et al. (1994)<br />
Masatepe, Nicaragua 1 98 (10) N/A (10) Viquez et al. (1994)<br />
Floodplain Jamuna 17 125 (48) N/A Yoshino and Ando<br />
tributary,<br />
Bangladesh<br />
(1999)<br />
Dhamrai, Bangladesh 243 N/A (71) N/A Millat-e-Mustafa<br />
et al. (2001)<br />
Deltaic, dry land, 200 120 (31) N/A Millat-e-Mustafa<br />
hilly, and plain<br />
regions,<br />
Bangladesh<br />
et al. (2002)<br />
Eastern Cuba 31 101 (39) 18 to 24 (4) Wezel and Bender<br />
(2003)<br />
Tixcacaltuyub and N/A 301 (152) N/A Rico-Gray et al.<br />
Tixpeual, Mexico<br />
(1991)<br />
Kerala, India 252 127 (25) 3 to 25 Kumar et al. (1994)<br />
Kandy, Sri Lanka 50 125 (52) 37 to 65 Perera and<br />
Rajapakse (1991)<br />
Central Sulavesi,<br />
30 149 28 to 37 Kehlenbeck and<br />
Indonesia<br />
(2.8) Maass (2005)<br />
a Values in parentheses refer to medicinal species; N/A = information not available.<br />
Immigrants from Southeast Asia to USA continued the tradition of growing<br />
many species in homegardens wherever they settled – for family use as well as for<br />
sale in the Asian markets. A survey of 59 gardens of Laotian Hmong settlers in the<br />
central Sacramento Valley, California, USA, revealed 59 taxa of which 38 had food
213<br />
value, 36 had medicinal value and a few others had uses like fiber and ornamental.<br />
Nineteen taxa had exclusive medicinal value, 15 combined food and medicine, and<br />
one or two combined medicinal, with ornamental or fiber uses. Many species that<br />
are categorized as being used for both food and medicine were primarily used for<br />
food seasoning or as additives (Corlett et al., 2003).<br />
2.2. Diversity of MPs in homegardens<br />
The species diversity including medicinal species in homegardens primarily depends<br />
on climate, altitude, socioeconomic and cultural factors, and nearness to markets.<br />
The diversity and density of plants generally increase with rainfall and elevation. In<br />
Venezuela, high diversity was positively correlated with age and remoteness of the<br />
garden, its use for subsistence, age of the farmer, and extent of participation of<br />
family labor in the activities of the garden (Mulas et al., 2004). In Bangladesh,<br />
species number decreased with increase in homegarden size and from deltaic region<br />
to dry region (Millat-e-Mustafa et al., 2002). <strong>Homegardens</strong> in West Java, Indonesia,<br />
contained the greatest diversity with an average number of 56 species per garden,<br />
the number of species being more in the wet season than in the dry season<br />
(Soemarwoto, 1987). In contrast, species composition of Cuban gardens differed<br />
across sites, especially in terms of medicinal plants, with gardens in the semiarid<br />
climate showing greater range than those in the humid region (Wezel and Bender,<br />
2003). Medicinal plants were recognized as the second most important group next<br />
only to cash value species in Sri Lanka (Perera and Rajapakse, 1991) and<br />
Bangladesh (Millat-e-Mustafa et al., 2002), food crops in Peruvian Amazon (Padoch<br />
and de Jong, 1991) and fruits in Cuba (Wezel and Bender, 2003) and Peruvian<br />
Amazon (Lamont et al., 1999). <strong>Homegardens</strong> close to cities were noted to capitalize<br />
on their relatively easy access to market in exploiting medicinal/other plants<br />
(Padoch and de Jong, 1991; Drescher et al., 2006).<br />
Aromatic species are less common compared to medicinal species in<br />
homegardens. Vetiver (Vetiveria zizanioides) cultivation was, however, observed in<br />
the homegardens of Kerala, India (Nair and Sreedharan, 1986) and the Chagga<br />
gardens on Mt. Kilmanjaro in Tanzania (Fernandes et al., 1984). Likewise,<br />
lemongrass (Cymbopogon citratus) was found in the homegardens of Thailand<br />
(Boonkird et al., 1984), Kerala (Nair and Sreedharan, 1986), and Nicaragua<br />
(Mendez et al., 2001), and citronella (Cymbopogon nardus) in the Kandyan<br />
homegardens of Sri Lanka (Perera and Rajapakse, 1991). <strong>Homegardens</strong> in Ethiopia<br />
also contained aromatic plants (Zemede and Ayele, 1995).<br />
2.3. Uses of MPs grown in homegardens<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
The MPs grown in homegardens are used to treat a variety of ailments ranging from<br />
common colds, fevers, headache, snake bites, and digestive problems to infectious<br />
and complicated diseases (Tables 1, 3, and 4). Thus, we find species yielding<br />
curatives, preventives, placebos, palliatives, nutrition supplements, and energizers.<br />
Some of the species provide medicaments to treat livestock diseases, fish baits, and
214<br />
M.R. RRAO AND B.R. RRAJESWARA RAO R<br />
piscicides. Medicinal and aromatic species found in the homegardens are also<br />
used as biopesticides. For example, leaves of sacred basil (Ocimum sanctum/<br />
O. enuiflorum) are traditionally used as a toxicant against insect pests in grain legume<br />
storage. Clove (Syzygium aromaticum) powder was found to cause adult mortality of<br />
bruchids (Callosobruchus maculatus; Rajapakse et al., 2002). Essential oils of<br />
citronella, Eucalyptus citriodora, and lemongrass are widely used as mosquito<br />
repellants. Parts of MPs used for medicinal purpose could be whole plants, young<br />
shoots, flowers, young leaves, stem, seed, bark, pods, rhizomes, bulbs, fruits, roots,<br />
and inflorescence depending on the species (see Tables 1, 3, and 4).<br />
3. GENDER ISSUES AND MEDICINAL PLANTS<br />
In many traditional societies, women are actively involved in the cultivation of food<br />
crops, while men are more concerned with the cash crops. This is true generally for<br />
Africa, the Ngöbe community of Panama (Samaniego and Lok, 1998), and the<br />
natives of Soqotra Island, Yemen (Ceccolini, 2002). Commercialization of certain<br />
products in the homegardens, however, reduced the diversity of species and income<br />
to women in a number of communities in Latin America (Howard, 2006). The<br />
proverbial reference to household treatment for common ailments, which generally<br />
are based on MPs as ‘grandmother’s remedies’, perhaps indicates the understanding<br />
of women on these aspects. Women also may have as much role as men, if not more,<br />
in the cultivation of traditional medicinal plants, use, and sale of herbal products in<br />
village markets because of proximity. In Nicoya, Costa Rica, it was noted that<br />
although men and women had equal knowledge of the parts used, women had<br />
greater knowledge of medicinal species, the forms of preparation, and application<br />
than men (Ochea et al., 1999; Howard, 2006). In Tanzania, men harvest fuel and<br />
fodder trees, while women harvest fodder grasses and herbs (Fernandes et al., 1984).<br />
Understanding the role of women in homegardens in general and possible impact of<br />
introduction of high value medicinal plants in homegardens on gender equity and<br />
well-being of women within the family and society is important; yet, in-depth<br />
studies are lacking on these aspects.<br />
4. SHADE TOLERANCE OF MEDICINAL PLANTS<br />
Several MPs, especially those grown in homegardens, require or can tolerate<br />
overstorey shade. Ginger (Zingiber officinale) can withstand light interception by<br />
the overstorey up to 48% without experiencing appreciable yield reduction (Kumar<br />
et al., 2001). Yield and quality of galangal or kacholam (Kaempferia galanga) – a<br />
medicinal and aromatic oil-yielding herbs were, however, not affected by light<br />
interception levels by the upperstorey canopy up to 82% of the open (Kumar et al.,<br />
2005). In fact, rhizome yield of galangal as an intercrop in coconut garden was 6.1<br />
Mg ha –1 compared with 4.8 Mg ha –1 in the open in Kerala, India. Essential oil and<br />
oleoresin contents were also greater in the rhizomes of the intercropped kacholam<br />
(Maheswarappa et al., 1998). Likewise, Plumbago rosea, K. galanga, and<br />
Asparagus racemosus performed better as intercrops in 20 year-old coconut
Table 3. Multipurpose trees with medicinal uses grown in, or suitable for, r homegardens.<br />
Latin name Family Parts used Medicinal uses (treatment of the diseases<br />
Where grown<br />
mentioned) and other applications<br />
at present?<br />
Albizia lebbeck k (siris tree) Mimosaceae flowers, seeds,<br />
bark<br />
asthma, thoracic pain, skin diseases, leprosy,<br />
sprains, wounds, ulcers, neuralgia, night blindness,<br />
diarrhea<br />
asthma, bronchitis, leprosy, ulcers, fevers, tumors,<br />
India, Africa<br />
Alstonia scholaris/A. // boonei Apocynaceae leaves, bark,<br />
India, Africa<br />
(devil tree)<br />
milky exudates cardiopathy, helminthiasis, debility, elephantiasis<br />
Azadirachta indica (neem tree) Meliaceae leaves, sticks, bronchitis, diabetes, ulcers, haemorrhoides, skin India, Africa<br />
flowers, seeds, diseases, tumors, syphilis, antiseptic, dandruff,<br />
oil, bark contraception, dental care, insecticide<br />
Bombax buonopozense (bombax) Bombacaceae leaves antipyretic Africa<br />
Cinnamomum camphora/ C. Lauraceae leaves fever, eruptions, measles, delirium, whooping India, China,<br />
parthenoxylon (camphor tree)<br />
cough, melancholia, chronic bronchitis, uterine Sri Lanka<br />
pains, myalgia<br />
Cedrela odorata (cedro) Meliaceae bark snake bites, fever Peru, Brazil,<br />
East Africa<br />
Commiphora mukul<br />
Burseraceae stem, leaves, rheumatic disorders, hyperchhloroesterolaemia India<br />
(Indian bedellium tree)<br />
gum, resin<br />
Croton lechleri<br />
Euphorbiaceae latex swellings, gastric ulcers, contraception Peru<br />
(sangre de drago)<br />
Table 3 (cont.)<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
215
Latin name Family Parts used<br />
Medicinal uses (treatment of the diseases<br />
mentioned) and other applications<br />
Where grown<br />
at present?<br />
Emblica officinalis<br />
Euphorbiaceae fruits aging and general debility, acid-peptic diseases, hair India<br />
(Indian gooseberry)<br />
loss, dyspepsia, laxative, cooling, diuretic, ulcers<br />
Erythrina spp. (Indian coral tree) Fabaceae bark, leaves sedative, vulnerary, lactogogue, collyrium, sterility India, Africa<br />
in women, diabetes, dysentery, eye infections,<br />
insomnia, worms, joint pains, whooping cough<br />
Eucalyptus citriodora (lemon- Myrtaceae leaves essential oil, perfumery, mosquito repellant India, China<br />
scented gum)<br />
Eucalyptus spp. Myrtaceae leaves essential oil, colds India, China<br />
Euterpe precatoria<br />
Arecaceae roots diabetes, vaginal infections Peru<br />
(chonta, pana)<br />
Garcinia cola,<br />
Clusiaceae branch sticks, anthelmenthic, cardiotonic, astringent, demulcent, India, Africa<br />
G. afzelii, G. efunctata<br />
seeds, fruits emollient, antiobesity, dental care<br />
Ginkgo biloba<br />
Ginkgoaceae leaves, fruits old-age problems, memory enhancer, general tonic, China, Japan,<br />
(ginkgo)<br />
adaptogenic<br />
other east<br />
Asian<br />
countries<br />
Gliricidia sepium (Mexican lilac) Fabaceae leaves insecticide India, Africa<br />
Hagenia abyssinica (cusso) Rosaceae flowers anthelminthic, purgative Africa<br />
Jatropha curcas (furging tree) Euphorbiaceae leaves, stem, laxative, lactogogue, leprosy, rheumatism, eczema, India, Africa,<br />
seeds, oil blisters, inflammations, ear-ache<br />
China<br />
216 M.R. RAO R AND B.R. RR RAJESWARA RAO R
Leucaena leucocephala (lead tree) Mimosoideae<br />
bark, root emmengogue, ecbolic, depilatory, contraceptive India, Africa,<br />
America<br />
Madhuca longifolia<br />
Sapotaceae bark, heartwood, inflammations, sprains, pruritus, epilepsy, India<br />
(butternut tree)<br />
flowers, seeds strangury, verminasis, haemotysis, hepatopathy,<br />
dipsia, bronchitis, dermatopathy, cephalalgia,<br />
rheumatism, skin diseases<br />
Maytenus macrocarpa<br />
Celastraceae bark arthritis, diarrhea, stomach disorders, anemia Peru<br />
(chuchuhuasha)<br />
Melia volkensii,<br />
Meliaceae different parts insecticide, anthelmintic, antiseptic, astringent, India, Africa<br />
M. azadirach<br />
emetic, febrifuge, anti-rheumatic<br />
Okoubaka aubrevillei (oku) Octoknamaceae bark vomiting, influenza, infections, diarrhea, gastritis Africa<br />
Pausinystalia johimbe (yohimbe) Rubiaceae bark male impotency, aphrodisiac, hypotensive, cardio Central Africa<br />
tonic<br />
Prunus africana Rosaceae bark prostatitis Central Africa<br />
Saraca asoca (ashoka) Caesalpiniaceae bark, leaves, luecorrhoea, anthelmintic styptic, dyspepsia, ulcers, India<br />
flowers, seeds visceromegaly, pimples, cervical adenitis, vesicle<br />
calculi, haemorrhagic dysentery, diabetes<br />
Terminalia arjuna Combretaceae bark, leaves, wounds, ear-ache, heart diseases, fractures, India, Africa<br />
fruits<br />
contusions, febrifuge, dysentery, diuretic, tonic,<br />
deobstruent, hypertension<br />
Table 3 (cont.)<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
217
Latin name<br />
Terminalia bellirica<br />
(beleric myrobalan)<br />
Family Parts used Medicinal uses (treatment of the diseases<br />
Where grown<br />
mentioned) and other applications<br />
at present?<br />
Combretaceae bark, fruits, gum bronchitis, sore throat, biliousness, inflammations,<br />
strangury, asthma, astringent, dropsy, diarrhea,<br />
leprosy, gum purgative<br />
Combretaceae fruits asthma, soar throat, vomiting, hiccoughs, eye<br />
diseases, heart, and bladder diseases<br />
India, Africa<br />
Terminalia chebula<br />
India, Africa<br />
(cherubulic myrobalan)<br />
Raphia hookeri (raphia palm) Arecaceae exudates cosmetics, wine Africa<br />
Uncaria tomentosa<br />
Rubiaceae bark infections, cancer, gastritis, birth control, allergies Peru, Brazil<br />
(uña de gato)<br />
Zanthoxylum rhoifolium Rutaceae leaves, fruits chest infection, dental care, analgesic, antibacterial Brazil,<br />
Columbia<br />
Source: Saint-Pierre (1991), Chadha and Gupta (1995), and Rao et al. (1999; 2004).<br />
218<br />
M.R. RRAO AND B.R. RR RAJESWARA RAO R
Table 4. Shade-tolerant medicinal species of commercial potential that can be grown in homegardens.<br />
Latin name (common name) Family Habit Part(s) used Uses (treatment of the diseases mentioned) and<br />
other applications<br />
Ranunculaceae herb tuberous root astringent tonic, anti-diarrhea, dyspepsia,<br />
Where grown at<br />
present?<br />
Aconitum heterophyllum<br />
East Asia,<br />
(monks hood)<br />
cough, alexitexic, anti-periodic, anthelmenthic, Western<br />
hemorrhoids, general debility<br />
Himalayas<br />
Adhatoda zeylanica,<br />
Acanthaceae shrub leaves, roots, asthma, menorrhagia, psoriasis, chronic India<br />
A. beddomei (Malabar nut tree)<br />
stem bark bronchitis, cough, body inflammation<br />
Aloe vera, A. barbadensis (aloe) Liliaceae herb leaves health drink, burns, cuts, skin diseases, leprosy, Many countries<br />
piles, liver ailments, dysentery<br />
Alpinia galanga, A. calcarata Zingiberaceae herb rhizomes asthma, bronchitis, hiccoughs, dyspepsia, Malaysia,<br />
(galangal)<br />
diabetes, obesity, rheumatoid arthritis, Indonesia<br />
stimulant, tonic<br />
Ammomum villosum Apiaceae herb fruit stomachic, carminative, expectorant, tonic, China<br />
antiemetic, antispasmodic<br />
Andrographis paniculata Acanthaceae herb shoots antipyretic, antiperiodic, anti-inflammatory, India<br />
(king of bitters)<br />
ulcers, bronchitis, skin diseases, intestinal<br />
worms, jaundice, leprosy, hemorrhoids<br />
Asparagus racemosus, Liliaceae climber tuberous lactogogue, urinary and gynecological India<br />
A. adscendens (asparagus)<br />
roots disorders, diseases of nervous system,<br />
hyperacidity, gastritis, cardiac debility,<br />
hypertension, oligospermea<br />
Table 4 (cont.)<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
219
Latin name (common name) Family Habit Part(s) used Uses (treatment of the diseases mentioned) and<br />
other applications<br />
Boerhaavia diffusa (hog weed) Nyctaginaceae creeping<br />
herb<br />
Cassia senna, C. acutifolia<br />
(senna)<br />
Catharanthus roseus<br />
(periwinkle)<br />
Centella asiatica (Indian<br />
pennywort/goticola)<br />
Chlorophytum borivilianum,<br />
C. tuberosum,<br />
C. arundinaceum (safed musli)<br />
whole plant aphrodisiac, diuretic, cardiac disorders,<br />
stimulant, diaphoretic, anti-inflammatory,<br />
jaundice, anemia, general debility, myalgia,<br />
scabies, oedema<br />
Where grown at<br />
present?<br />
many tropical<br />
countries<br />
Caesalpiniaceae shrub leaves, pods constipation, skin diseases India, Sudan<br />
Apocynaceae small<br />
shrub<br />
leaves, roots cancer therapy (leaves), hypertension (roots) India, China,<br />
Apiaceae creeper whole plant memory enhancer, anxiety, neurosis, general India<br />
debility, wound healing, leprosy, eczema,<br />
sporiasis<br />
Liliaceae herb tubers aphrodisiac, nervine tonic, India<br />
Costus speciosus Zingiberaceae shrub rhizomes contraception, aphrodisiac astringent, digestive, India<br />
skin diseases, fevers<br />
Curculigo orchioides<br />
Amaryllidaceae herb roots erectile impotency, mm spermatorrhoea, general India<br />
(black musli)<br />
weakness, burning and fatigue piles,<br />
menorrhagia, jaundice<br />
Curcuma angustifolia Zingiberaceae herb tubers anti-diarrheal, anti-dysenteric, coolant, health India<br />
(arrow root)<br />
drink<br />
Central and South<br />
America<br />
220 M.R. RRAO AND B.R. RR RAJESWARA RAO R
Cymbopogon martinii var. Poaceae shrubby flowering perfumery, flavoring, joint pains, galactogogue, South and<br />
motia<br />
grass shoots febriluge, aromatherapy<br />
Southeast Asia<br />
Decalepis hamiltonii Asclepiadaceae twining tuberous health drink, tonic, promotes digestion, cures India<br />
straggler roots fever<br />
Dioscorea deltoides/<br />
Dioscoreaceae tuberous tubers steroidal drugs, contraception, anthelmintic, India<br />
D. floribunda (medicinal yam)<br />
twines<br />
leprosy<br />
Gloriosa superba (glory lily) Liliaceae climbing tuber, seeds gout, polyploidy, rheumatism, abortifacient, India<br />
herb<br />
chronic ulcers, piles, diarrhea, antiperiodic,<br />
anthelmintic, snake bites, scorpion stings,<br />
gonorrhea<br />
Glycyrrhiza glabra (liquorice) Fabaceae shrub roots cough, general tonic, acid peptic disease anti- India, China,<br />
inflammatory, sweetener<br />
Eurasia<br />
Gymnema sylvestre (pepricola Asclepiadaceae climbing leaves antidiabetic, cardiac stimulant, eye diseases, India<br />
of the wood)<br />
shrub<br />
diuretic<br />
Hippophae rhamnoides Elaeagnaceae shrub berries skin care, analgesic, antioxidant, antibacterial, USA, Canada,<br />
(seabuckthorn)<br />
anti-inflammatory, nutraceutical<br />
Europe, India<br />
Holostemma adakodien Asclepiadaceae climber roots opthalmopathy, fever, arthritis, cough, burning India<br />
(swallow wort/ring coronet)<br />
sensation, stomachalgia<br />
Mucuna pruriens (velvet bean) Fabaceae climbing seeds Parkinson’s disease, anthelmintic, laxative, many countries<br />
shrub<br />
tonic for male virility, elephantiasis<br />
Table 4 (cont.)<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
221
Latin name (common name) Family Habit Part(s) used Uses (treatment of the diseases mentioned) and<br />
Where grown at<br />
other applications<br />
present?<br />
Ocimum sanctum<br />
Lamiaceae herb flowering cold, cough, bronchospasm, general debility, India, West<br />
(sacred basil)<br />
shoots stress disorders, skin infections, wounds, Indies<br />
indigestion, nausea, essential oil in flavoring,<br />
perfumery<br />
Phyllanthus amarus Euphorbiaceae herb whole plant hepatoprotective, oedema, anorexia many countries<br />
Piper longum (long pepper) Piperaceae climbing fruit, stem, bronchial asthma, throat infections, flatulence, India<br />
shrub roots dyspepsia, respiratory diseases, analgesic,<br />
carminative, sedative, insomnia, epilepsy,<br />
abortifacient<br />
Plumbago zeylanica,<br />
Plumbaginaceae herb roots acro-narcotic poison, abortifacient, rheumatic India<br />
P. rosea<br />
and paralytic affections, ulcers, leprosy,<br />
(white/red flowered lead wort)<br />
enlarged spleen, rubefacient, piles, skin<br />
diseases, leucoderma, syphilis, influenza<br />
Pueraria tuberosa<br />
Fabaceae large tubers arthritis, agalactia, cardiac debility, pharyngitis, India<br />
(Indian kudzu)<br />
climber<br />
leprosy, tuberculosis, spermatorrhoea<br />
Rauvolfia serpentina, Apocynaceae shrub roots leaves hypertension, insanity, insomnia, psychological India, Malaysia,<br />
R. tetraphylla,<br />
disorders, epilepsy eczema, skin diseases Indonesia<br />
R. vomitoria (serpentine root)<br />
opacities of cornea, psoriasis, snake and insect<br />
bites, toxic goiter, angina pectoris<br />
222 M.R. RRAO AND B.R. RR RAJESWARA RAO R
Rosemarinus officinalis Lamiaceae herb flowering perfumery, aromatherapy, digestive, nervine Mediterranean<br />
shoot tonic, stimulates kidneys<br />
region<br />
Salvia officinalis<br />
Lamiaceae herb leaves, oil perfumery, digestive, nervine tonic, antiseptic, Mediterranean<br />
(sage)<br />
deodorant, diaphoretic<br />
region<br />
Stevia rebaudiana (stevia) Asteraceae herb leaves sweetener Brazil, Japan,<br />
Paraguay,<br />
China,<br />
Indonesia,<br />
Thailand<br />
Tinospora cordifolia (tinospora) Menispermaceae woody stem seminal weakness, urinary affections, tonic, India<br />
climber<br />
fever, jaundice, syphilis, rheumatism, general<br />
debility, leprosy<br />
Source: Saint-Pierre (1991), Rao et al. (1999), Chadha and Gupta (1995), and Kehlenbeck and Maass (2005).<br />
MEDICINAL M PLANTS IN HOMEGARDENS<br />
223
224<br />
M.R. RRAO AND B.R. RRAJESWARA RAO R<br />
plantations spaced at 7.5 x 7.5 m, and gave 69 to 97% higher net returns compared<br />
to sole crops. The performance of Adhatoda beddomei and Holostemma adakodien,<br />
however, was unaffected by the cropping systems (Kurien et al., 2003), implying<br />
that they could perform well under disparate cropping situations. Patchouli<br />
(Pogostemon patchouli), an important aromatic crop, is grown as an intercrop in the<br />
coconut gardens of India. Its biomass yield and quality of oil were better under<br />
shade than when grown in the open (E.V.S. Prakasa Rao, CIMAP Resource Centre,<br />
Bangalore, India, pers. comm., July 2005). Black musli or golden eye grass<br />
(Curculigo orchioides) planted at 10 x 10 cm spacing under 25% shade performed<br />
better than the crop in the open in terms of vegetative growth, rhizome yield, harvest<br />
index, and nutrient uptake 4 .<br />
Most of the medicinal plants harvested from forests are shade tolerant or prefer<br />
some degree of shade, so that they can be cultivated in the homegardens as well,<br />
provided they are adapted to the prevailing climatic and soil conditions. A number<br />
of medicinal and aromatic crops that are traditionally grown outside forests can also<br />
withstand some shade (Jha and Gupta, 1991; Nair et al., 1991) and such species too<br />
can be integrated into homegardens. Tables 3 and 4 list a number of species that can<br />
be promoted in the homegardens. Species requiring mild shade may be grown in the<br />
early years of newly established homegardens or in patches under partial shade,<br />
whereas those that withstand intense shade can be grown in ‘mature’ homegardens.<br />
5. PROMOTING MEDICINAL CROPS IN HOMEGARDENS<br />
With the future of homegardens themselves being uncertain (Kumar and Nair, 2004;<br />
Wiersum, 2006), its role in providing a steady supply of medicinal plants and other<br />
products is unclear. Consistent with this, some reports indicate a reduction in the<br />
supply of MPs from homegardens. For example, an analysis of the species<br />
composition of homegardens in West Java, Indonesia in 1980 and 1999 revealed that<br />
fruit trees and ornamentals constituted a high proportion of plant species in both the<br />
years. There was, however, a decrease in the number of useful species from 126 to<br />
100 during the 1999 enumeration. The utilization of useful plants, except for fruit<br />
trees and plants for miscellaneous uses largely changed in the past 20 years<br />
especially in the case of vegetable, industrial, and ornamental plants (Kubota et al.,<br />
2002). In Catalonia, MPs declined because of the loss of original significance of<br />
certain species and death of people with particular knowledge on the cultural<br />
requirements of some plants (Agelet et al., 2000).<br />
In spite of the above uncertainties, homegardens offer an opportunity to produce<br />
some high value medicinal crops and help smallholders earn additional incomes. For<br />
instance, in the Ba Vi National Park in northern Vietnam, the Dao people have taken<br />
up cultivation in the homegardens some of the 44 commercially important medicinal<br />
species identified in the area including Alstonia scholaris, Cinnamomum<br />
zeylanicum, Tradescantia zebrine, Piper retrofractum, and Travesia palmatet t (On<br />
et al., 2001). Ammomum villosum in China (Saint-Pierre, 1991) and Piper longum<br />
and Kaempferia galanga in India (Kumar et al., 2005) are similarly grown for<br />
commercial purposes. In the Peruvian Amazon, younger generations were as keen as<br />
the older ones to add species potentially useful as medicine, food, cosmetics, and
MEDICINAL M PLANTS IN HOMEGARDENS<br />
225<br />
other items to their collections as well as gathering knowledge on such plants<br />
(Padoch and de Jong, 1991).<br />
In the humid tropics, the active slash-and-burn agriculture (120 million ha),<br />
secondary forest fallow (203 million ha), logged forests (136 million ha), secondary<br />
forest fallows (203 million ha), Imperata-infested grasslands in Southeast Asia (40<br />
million ha), and degraded pastures in the Amazon (10 million ha) present vast<br />
degraded and abandoned areas, some of which can be put under permanent crop<br />
production systems (Sanchez et al., 1994). <strong>Homegardens</strong> and multistrata systems are<br />
regarded as some of the best bet alternatives to slash-and burn system both for the<br />
newly cleared lands as well as to bring degraded lands into permanent production. In<br />
the uplands of northern Vietnam, the need for improved homegardens using<br />
medicinal crops, rattan, quality timber, and livestock was recognized to replace<br />
shifting cultivation and to prevent opium m production (Tai et al., 1995). <strong>Homegardens</strong><br />
have been taken up by smallholders in the re-settlement projects in Southeast Asia<br />
(e.g., Indonesia) and Amazon (e.g., Brazil). The native people and migrants in the<br />
course of developing their homegardens have used a wealth of plant materials<br />
including recently developed germplasm. A survey of 33 homegardens in the<br />
uplands and 18 in the floodplains of Brazilian Amazon revealed that a total of 77<br />
and 80 commercially valuable perennial species respectively are present (Smith,<br />
1996). These species included, in addition to those providing food, beverages,<br />
juices, nuts, oils, thatch, and wood, those that provided folk remedies such as juca<br />
(Caesalpinia ferrea), piao roxo (Jatropha gossypiifolia), yellow mombim or<br />
taperebá (Spondias mombim), fish bait (e.g., Colossoma macropomum, C. bidens,<br />
and Brycon sp), and piscicide (e.g., Ichthyothere cunabi). The species diversity was<br />
greater if medicinal, ornamental, and vegetable species meant mostly for family use<br />
were also considered. The number of such species in gardens ranged from 4 to 27.<br />
<strong>Homegardens</strong> established recently as alternatives to slash-and-burn agriculture in<br />
cleared forests or degraded lands, however, did not contain as many medicinal<br />
species as the traditional gardens. Similarly, recently established homegardens in<br />
southern Andaman, India did not contain medicinal plants (Pandey et al., 2002).<br />
Official recognition of traditional medicine will promote growing of medicinal<br />
plants, which in turn would help farmers earn better price to their products and<br />
citizens to get healthcare at reduced costs. <strong>Homegardens</strong> and health resorts could<br />
also promote ecotourism or ‘health tourism’, as is happening in the Kerala state of<br />
India (www.ktdc.com and www.keralatourism.org; last accessed: December 2005).<br />
The social benefits include revival of local traditions and protection of traditional<br />
knowledge. It is possible to patent indigenous knowledge about medicinal plants and<br />
preparations of products so that the society associated with the development of such<br />
knowledge derive the economic benefits thereof. Patenting of the stress relieving<br />
properties of Trichopus zeylanicus, a medicinal plant used by the Kani tribals of<br />
Agasthyar hills in Kerala is worth mentioning in this context (TBGRI, 2003).<br />
Indeed, a share of the royalty paid by the firm, which commercialized the<br />
technology, has been passed on to the tribal community that possessed this<br />
knowledge as part of their traditions. Value-addition and product development at<br />
local level wherever possible would also increase the earnings of farmers as well as<br />
create rural employment to skilled people and reduce migration to cities.
226<br />
M.R. RRAO AND B.R. RRAJESWARA RAO R<br />
6. MARKETING OF MEDICINAL PLANTS FROM HOMEGARDENS<br />
Local markets may not be adequate in most cases to absorb all the commercially<br />
valuable MPs and offer an equitable price to the producer; prices offered at these<br />
markets are often only a small fraction of those at the national and international<br />
markets. Lack of organized market channels, poor infrastructure, and involvement of<br />
middlemen in the supply chain from farm to factory deprive the farmers of<br />
remunerative prices to their produce. Strategies that will promote marketing of MPs<br />
and offer competitive prices to farmers are needed; these include establishment of<br />
farmers’ <strong>coop</strong>eratives, contract farming with ‘buyback’ arrangements by the industry,<br />
declaration of minimum support price to promising MPs, and subsidies to exporters<br />
of MPs as in other sectors. Examples of such proactive policies include development<br />
of a marketing network for Piper longum in Andhra Pradesh (India) and the<br />
intervention by government agencies in the case of Ammomum villosum in China,<br />
which encouraged large-scale cultivation of these MPs in homegardens. The Girijan<br />
Cooperatives in many Indian states also help the tribals living at forest margins to<br />
market non-wood forest products. Likewise, the Mayan farmers in the Yucatan<br />
region of Mexico have organized a <strong>coop</strong>erative project for the sale of aloe ( (Aloe<br />
barbadensis) and orange juice produced from forest gardens (Neugebauer and<br />
Mukul, 2000). Dabur India Ltd., a pharmaceutical company that makes herbal<br />
medicines, relies on contract farming for the supply of Indian gooseberry (Emblica<br />
officinalis), Rauvolfia, and Piper longum. Maintaining quality of the produce all<br />
through the supply chain is, however, very important to earn a premium price for<br />
which the farmers, transporters, and processors should be trained properly.<br />
7. OUTLOOK AND RESEARCH NEEDS<br />
Clearly, not all medicinal and aromatic plants found in the homegardens are used by<br />
people, and the relative importance of these plants to local societies also varies<br />
greatly from place to place. As a first step, therefore, priority species need to be<br />
identified based on their medicinal importance, ailments for which they are used,<br />
commercial value, cost effectiveness of alternate medicines, and the potential for<br />
synthesizing alternative compounds. Research efforts could then concentrate on a<br />
few priority species in terms of improving germplasm and developing agronomic<br />
techniques, particularly effective propagation techniques, and field establishment in<br />
homegardens and forest gardens. Sustainable harvesting methods have to be<br />
developed, especially for species harvested from the wild.<br />
Basic research is needed on the response of important medicinal species that are,<br />
and can be grown, in the homegardens to variations in quantity and quality of light;<br />
and to determine the effects of varying light regimes and organic and inorganic<br />
sources of nutrients on yield and quality. Such information helps to develop<br />
appropriate canopy management practices for multistrata systems to facilitate the<br />
growth of understorey crops. The use of MPs is based on indigenous knowledge and<br />
customs passed down from generations; the principal chemical compounds in many<br />
of these plants and their curative properties and mode of action have not yet been
MEDICINAL M PLANTS IN HOMEGARDENS<br />
227<br />
elucidated properly. Such studies will give authenticity to the use of traditional<br />
medicines and help protect genuine herbalists from unscrupulous practitioners.<br />
Globalization of agricultural trade under the World Trade Organization (WTO)<br />
regime brought with it several challenges and opportunities in the medicinal and<br />
aromatic plants sector too. The challenges include price competition, maintenance of<br />
quality, and scientific validation of claims for traditional medicines. The<br />
opportunities include global positioning of natural products obtained from medicinal<br />
plants, which have large demand. Bioprospecting for molecules of pharmaceutical or<br />
flavor/fragrance value from these plants and patenting of these molecules is going to<br />
be a future source of conflict between developed and developing countries. While<br />
the developed countries have the technology and fiscal resources, the developing<br />
countries in the tropics, where most of these MPs are grown, lack such resources. As<br />
a first step, therefore, tropical countries should make efforts to develop databases on<br />
MPs, indigenous medicinal practices, and herbal preparations in use. These will not<br />
only prevent loss of indigenous knowledge but also help promote the use of MPs.<br />
Documentation further helps native communities to protect their intellectual<br />
property rights on their genetic resources and indigenous knowledge systems and<br />
safeguard from biopiracy (Jose, 2004).<br />
7.1. Processing of homegarden produced MPs<br />
Medicinal and aromatic plants in homegardens can be produced at a lower cost<br />
compared to input intensive sole crops, as they benefit from common field<br />
operations and minimal use of chemical inputs. Organically produced MPs may also<br />
attract premium prices in the international markets. Processing and packaging of<br />
MPs at local level instead of selling the raw materials will further increase the value<br />
of the products and benefit the growers. Some typical value-addition practices are:<br />
(1) drying and powdering of relevant plant parts, (2) distillation of aromatic plants,<br />
(3) isolation of menthol crystals from mentha oil (Mentha spp.) following chilling<br />
and centrifuging, (4) pulverizing and encapsulation (e.g., peeled and dried tubers of<br />
Chlorophytum borivilianum in India), (5) preparation of herbal extracts, and (6)<br />
preparation of simple products such as incense sticks, perfumed candles, soaps, and<br />
herbal drugs. Powdering medicinal plant parts is the simplest activity, which can be<br />
taken up at the farm-level; e.g., tribals cultivating Curcuma angustifolia in Andhra<br />
Pradesh state, India, prepare a white powder from the tubers of this plant. Other<br />
processes may need establishment of facilities at village- or community-level as<br />
cottage industries. Nevertheless, it may increase profits to the farmers and generate<br />
employment to the local people. For instance, in Karnataka state of India, incense<br />
sticks are made mostly by women and children using plant-derived raw materials,<br />
adding value to these products and enhancing household incomes. Farmers have to<br />
be encouraged and trained, if necessary, to take such value-addition processes either<br />
individually or collectively at the farm- or village-level to realize better prices for<br />
their products. Good packaging, branding, organic labeling, and quality certification<br />
by authorized agencies for finished products will also increase the value of herbal<br />
medicines and its consumer acceptability.
228<br />
M.R. RAO R AND B.R. RRAJESWARA RAO R<br />
8. CONCLUSIONS<br />
<strong>Homegardens</strong> will continue to be an important land use system for the small-scale<br />
farmers in humid and subhumid tropics. They can be turned into future ‘biofactories’ for<br />
the production of commercially important phytochemicals. Furthermore, organically<br />
grown MPs can be an important income and employment generating village<br />
enterprise in many rural localities. Promotion of ecotourism to herbal/homegardens<br />
and health resorts catering to aromatherapy or herbal therapy will have its spin off in<br />
terms of additional income and rural employment. Training farmers in improved<br />
cultivation and processing practices, contract farming, and establishment of<br />
institutions that provide market information and ensure quality standards will go a<br />
long way in promoting MPs in the homegardens.<br />
ACKNOWLEDGEMENTS<br />
The authors sincerely thank Dr. J.S.K. Prasad, Central Research Institute for<br />
Dryland Agriculture (CRIDA), Santhoshnagar, Hyderabad 500 030, India, and Mr.<br />
Solomon G. Haile, School of Forest Resources and Conservation, University of<br />
Florida, Gainesville, Florida, USA for providing some useful literature in the course<br />
of preparing this paper.<br />
ENDNOTES<br />
1. World Bank stresses importance of coming phytomedicines. Newsletter of the<br />
Asian Network on Medicinal and Aromatic Plants. 23: 5 – 6 (1997).<br />
2. DGCIS 2004. Monthly statistics of foreign trade of India. Annual report for<br />
2003 – 2004 (Vol. 1). Exports including re-exports. Directorate General of<br />
Commercial Intelligence and Statistics, Ministry of Commerce, Kolkata.<br />
3. Emerging trends in productivity of medicinal and aromatic plants. Newsletter of<br />
the Asian Network on Medicinal and Aromatic Plants. 18: 7 (1996).<br />
4. Joy P.P., Savithri K.E., Mathew S. and Thomas J. 2005. Optimum shade and<br />
spacing for black musli (Curculigo orchioides Gaertn.). In: Book of Abstracts<br />
‘National seminar on achievements and opportunities in post-harvest<br />
management and value addition in roots and tuber crops’, 19 – 20 July 2005,<br />
Central Tuber Crops Research Institute, Thiruvananthapuram, Kerala, p114.<br />
REFERENCES<br />
Agelet A., Bonet M.A. and Valles J. 2000. <strong>Homegardens</strong> and their role as a main source of<br />
medicinal plants in mountain regions of Catalonia (Iberian peninsula). Econ Bot 54:<br />
295 – 309.<br />
Albuquerique de U.P. and Andrade L. de H.C. 2002. Conhecimento botánico tradicional e<br />
conservação em uma área de caatinga no estado de Pernambuco, nordeste do Brasil. Acta<br />
Botanico do Brasil 16: 273 – 285.<br />
Boonkird S.A., Fernandes E.C.M. and Nair P.K.R. 1984. Forest villages: an agroforestry<br />
approach to rehabilitating forest land degraded by shifting cultivation in Thailand.<br />
Agroforest Syst 2: 87 – 102.
MEDICINAL M PLANTS IN HOMEGARDENS<br />
229<br />
Ceccolini L. 2002. The homegardens of Soqotra island, Yemen: an example of agroforestry<br />
approach to multiple land use in an isolated location. Agroforest Syst 56: 107 – 115.<br />
Chadha K.L. and Gupta R. 1995. Medicinal and aromatic plants. Advances in horticulture<br />
(Vol 11). Malhotra Publishing House, New Delhi, 932p.<br />
Corlett J.L., Dean E.A. and Grivetti L.E. 2003. Hmong gardens: Botanical diversity in an<br />
urban setting. Econ Bot 57: 365 – 379.<br />
De Clerck F.A.J. and Negreros-Castillo P. 2000. Plant species of traditional Mayan<br />
homegardens of Mexico as analogs for multistrata agroforests. Agroforest Syst 48:<br />
303 – 317.<br />
Drescher A.W., Holmer R.J. and Iaquinta D.L. 2006. Urban homegardens and allotment<br />
gardens for sustainable livelihoods: Management strategies and institutional environments<br />
In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of<br />
sustainable agroforestry, pp 317 – 338. Springer Science, Dordrecht.<br />
EXIM Bank 2003. Export potential of Indian medicinal plants and products. Publication No.<br />
OP 98. Export and Import Bank of India (EXIM Bank), Mumbai, India (see also<br />
www.eximbankindia.com/publications; last accessed: October 9, 2005).<br />
Farnsworth N.R. and Soejarto D.D. 1991. Global importance of medicinal plants. In: Akerele<br />
O., Heywood V., and Synge H. (eds), The conservation of medicinal plants, pp. 25 – 51.<br />
Cambridge University Press, Cambridge.<br />
Fernandes E.C.M., O’kting’ati A. and Maghembe J.M. 1984.The Chagga homegardens: a<br />
multistoreyed cropping system on Mount Kilimanjaro (Northern Tanzania). Agroforest<br />
Syst 2: 73 – 86.<br />
Food and Agricultural Organization (FAO) 1996. Forests, food and health.<br />
www.fao.org/forestry/site /28813/em m (last accessed: October 10, 2005).<br />
Howard P.L. 2006. Gender and social dynamics in swidden and homegardens in Latin<br />
America In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 159 – 182. Springer Science, Dordrecht.<br />
Jha K.K. and Gupta C. 1991. Intercropping of medicinal plants with poplar and their<br />
phenology. Indian For 117: 535 – 544.<br />
Jose R. 2004. US firm hijacks Kerala patent. www.huk.org/articles/0204/78.html (last<br />
accessed: October 10, 2005).<br />
Kehlenbeck K. and Maass B.L. 2005. Crop diversity and classification of homegardens in<br />
Central Sulavesi, Indonesia. Agroforest Syst 63: 53 – 62.<br />
Kubota N., Hadikusumah H.Y., Abdoellah O.S. and Sugiyama N. 2002. Changes in the<br />
performance of the homegardens in West Java for twenty years. 2. Changes in the<br />
utilization of cultivated plants in the homegardens. Jpn J Trop Agr 46: 152 – 161.<br />
Kubota N., Shimamura K. and Ogo T. 1992. Useful plant species observed in homegardens,<br />
fields and local markets in Java and Sumatra islands. 2. Spice, medicinal, industrial and<br />
miscellaneous plants. Jpn J Trop Agr 36: 298 – 308.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst<br />
61/62: 135 – 152.<br />
Kumar B.M., George S.J. and Chinnamani S. 1994. Diversity, structure and standing stock of<br />
wood in the homegardens of Kerala in Peninsular India. Agroforest Syst 25: 243 – 262.<br />
Kumar B.M., Kumar S.S., and Fisher R.F. 2005. Galangal growth and productivity related<br />
light transmission in single-strata, multistrata and no-over-canopy systems. J New Seeds<br />
7: 111 – 126.<br />
Kumar B.M., Thomas, J. and. Fisher, R.F. 2001. Ailanthus triphysa at different density and<br />
fertilizer levels in Kerala, India: tree growth, light transmission and understorey ginger<br />
yield. Agroforest Syst 52: 133 – 144.<br />
Kurien A., Augustin A. and Nybe E.V. 2003. Economic analysis of resource-based cropping<br />
in selected medicinal species. In: Mathur A.K., Dwivedi S., Patra D.D., Bagchi G.D.,
230<br />
M.R. RAO R AND B.R. RRAJESWARA RAO R<br />
Sangwan N.S., Sharma A., and Kanuja S.P.S. (eds), Proceedings of the first national<br />
interactive meet on medicinal and aromatic plants, pp. 47 – 49. Central Institute of<br />
Medicinal and Aromatic Plants, Luknow.<br />
Lamont S.R., Eshbaugh W.H. and Greenberg A.M. 1999. Species composition, diversity and<br />
use of homegardens among three Amazonian villages. Econ Bot 53: 312 – 326.<br />
Maheswarappa H.P., Hegde M.R, and Nanjappa M.V. 1998. Kacholum (Kaempferia galanga)<br />
– a potential medicinal-cum-aromatic crop for coconut gardens. Indian Coconut J<br />
(Cochin) 29 (5): 4 – 5.<br />
Mendez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Michon G., Mary F. and Bompard J. 1986. Multistoreyed agroforestry garden system in west<br />
Sumatra, Indonesia. Agroforest Syst 4: 315 – 338.<br />
Millat-e-Mustafa M., Khodeja Begum, Mohammed-Al-Amin and Shafiul Alam Md. 2001.<br />
Medicinal plant resources of the traditional homegardens in Bangladesh. J Trop Med<br />
Plants 2: 99 – 106.<br />
Millat-e-Mustafa M., Teklehaimanot Z. and Haruni A.K.O. 2002. Traditional uses of<br />
perennial homestead garden plants in Bangladesh. Forests Trees Livelihoods 12:<br />
235 – 256.<br />
Mpoyi K., Lukebakio N., Kapendo K. and Paulus J. 1994. Inventaire de la flore domestique<br />
des parcelles d’habitation. Cas de Kinshasa (Zaire). Revue de Mé et Pharmacopée<br />
Africaine 8(1): 55–66.<br />
Mulas M.G., Quiroz C., Perez S. D.M., Rodriguez D., Perez T., Marques A. and Pacheco W.<br />
2004. Conservacion in situ de diversas especies vegetales en ‘conucos’ (home gardens) in<br />
the states of Carabobo y Trujillo de Venezuela. Plant Gen Resour Newsl 137: 1 – 8.<br />
Nair P.K.R. and Kumar B.M. 2006. Introduction. In: Kumar B.M. and Nair P.K.R. (eds),<br />
<strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp 1 – 10.<br />
Springer Science, Dordrecht.<br />
Nair M.A. and Sreedharan C. 1986. Agroforestry farming systems in the homesteads of<br />
Kerala, southern India. Agroforest Syst 4: 339 – 363.<br />
Nair G.S., Sudhadevi P.K. and Kurian A. 1991. Introduction of medicinal and aromatic plants<br />
as intercrops in coconut plantations. a In: Raychaudhuri S.P. (ed.), Recent advances in<br />
medicinal, aromatic and spice crops, pp 163 – 165. Today and Tomorrow’s Printers &<br />
Publishers, New Delhi.<br />
Neugebauer B. and Mukul Ek A. 2000. Trees for people – a Mayan strategy towards organic<br />
agriculture. In: Alfoldi T.T., Lockeretz W., and Niggli U. (eds), The world grows organic:<br />
Proceedings 13th International IFOAM scientific conference (28-31 August 2000),<br />
IFOAM, Basel, Switzerland, 428p.<br />
Ochea L., Fassaert C., Somarriba E. and Schlonvoight A. 1999. Medicinal and food plants in<br />
Nicoya, Costa Rica: there are differences in what men know and women know.<br />
Agroforest Today 11: 1–2, 11 – 12.<br />
Okafor J.C. and Fernandes E.C.M. 1987. The compound farms of southeastern Nigeria: a<br />
predominant agroforestry homegarden system with crops and small livestock. Agroforest<br />
Syst 5: 153 – 168.<br />
O’Kting’ati A., Maghembe J.A., Fernandes E.C.M. and Weaver G.H. 1984. Plant species in<br />
the Kilimajaro agroforestry system. Agroforest Syst 2: 177 – 186.<br />
On T.V., Quyen D., Bich L.D., Jones B., Wunder J., and Russel-Smith J. 2001. A survey of<br />
medicinal plants in Ba Vi National Park Vietnam: methodology and implications for<br />
conservation and sustainable use. Biol Conserv 97: 295 – 304.<br />
Padoch C. and de Jong W. 1991. The house gardens of Santa Rosa: diversity and variability in<br />
an Amazonian agricultural system. Econ Bot 45: 166 – 175.
MEDICINAL M PLANTS IN HOMEGARDENS<br />
231<br />
Pandey C.B., Kanak Lata, Venkatesh A., Medhi R.P. and Lata K. 2002. <strong>Homegardens</strong>: its<br />
structure and economic viability in South Andaman. Indian J Agroforest 4: 17 – 23.<br />
Perera A.H. and Rajapakse R.M.N. 1991. A baseline study of Kandian forest gardens of Sri<br />
Lanka: structure, composition and utilization. For Ecol Manag 45: 269 – 280.<br />
Principe P.P. 1991. Valuing the biodiversity of medicinal plants. In: Akeele O., Heywood V.,<br />
and Synge H. (eds), The conservation of medicinal plants, pp 79 – 124. Cambridge<br />
University Press, Cambridge.<br />
Rao B.R.R. 1999. Medicinal plants for dry areas. In: Singh R.P. and Osman M. (eds),<br />
Sustainable alternate land use systems for drylands, pp 139 – 156. Oriental Enterprises,<br />
Dehra Dun.<br />
Rao M.R., Palada M.C. and Becker B.N. 2004. Medicinal and aromatic plants in agroforestry<br />
systems. Agroforest Syst 61/62: 107 – 122.<br />
Rao P.S., Venkaiah K. and Padmaja R. 1999. Field guide on medicinal plants. Forest<br />
Department, Government of Andhra Pradesh, Hyderabad, India, 208p.<br />
Rajapakse R., Rajapakse H.L. de Z. and Ratnasekera D. 2002. Effect of botanicals on<br />
oviposition, hatchability and mortality of Callosobruchus maculatus L. (Coleoptera:<br />
Bruchidae). Entomon 27: 93 – 98.<br />
Rico-Gray V., Chemas A. and Mandujano S. 1991. Use of tropical deciduous forest species<br />
by the Yucatecan Maya. Agroforest Syst 14: 149 – 161.<br />
Rugalema G.H., Okting’ati A. and Johnsen F.H. 1994. The homegarden agroforestry system<br />
of Bukoba district, Northwestern Tanzania. 1. Farming system analysis. Agroforest Syst<br />
26: 53 – 64.<br />
Saint-Pierre C. 1991. Evolution of agroforestry in the Xishuangbanna region of tropical<br />
China. Agroforest Syst 13:159 – 176.<br />
Samaniego G. and Lok R. 1998. Valor de la percepcion y del conocimiento local de indigenas<br />
Ngöbe, en Chiriqui, Panama. Agroforesteria en las Americas 5 (17/18): 12 – 16.<br />
Sanchez P.A., Woolmer P.L. and Palm C.A. 1994 Agroforestry approaches for rehabilitating<br />
degraded lands after tropical deforestation. In: Rehabilitation of degraded forest lands in<br />
the tropics: Technical approach. JIRCAS International Symposium Series No. 1, pp<br />
108 – 119. Japan International Research Centre for Agriculture Research, Tsukuba,<br />
Ibaraki.<br />
Smith N.J.H. 1996. <strong>Homegardens</strong> as a springboard for agroforestry development in<br />
Amazonia. Int Tree Crops J 9: 11 – 30.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: a traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development,<br />
pp 157 – 170. ICRAF, Nairobi.<br />
Tai N.D., Nhan H.D., Yen N.T. and Cameron D.M. 1995. Socio-economic considerations in<br />
the planning of agroforestry systems for the acid uplands of northern Vietnam. In: Date<br />
R.A., Grundon N.J., Rayment G.E., and Probert M.E. (eds), Plant–soil interactions at low<br />
pH: Principles and management. Proceedings of the Third International Symposium<br />
(September 12–16, 1993), pp 697 – 702. Brisbane, Queensland.<br />
<strong>Tropical</strong> Botanic Garden and Research Institute (TBGRI) 2003. TBGRI, Palode,<br />
Thiruvananthapuram, Kerala, www.tbgri.org/tbgri/patent/htm (last accessed: October 10,<br />
2005).<br />
Viquez E., Prado A., Onoro P., Solano R. and Solano A.R. 1994. Characterization of the<br />
tropical mixed garden ‘La Asuncion’, Masatepe, Nicaragua. Agroforesteria en las<br />
Americas 1(2): 5 – 9.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
World Health Organization (WHO) 2004. www.WHO.int/entity/mediacentre/news/notes/<br />
2004/np3/en (last accessed: October 9, 2005)
232<br />
M.R. RRAO AND B.R. RRAJESWARA RAO R<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.<br />
Yoshino K. and Ando K. 1999. Utilization of plant resources in homestead (bari-bhiti) in<br />
floodplain in Bangladesh. Japanese J Trop Agric 43: 306 – 318.<br />
Zemede A. and Ayele N. 1995.<strong>Homegardens</strong> in Ethiopia: Characteristics and plant diversity.<br />
Sinet-An Ethiopian J Sci 18(2): 235 – 266.
CHAPTER 13<br />
COMMERCIALIZATION OF<br />
HOMEGARDENS IN AN INDONESIAN<br />
VILLAGE: VEGETATION COMPOSITION<br />
AND FUNCTIONAL CHANGES<br />
O.S. ABDOELLAH 1* , H.Y. HADIKUSUMAH 2 , K. TAKEUCHI 3 ,<br />
S. OKUBO 3 , AND PARIKESIT 2<br />
1 Institute of Ecology and Department of Anthropology and 2 Department of Biology,<br />
Padjadjaran University, Bandung, Indonesia. 3 Department of Ecosystem Studies,<br />
Graduate School of Agricultural and Life Sciences, University of Tokyo, Japan; *<br />
E-mail: <br />
Note: Adapted from: Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo S. and<br />
Parikesit. 2006. Commercialization of homegardens in an Indonesian village: Vegetation<br />
composition and functional changes. Agroforestry Systems (in press).<br />
Keywords: Agricultural transformation, Cash crops, Owner preferences, Plant diversity.<br />
Abstract. With rapid development of Indonesia’s agricultural sector in response to market<br />
pressures, homegardens and other traditional forms of agriculture are increasingly being<br />
transformed into income-generating enterprises through the introduction of cash crops. We<br />
examined the impact of this commercialization on the structure and function of homegardens in the<br />
upland area of the Citarum watershed, West Java, Indonesia, and analyzed the ecological, social,<br />
and economic implications of these changes. Results of a vegetation survey and a survey of 94<br />
respondents indicated plant diversity in commercialized (intensively managed) homegardens<br />
decreased owing to the introduction of commercial crops. The change from subsistence to<br />
commercial farming was accompanied by decreased plant diversity, higher risks, higher external<br />
input use, increased instability, and reduced social equitability. The needs and preferences of the<br />
owners and market pressures were the main factors that triggered the development of intensive<br />
agriculture and increased the commercialization of homegardens. Commercialization adversely<br />
impacted the socio-cultural value that homegardens have traditionally provided to the society.<br />
Likewise, the long-term impacts and sustainability of commercial homegardens are also<br />
uncertain.<br />
233<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 233–250.<br />
© 2006 Springer. Printed in the Netherlands.
234 O.S. ABDOELLAH ET AL .<br />
1. INTRODUCTION<br />
Traditional homegardens have received special attention in Indonesia since the<br />
1970s, when the Institute of Ecology of Padjadjaran University discussed the role of<br />
these homegardens in rural development. Soemarwoto (1987) defined homegardens<br />
as a land surrounding houses in which the structure resembles that of a forest,<br />
combining the natural aspects of a forest with solutions to the socioeconomic and<br />
cultural needs of the people. <strong>Homegardens</strong> are centuries-old components of the rural<br />
ecosystems and are usually cultivated with a mixture of annual and perennial plants<br />
that can be harvested on a daily or seasonal basis.<br />
The structure of homegardens, however, varies from place to place according to<br />
the local physical environment, ecological characteristics, and socioeconomic and<br />
cultural factors (Christanty et al., 1986; Abdoellah, 1990; Karyono, 1990; Ceccolini,<br />
2002; Kumar and Nair, 2004). The high diversity of plant species in these<br />
homegardens and the mixture of annuals and perennials at different heights result in<br />
a complex horizontal and vertical structure. The multi-layered plant canopies prove<br />
to be beneficial in terms of the utilization of sunlight and in terms of water and soil<br />
conservation (Wiersum, 1982; Brownrigg, 1985; Torquebiau, 1992).<br />
<strong>Homegardens</strong> have several functions: economic, social and cultural, esthetic, and<br />
ecological (Abdoellah, 1990; Soemarwoto and Conway, 1991; Wezel and Bender,<br />
2003). In addition, the multiple uses of homegarden products contribute significantly<br />
to meeting the various needs (such as nutrition and income) of the households<br />
(Abdoellah and Marten, 1986; Christanty et t al., 1986; Abdoellah, 1990; Karyono,<br />
1990; Michon and Mary, 1994; Ceccolini, 2002; Blanckaert et al., 2004). Income<br />
derived from homegardens in West Java, for example, ranged from 6.6% to 55.7%<br />
of the family’s total income (Soemarwoto, 1987). The diversity of plants in<br />
traditional homegardens is beneficial from the nutritional point of view as well.<br />
Many of the plants are important sources of non-food necessities such as fuelwood<br />
and building materials too (see Shanavas and Kumar, 2003).<br />
Apart from their economic and ecological functions, rural homegardens also play<br />
important social roles (Abdoellah, 1990; Soemarwoto and Conway, 1991). For many<br />
rural people, the homegarden is an important place for socializing with family and<br />
neighbors. Many homegarden products also have social functions, since it is<br />
common for neighbors to let each other obtain such products freely. Many species<br />
are believed to have “magical” values or to serve as weather indicators. The<br />
homegarden is also an important status symbol; those who do not have their own<br />
homegardens and who must build their homes in another’s homegarden are<br />
considered poor. In the light of these multiple functions, many authors have<br />
concluded that homegardens are sustainable production systems (Karyono, 1990;<br />
Soemarwoto and Conway, 1991; Ceccolini, 2002; Nair, 2001; Wezel and Bender,<br />
2003; Blanckaert ett al., 2004; Kumar and Nair, 2004).<br />
However, during the rapid development of f Indonesia’s agricultural sector in<br />
response to market pressures, commercialization and the adoption of new<br />
technologies have been forcing major changes upon the agroecosystems, and<br />
homegardens are no exception to that general rule (Abdoellah et al., 2001; Kumar<br />
and Nair, 2004). Some villagers are already transforming their homegardens to meet
235<br />
the need for more cash as consumer goods become increasingly available. The<br />
introduction of commercial crops into this system to generate income is a potential<br />
source of structural and functional changes. Coincidentally, some homegardens have<br />
become dominated by few plant species; or have even become similar to<br />
monocultures. Examples include the gardens comprising of cash crops such as<br />
vegetables that are in high demand in urban markets.<br />
We examined the impact of this commercialization on the structure and function<br />
of homegardens in Sukapura village in the upland area of Citarum watershed, West<br />
Java, Indonesia. We addressed the following key questions: Does commercialization<br />
of homegardens affect their structure? Does it affect their social and economic<br />
functions? The answers to these questions will increase our understanding of the<br />
homegardens in relation to the multidimensional socioeconomic, ecological, and<br />
cultural dynamics of the people in this region.<br />
2.1. Study site<br />
2. METHODS<br />
The study was conducted in the Upper Citarum watershed, West Java, Indonesia<br />
during 2000 and 2001. With a total catchment area of approximately 6000 km 2 , the<br />
watershed covers seven districts, and its main river, the Citarum, runs approximately<br />
350 km northward from Mount Wayang to the Java Sea. This watershed, particularly<br />
in the upper part, has been experiencing rapid agricultural development since the<br />
1970s (after the Green Revolution), which caused major changes in its agricultural<br />
landscape, with a strong trend towards homogenization.<br />
Sukapura village, the study site, is located about 30 km southeast of Bandung<br />
Municipality and 20 km to the Majalaya sub-district, a center of the textile industry,<br />
in the Upper Citarum river basin. Most families in Sukapura depend on agriculture<br />
with little influence of urbanization. A major share of the total land area of the<br />
village (163.5 ha out of the administrative area of 187 ha) is devoted to agriculture<br />
consisting of cash crop gardens and mixed gardens. Being located at about 1250 m<br />
above sea level, the climate of the village is slightly cooler than in the lower part of<br />
the watershed and thus more suitable for cultivation of leafy vegetable such as<br />
Chinese cabbage (Brassica sinensis) and green onion ( (Allium fistulosum).<br />
The soil,<br />
which is an Andosol, volcanic in origin, is well drained and quite fertile. The village<br />
also has easy access, with an asphalt road, to nearby urban centers (Majalaya and<br />
Bandung), which allows the villagers to easily market their agricultural products.<br />
Because of these, cash crop cultivation in homegardens is nowadays quite common<br />
in this village. Thus, we considered Sukapura village as one of “typical” places that<br />
have the potential for homegarden commercialization.<br />
2.2. Sampling design<br />
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
We interviewed respondents by using a standard questionnaire. In addition, we<br />
conducted a vegetation survey to characterize the composition and structure of the<br />
homegardens.
236<br />
Sample selection: We determined the required number of homegardens by the<br />
following formula of Lynch et al. (1974):<br />
n =<br />
NZ 2 p(1– p)<br />
Nd 2 +Z 2 p p(1– p)<br />
Where<br />
n = number of samples,<br />
N = number of households in the study village,<br />
Z = the value of the normal variable (1.96) for a confidence level of 0.95,<br />
p = the highest possible proportion (0.5), and<br />
d = the sampling error (0.1).<br />
Using the above formula, we randomly selected 94 households out of 3433 for<br />
interviews and for our vegetation survey. This total number of households was based<br />
on data obtained from the village office. In fact, landlessness in the study village<br />
was very high (>50%). Based on preliminary interviews with the 94 landowners,<br />
we defined the homegardens as commercial (if more than half of the products from<br />
the homegarden were sold for cash) or non-commercial (if more than half of the<br />
products were consumed by the family). Fifty-nine homegardens were thus found to<br />
be non-commercial and 35 commercial homegardens. We also collected data on<br />
household profiles, including main occupation, income from the homegardens,<br />
resources used as inputs, and the presence or absence of livestock, fences, and<br />
buruan (places in front of the house used for socializing and as playgrounds) by<br />
conducting interviews and through direct observation. Regarding income, the data<br />
obtained on annual and currency bases (Indonesian Rupiah: IDR) were converted<br />
into the value equivalent to rice weight at the rate of 1250 IDR per kg rice, which<br />
was the average of selling rate for the variety being cultivated in the village during<br />
2001.<br />
Vegetation survey: For the vegetation survey, we recorded the following data:<br />
species name, number of individuals of each species per plot/farm, number of<br />
structural layers based on plant height, and the plant category based on the main use.<br />
Vegetables were categorized into cash crops (for sale) and subsistence foods (for<br />
own consumption). Land utilization in the homegardens for nursery for plants and<br />
for growing cash crops was also recorded.<br />
To describe the dominance of a given species, we calculated the summed<br />
dominance ratio (SDR; Numata, 1966) for each species. This index was based on the<br />
density and frequency of the species. We also calculated plant diversity using the<br />
Shannon–Wiener diversity index and Pielou’s evenness index (Magurran, 1988).<br />
2.3. Data analysis<br />
O.S. ABDOELLAH ET AL .<br />
The vegetation and interview data for each homegarden were summarized, and the<br />
differences between commercial and non-commercial homegardens were compared<br />
using non-parametric tests in the SPSS for Windows software, Version 10.0 (SPSS
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
237<br />
Inc., Chicago, Illinois, USA). The Mann–Whitney U-test was used for the number of<br />
species and individuals, diversity and evenness indexes, area of the homegarden,<br />
ownership of other agricultural land, and income from the homegarden. For a<br />
bivariate comparison between the types of homegardens and the presence or absence<br />
of fences, livestock, and buruan, the use or non-use of external inputs, and either<br />
off-farm activities or on-farm activities as the main occupation, the Chi-square test<br />
was used.<br />
3.1. Structure of homegardens<br />
3. RESULTS<br />
The size of the homegardens averaged 341.73 m 2 , but commercial homegardens<br />
were larger than the non-commercial gardens (Table 1). The correlation between the<br />
number of species and the size of the homegarden was, however, poor for both types<br />
(Fig. 1a). The number of individual plants tended to increase with increasing size of<br />
the commercial homegardens, but not for the non-commercial ones (Fig. 1b).<br />
Table 1. Plant diversity parameters in commercial and non-commercial homegardens in<br />
Sukapura village, West Java, Indonesia.<br />
Structural attributes Commercial Non-commercial<br />
Area (m<br />
homegardens (n = 35) homegardens (n = 59)<br />
2 )<br />
Average 461.54 270.66<br />
Range<br />
Number of species<br />
120 – 2000 85 – 1400<br />
Total 145 181<br />
Average 15.71 15.37<br />
Range<br />
Number of all plants<br />
4 – 49 4 – 41<br />
Total 42952 3893<br />
Average 1227.20 65.98<br />
Range<br />
Shannon–Wiener diversity index<br />
95 – 8388 6 – 159<br />
Average 1.11 2.03<br />
Range<br />
Pielou’s evenness index<br />
0.16 – 2.00 0.96 – 3.12<br />
Average 0.42 0.78<br />
Range 0.07 – 0.86 0.39 – 0.95<br />
The numbers of species and individual plants in the commercial and noncommercial<br />
homegardens are shown in Table 1. The total number of species found<br />
in both types of homegarden was 127 (out of 199 species; data not presented). There
238<br />
O.S. ABDOELLAH ET AL .<br />
was no significant difference in the number of species per homegarden between the<br />
commercial and non-commercial gardens (Fig. 1a and Table 1); however, there was<br />
significant difference in the number of individuals between the two categories<br />
(U- U test, p
239<br />
between the number of individuals and area particularly in the commercial ones. A<br />
comparison of the area density (individuals/ha) between the two types of<br />
homegardens, showed that the average number in commercial ones was significantly<br />
higher than that in the non-commercial ones (27 154 and 3486 individuals/ha<br />
respectively; U-test, p < 0.01). The average Shannon–Wiener diversity and evenness<br />
indexes in commercial homegardens were significantly lower than that in the noncommercial<br />
homegardens (U-test, p < 0.01). The five most-dominant species in the<br />
commercial homegardens were vegetables (Table 2). Green onion ( (Allium<br />
fistulosum) had by far the highest SDR among these species. In the non-commercial<br />
homegardens Duranta erecta was dominant because of its use as a living hedge<br />
surrounding the homegardens. The numbers of species and their SDR values suggest<br />
that although the number of species did not differ significantly between homegarden<br />
types, the species distribution differed.<br />
Table 2. The five most-dominant species (based on the summed dominance ratio) in the<br />
commercial and non-commercial homegardens in Sukapura village, West Java, Indonesia.<br />
Type of<br />
homegarden<br />
and species<br />
rank order<br />
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
Dominant species Relative<br />
density<br />
Relative<br />
frequency<br />
Summed<br />
dominance<br />
ratio<br />
Commercial<br />
1 Allium fistulosum 39.27 3.27 21.27<br />
2 Daucus carota 12.15 2.00 7.07<br />
3 Ipomoea batatas 10.48 2.00 6.24<br />
4 Brassica sinensis 9.87 0.55 5.21<br />
5 Raphanus sativus 8.38 0.36 4.37<br />
Non-commercial<br />
1 Duranta erecta 15.75 3.64 9.69<br />
2 Manihot esculenta 4.98 1.87 3.43<br />
3 Psidium guajava 3.39 3.31 3.35<br />
4 Alternanthera philoxeroides 4.44 1.43 2.94<br />
5 Musa paradisiaca 3.06 2.32 2.69<br />
Based on the main use of each species, we defined eight plant categories in all<br />
sampled homegardens: vegetable, ornamental, food, fruit, spice, medicinal, building<br />
material, and “other” (Table 3). In commercial homegardens, vegetables were<br />
dominant, whereas ornamental plants were dominant in the non-commercial<br />
gardens. Table 4 presents the proportion of individuals in each plant category as a<br />
function of the size of the homegarden and it shows that the proportion of vegetables<br />
was highest for all sizes of commercial homegardens. These figures suggest that<br />
villagers who used homegardens for commercial purposes did so regardless of the<br />
size of the gardens. In addition, 65% to 93% of the total area of the commercial
240<br />
homegardens was planted with vegetable crops (data not shown). In contrast, in the<br />
non-commercial homegardens, inedible ornamental plants were dominant (Table 3).<br />
The relative proportion of the number of individuals in each plant category did not<br />
seem to be related to the size of the homegarden, but ornamental plants occupied the<br />
highest percentage for all size categories (Table 4). Even though the average number<br />
of individuals of ornamental plants was not different between the commercial and<br />
non-commercial homegardens, there were pronounced variations in this respect<br />
concerning vegetables and food crops (Table 4).<br />
Table 3. Dominance ratios of the main categories of plants in commercial and noncommercial<br />
homegardens in Sukapura village, West Java, Indonesia.<br />
Plant category Summed dominance ratio<br />
Commercial Non-commercial<br />
Vegetable 44.30 9.61<br />
Ornamental 23.63 56.51<br />
Food 14.53 7.85<br />
Fruit 11.30 16.76<br />
Spice 1.80 3.06<br />
Medicinal 1.35 2.46<br />
Building 1.25 1.52<br />
Other 1.84 2.23<br />
Total 100.0 100.0<br />
In terms of growth form, 88.6% of the individual plants in commercial<br />
homegardens occupied the first (ground) strata of the vegetation structure and were<br />
shorter than 1 m tall (Fig. 2). Of this, 90.1% comprised commercial crops such as<br />
Allium fistulosum, D. carota, Ipomoea batatas, Brassica sinensis, and Raphanus<br />
sativus. Figure 2a also indicates that the non-commercial homegardens kept the<br />
multistrata structure better than the commercial gardens.<br />
3.2. Functions of homegardens<br />
O.S. ABDOELLAH ET AL .<br />
In general, homegarden functions depended on their species composition. In the<br />
commercial homegardens, the choice of species is determined largely by market<br />
demands. The number of respondents conducting off-farm activities as the main<br />
occupation was about 22% in the non-commercial category and about 11% for the<br />
commercial-homegarden owners; albeit t the differences were not significant (Table<br />
5; Chi-square test, p = 0.20). Moreover, based on the area of other agricultural lands<br />
owned by farmers, there was no difference between the types of the homegardens<br />
(U-test, U p > 0.05; n = 31 for the commercial homegardens and n = 46 for the noncommercial<br />
class).
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
241<br />
Figure 2. Vertical structural differences between the commercial and non-commercial<br />
homegardens in Sukapura Village, West Java, Indonesia. (a) Relative proportion of the<br />
number of species in each story of the vegetation structure to the total number of species. (b)<br />
Proportion of the total number of individuals in each story.<br />
Table 5 also shows the income derived from homegardens. The annual income<br />
from commercial homegardens was significantly higher than that from noncommercial<br />
homegardens (14 553 versus 2467 kg rice equivalent per ha). It is<br />
interesting to note that income per unit area in each sample was almost similar<br />
among the commercial gardens, but it varied among the non-commercial<br />
homegardens. The actual income in each sample (kg rice per year) had a significant<br />
and positive correlation with the area of the gardens only for the commercial<br />
category (Table 6). It was also significantly correlated with the number of<br />
individuals of all species, as well as the numbers of vegetable species, timber<br />
species (producing building materials), and food plants in commercial homegardens<br />
(Table 6). Although there was significant correlation between the income (kg rice<br />
per year) and the number of plants in non-commercial homegardens, it was only<br />
correlated with the number of fruit- and food plants (Table 6). These results imply<br />
that the main sources of income in the commercial homegardens were commercial
Table 4. Proportion of the individual plants in each of the eight plant categories as a function of the size of the homegardens in Sukapura village,<br />
West Java, Indonesia.<br />
Type of<br />
homegardens and<br />
area classes<br />
n<br />
Proportion of the number of individuals in each plant category (%)<br />
Vegetable Ornamental Food Fruit Spices Medicinal Building<br />
material<br />
Commercial<br />
< 100 m 2 0 – – – – – – – –<br />
101–200 m 2 7 45.9 (295) 23.2 (149) 25.6 (164) 1.5 (10) 0.6 (5) 0.2 (1) 0.0 (1) 3.0 (19)<br />
201–300 m 2 11 69.9 (410) 7.2 (43) 18.3 (108) 3.1 (19) 0.4 (3) 0.7 (5) 0.2 (1) 0.2 (1)<br />
301–400 m 2 5 81.8 (1061) 4.1 (54) 11.2 (145) 2.0 (26) 0.0 (1) 0.1 (1) 0.4 (6) 0.5 (7)<br />
401–500 m 2 5 74.2 (313) 15.5 (66) 6.8 (29) 2.9 (13) 0.0 (1) 0.2 (1) 0.1 (1) 0.2 (1)<br />
> 501 m 2 7 73.8 (2470) 2.3 (79) 22.6 (758) 0.8 (28) 0.0 (1) 0.0 (1) 0.4 (15) 0.0 (1)<br />
Non-commercial<br />
< 100 m 2 7 11.5 (6) 66.9 (35) 1.1 (1) 16.1 (9) 2.2 (2) 0.3 (1) 0.3 (1) 1.6 (1)<br />
101–200 m 2 24 5.3 (4) 60.3 (36) 12.4 (8) 15.5 (10) 0.8 (1) 1.2 (1) 2.1 (2) 2.4 (2)<br />
201–300 m 2 8 13.6 (10) 62.2 (44) 10.1 (8) 6.7 (5) 1.6 (2) 4.2 (3) 1.1 (1) 0.5 (1)<br />
301–400 m 2 7 5.9 (5) 57.5 (44) 14.3 (11) 16.4 (13) 2.5 (2) 1.0 (1) 0.4 (1) 2.1 (2)<br />
401–500 m 2 7 16.4 (13) 46.6 (35) 19.3 (15) 7.4 (6) 4.2 (4) 4.0 (3) 0.6 (1) 1.5 (2)<br />
> 501 m 2 6 7.6 (7) 66.6 (55) 1.0 (1) 12.1 (10) 10.5 (9) 0.6 (1) 1.6 (2) 0.0 (0)<br />
n = number of homegardens; parenthetic values are the average number of individuals per homegarden.<br />
Other<br />
242 O.S. ABDOELLAH ET AL .
243<br />
Table 5. Differences in socioeconomic data for the owners of commercial and noncommercial<br />
homegardens in Sukapura village, West Java, Indonesia.<br />
Socioeconomic attributes<br />
Type of homegarden<br />
Commercial<br />
(n = 35)<br />
Ownership a of agricultural lands (m 2 )<br />
Paddy field 23.57<br />
(0 – 700)<br />
Crop field 881.20<br />
(0 – 1000)<br />
Mixed garden 116.00<br />
(0 – 2100)<br />
Total agricultural lands (area) 1020.77<br />
(0 – 11600)<br />
Income from homegarden b<br />
(kg rice equivalent ha –1 yr –1 ) (13757 – 14960)<br />
Off-farm activities as the main<br />
occupation c<br />
Use of external inputs in the<br />
homegarden c<br />
Existence of fences around the<br />
homegarden c<br />
Raising livestock in the<br />
homegarden c<br />
Presence of buruan in the<br />
homegarden c<br />
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
Noncommercial<br />
(n = 59)<br />
90.68<br />
(0 – 2400)<br />
653.05<br />
(0 – 6000)<br />
28.98<br />
(0 – 840)<br />
772.71<br />
(0 – 6000)<br />
Statistical<br />
significance d<br />
NS<br />
NS<br />
NS<br />
NS<br />
14565 2467<br />
(144 – 12318)<br />
**<br />
4 13 NS<br />
33 16 **<br />
29 13 **<br />
7 42 **<br />
10 51 **<br />
a 2<br />
Mean followed by range in parentheses (m ).<br />
b –1<br />
Mean followed by range in parentheses (kg rice equivalent ha a yr –1<br />
r ).<br />
c<br />
Number of respondents doing off-farm activities as the main occupation, using external<br />
inputs such as chemical fertilizers and pesticides in their homegardens, having fences around<br />
their homegardens, raising livestock in their homegardens and having a buruan in front of<br />
their homegardens.<br />
d<br />
For comparing commercial and non-commercial homegardens, we used the Mann–Whitney<br />
U-test for the three types of ownership of agricultural lands and for income from the<br />
homegarden and Chi-square test—for external inputs, existence of fences and livestock, and<br />
presence of a buruan; ** p < 0.01; NS = no significant difference or correlation.<br />
crops such as vegetables and timber species while in the non-commercial<br />
homegardens it was the fruit-producing species. This relatively higher income from<br />
the commercial homegardens reflected the change in function of homegardens from<br />
subsistence to commercial purposes. Based on our observation, the owners of
244<br />
O.S. ABDOELLAH ET AL .<br />
commercial homegardens managed their homegardens much more intensively, for<br />
example, by routinely watering the homegarden plants and using external inputs<br />
such as chemical fertilizers and pesticides (Table 5). Thus, commercialization of the<br />
homegardens increased the demand for external inputs. Almost all respondents with<br />
commercial homegardens (94.3%) used these inputs to enhance crop yields and to<br />
protect crops from pests. There was a significant correlation between the use of<br />
external inputs and the type of homegarden: most commercial gardens used those<br />
inputs, while very few non-commercial gardens did.<br />
Table 6. Pearson’s correlation coefficient between the annual income from the<br />
homegardens and some homegarden characteristics in Sukapura village, West Java,<br />
Indonesia.<br />
Correlation coefficient to the annual<br />
income (kg rice equivalent yr –1 Homegarden characteristics<br />
)<br />
Commercial Non-commercial<br />
homegardens homegardens<br />
(n = 35)<br />
(n = 59)<br />
Size of homegarden<br />
Number of individuals<br />
1.00** –0.09<br />
Total 0.77** 0.31*<br />
Vegetable 0.72** 0.18<br />
Ornament 0.11 0.04<br />
Food 0.53** 0.26*<br />
Fruit 0.14 0.29*<br />
Spice –0.21 0.24<br />
Medicinal –0.06 0.15<br />
Building material 0.60** 0.05<br />
Other –0.15 –0.17<br />
To protect the commercial homegardens, 82.9% owners established fences,<br />
although the fences did not completely enclose the homegardens. In contrast, 78% of<br />
the owners of non-commercial homegardens did not establish fences (Table 5). In<br />
addition, 80% of the owners of commercial homegardens did not raise animals such<br />
as chickens, goats, and sheep in their homegardens, partly because of lack of space<br />
to raise the animals and build livestock pens, and partly because of the desire to<br />
protect their cash crops from grazing animals. Conversely, 71.2% of the owners of<br />
non-commercial homegardens raised livestock, and this difference was significant.<br />
Furthermore, 71.4% of commercial homegardens lacked a buruan where children<br />
could play in front of the house. The main reason for the decision not to create a<br />
buruan was, again, the lack of space and the worry that the children might damage<br />
the crops. In contrast, 86.4% of the non-commercial homegardens had a buruan, and<br />
this difference was significant.
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
4. DISCUSSION<br />
245<br />
Although the average number of species present did not differ significantly between<br />
the types of homegardens and many species were planted in both types, floristic<br />
composition of commercial homegarden was characterized by an increasing number<br />
of individuals of cash crops (vegetables) and a significantly decreasing diversity<br />
index. Implicit in this is that owners of both types of homegardens desired variety in<br />
products for both self-consumption and for sale, but that the latter goal probably<br />
outweighed the former for the owners of commercial homegardens.<br />
The total number of species found in all sampled homegardens in Sukapura did<br />
not differ from the results of the previous studies conducted in the lower part of the<br />
Citarum watershed. However, the dominant species in the present study (and<br />
especially those in the commercial homegardens) differed strongly from those of the<br />
previous studies. For example, in a study conducted by Chistanty et al. (1986), the<br />
homegardens greatly resembled the non-commercial homegardens of the present<br />
study, which were dominated by ornamental plants and had only few cash crops.<br />
This difference may have been strongly influenced by the specific needs and<br />
preferences of the landowners as well as by the different climatic and edaphic<br />
factors prevailing in the lower parts of the watershed. The fertile and well-drained<br />
soils and cooler climate of the upper watershed (present study) could have<br />
encouraged the local farmers to intensify the land use, including homegardening.<br />
The low correlation between the number of species and size of the homegardens<br />
in the present study suggests that homegarden size is probably not the main factor<br />
that governs species diversity. Instead, the structure and composition of the<br />
homegardens depended most likely on the role of various species required to fulfill<br />
the owner’s cultural, nutritional, social, and economic needs. For example, the fact<br />
that buruan were far more common in the non-commercial homegardens suggests<br />
that these landowners gave a high priority to the social and cultural roles<br />
traditionally supported by homegardens.<br />
Unlike in rural areas located at lower altitudes, the structure of the commercial<br />
homegardens in the present study was characterized by a more complex lower<br />
canopy, which, for example, was different from that described by Karyono (1990).<br />
In our study, some homegardens were dominated by only a few plant species that<br />
occupied the lower layers of the canopy structure, and some had even become<br />
monocultures, with the dominant species comprising cash crops such as vegetables<br />
that were usually found in the lowest layer (less than 1 m tall; 88.6% of the total).<br />
Interestingly, plants were more evenly distributed throughout the vertical structure<br />
of the non-commercial gardens. This indicates the presence of a multistrata canopy<br />
structure in most of these homegardens, as has been suggested by many others too<br />
(Karyono, 1990; Soemarwoto and Conway, 1991; Michon and Mary, 1994;<br />
Blanckaert et al., 2004). However, there were both inter-site and intra-site variations<br />
that complicate this inference. The specific needs and preferences of the owners<br />
were clearly important factors that influence the structure and the number of strata<br />
that were preserved or created in the homegardens (De Clerck and Negreros-<br />
Castillo, 2000).
246<br />
O.S. ABDOELLAH ET AL .<br />
Many authors have pointed out that the structural pattern of the vegetation cover<br />
is influenced by specific physical circumstances, ecological characteristics,<br />
economics, and social and cultural factors (Christanty et al., 1986; Abdoellah, 1990;<br />
Karyono, 1990; Soemarwoto and Conway, 1991; Wezel and Bender, 2003). Although<br />
the landowners in our study were living under similar biophysical conditions, the<br />
structural pattern of the vegetation cover differed between the commercial and noncommercial<br />
homegardens (Fig. 2). Given the high degree of variation among gardens<br />
(Tables 1 and 4), there was clearly no single “typical” homegarden. Although tree<br />
species taller than 10 m were found in both types of homegardens, they were clearly<br />
more common in the non-commercial category (Fig. 2). These tree species were<br />
grown by the owners of non-commercial homegardens without distinct spatial<br />
arrangements; these owners grew big trees in any part of the yard and on any side of<br />
the house. It is likely that the owners tried to make the better use of available space<br />
in their homegardens, besides, tree planting is an old custom aimed to fulfill<br />
subsistence needs and, to some extent, to provide a restful micro-environment<br />
around the house. Besides, as the non-commercial homegardens often function as<br />
spots for social activities, the presence of tree canopies providing shade may<br />
facilitate such activities. In contrast, the owners of commercial homegardens mostly<br />
planted such trees in the backyard areas to mark the border of their gardens. It was<br />
very rare to find a tall tree in front of or at the side of a house in a commercial<br />
homegarden where cash crops were planted. According to the owners, this was<br />
because growing a big tree would inhibit their ability to grow commercial vegetable<br />
crops due to excessive shading.<br />
This difference suggests that the structural pattern of the vegetation in the<br />
homegardens was strongly influenced by the specific needs and preferences of the<br />
owners. We assumed that the owner whose main occupation was farming and who<br />
did not have much agricultural lands might commercialize his/her homegarden and<br />
vice-versa. However, there was no significant correlation between the commercialization<br />
of an owner’s homegarden and ownership of other agricultural lands or<br />
the main occupation status (Table 5). Thus, the structure of the homegarden<br />
depended on the owner’s management objectives as has been reported by several<br />
previous workers too (De Clerck and Negreros-Castillo, 2000; Mendez et al., 2001;<br />
Kumar and Nair, 2004).<br />
Many authors, such as Abdoellah (1990) and Soemarwoto and Conway (1991),<br />
have also reported that the increased intensity of cultivation of homegardens and the<br />
domination of these homegardens by particular species has reduced the overall<br />
number of plant species. However, our study showed that the number of species did<br />
not change significantly because of commercialization, but that the diversity index<br />
did indeed decrease, most likely because of the greatly increased number of<br />
individuals of certain species (Table 1). One consequence of the rising demand for<br />
better vegetable crops is that the species evenness has decreased substantially. The<br />
increased reliance on a limited number of species is likely to increase the risk of pest<br />
and disease outbreaks, as has already occurred in the sweet orange (Citrus nobilis)<br />
and clove (Syzygium aromaticum) orchards of Java. Soemarwoto and Conway<br />
(1991) reported that these crops, which were being extensively introduced in the<br />
homegardens, were already being severely damaged by Phyllosticta spp. and by
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
247<br />
citrus vein phloem degeneration disease, respectively. Similar problems have also<br />
been reported by Ceccolini (2002) in the homegardens on Soqotra Island. This<br />
suggests that commercialization of homegardens may eventually create ecological<br />
instability, leading to an increased incidence of pests and diseases.<br />
Furthermore, commercialization of homegardens by focusing on cash crops has<br />
resulted in only short-term improvements in farmers’ incomes. It is, however, not<br />
certain whether the high initial levels of productivity can be sustained. Cash crops<br />
also require high-energy inputs in the form of fertilizers and pesticides (Abdoellah,<br />
1990; Abdoellah et al., 2001). Our study confirmed that the use of these external<br />
inputs is significantly higher in the commercial gardens (Table 5), and that such<br />
increased use of chemical fertilizers and pesticides are inevitable for the commercial<br />
gardeners. Thus, although the gross income is higher, at least in the short term, net<br />
income will increasingly suffer and the long-term stability of this income is also<br />
uncertain, particularly for the products for which market demand fluctuates greatly.<br />
An additional consequence is the need for credit from banks and other sources of<br />
capital. Inadequate credit facilities in the public sector, however, have driven the<br />
villagers to unscrupulous middlemen and moneylenders, potentially leading to future<br />
changes in land ownership, and making the continued existence of somewhat<br />
autonomous homegardens doubtful. This seemed to reflect what has been stated by<br />
Michon and Mary (1994) that apart from high population density, major factors that<br />
threatened the existence of traditional homegardens in West Java were increased<br />
scarcity of agricultural lands, conflicts between commercial agriculture and<br />
traditional food production system, and development of a market economy.<br />
Soemarwoto and Conway (1991) also stated that the income generated from the<br />
sale of homegarden products tended to be used for ceremonies and other forms of<br />
consumption. There is also a danger that the dietary role of homegardens in<br />
providing protein, vitamins, and minerals may be neglected or even lost (Abdoellah<br />
and Marten, 1986; Soemarwoto and Conway, 1991; Wezel and Bender, 2003;<br />
Blanckaert et al., 2004), because traditional vegetables with low commercial value<br />
but high nutritional value may be the first to disappear from the commercial<br />
homegardens. Furthermore, commercialization of these homegardens has led to a<br />
decline in animal husbandry, thereby eliminating another source of nutrition that<br />
might compensate for the loss of these vegetables. These factors, taken together,<br />
undoubtedly decrease the ecological and economic sustainability of the commercial<br />
homegarden production system.<br />
Commercialization of homegardens has eliminated or reduced some of their<br />
multiple functions also. Traditionally, many products such as fruits, vegetables, and<br />
other useful plants were shared within the local communities (Abdoellah, 1990;<br />
Soemarwoto and Conway, 1991), thereby adding a unique social role to these<br />
homegardens (Kumar and Nair, 2004). Commercialization, however, has impeded<br />
this practice and has thus reduced the equitability of farming. Soemarwoto and<br />
Conway (1991) already pointed out that traditionally the Sundanese who live in this<br />
area have abided by the prospect of living harmoniously (rukun) with both relatives<br />
and other members of the community. Soemarwoto and Conway (1991) reported<br />
that an important way of expressing rukun was by offering useful homegarden<br />
products to relatives or neighbors daily, and particularly to the poor or unfortunate
248<br />
O.S. ABDOELLAH ET AL .<br />
who needed this gift to survive, thereby maintaining, and strengthening social<br />
networks. Unfortunately, based on our interviews with several respondents, commercialization<br />
has decreased this sharing, even with relatives, and this has undermined the<br />
community’s social linkages, particularly concerning the poor.<br />
Commercialization of homegardens has forced more owners to establish fences<br />
around their homegardens (Table 5). Although these fences do not completely<br />
enclose the homegarden, they prevent people from entering or passing through<br />
freely, and force these people to request the owner’s permission to enter. This<br />
represents an important negative change from the traditional free access, as there<br />
was originally no concept of trespassing (Soemarwoto and Conway, 1991). This<br />
access has been retained in many non-commercial homegardens, which still mostly<br />
lack fences (Table 5). Most owners of non-commercial homegardens feel that<br />
establishing a fence around a homegarden is socially inappropriate, and that the<br />
owners of completely fenced homegardens are “conceited” (Soemarwoto and<br />
Conway, 1991). Furthermore, commercialization of homegardens has significantly<br />
decreased the number of buruans (Table 5). Children can no longer play in front of a<br />
house that lacks a buruan, thereby removing an important location for socializing<br />
with family and neighbors. Even more seriously, the buruan has traditionally been a<br />
place for children to learn cultural and social values from their elders (Soemarwoto<br />
and Conway, 1991). As a result of decreasing the sharing of products from<br />
homegardens and disrupting the social networks that are encouraged by free passage<br />
through homegardens and the existence of buruan, the commercialization of<br />
homegardens has done serious damage to the social fabric of these communities.<br />
5. CONCLUSIONS<br />
Our results suggest that the homegardens of Sukapura village, in the Upper Citarum<br />
watershed of Indonesia, have changed dramatically over the past two decades. The<br />
ecological characteristics and social roles of these homegardens have been adversely<br />
affected, and the traditional system of sustainable agriculture that has kept people<br />
safe and well fed for centuries may no longer be sustainable without external inputs.<br />
Although income from commercialized homegardens has increased, these gardens<br />
have decreased plant diversity and evenness, heightened the ecological and financial<br />
risks to the owners, increased the requirements for external inputs such as fertilizers<br />
and pesticides, lowered community equitability, and increased overall instability.<br />
To revitalize the traditional functions of homegardens, we must convince the<br />
owners that the complex vegetation structure of these homegardens is more<br />
advantageous in the long-term, than the simpler and less stable structures of the<br />
commercial homegardens. In order not to go to the dangerous extent of full<br />
commercialization, heavily relying on external inputs as occurring in the research<br />
site, efforts should be made to improve the economic functions of the homegardens<br />
by manipulating their species composition. To succeed in this endeavor, a detailed<br />
analysis of the plant associations in traditional homegardens is required. This will<br />
provide a better knowledge of the ecological and economic compatibility of various<br />
plant species. The perceptions of landowners related to the preferred plant species<br />
must also change to reflect these findings, leading to new planting patterns based on
COMMERCIALIZATION OF HOMEGARDENS IN INDONESIA I<br />
249<br />
improved selection of species, based on both their ecological roles and economic<br />
potential. Improved homegarden designs should also consider integrating crop-based<br />
activities with animal husbandry, both of which are crucial “social capital” for<br />
sustaining traditional homegardens and permitting future development. Post-harvest<br />
technology related to the products of these homegardens should also be investigated so<br />
as to reveal opportunities to add value to the products and create jobs that generate<br />
income without undermining the sustainability of the homegardens. Finally,<br />
supportive regional land use planning and management policies must be developed<br />
to encourage landowners to maintain the structure and function of traditional<br />
homegardens.<br />
ACKNOWLEDGMENTS<br />
This research was supported by the Institute of Ecology and the Research Institute of<br />
Padjadjaran University, and the Core University Program in Applied Biosciences<br />
funded by the Japanese Society for the Promotion of Science and the Directorate<br />
General of Higher Education, Indonesian Ministry of Education and Culture (Grantin-Aid<br />
for Scientific Research Category B #16405037, JSPS). The authors thank<br />
their students: Luppy Handinata, Deyna Handiyana, Dendi Muhamad, and Fazar R.<br />
Zulkarnaen, for assistance in fieldwork. This article was written while the first<br />
author was a visiting professor at the Laboratory of Landscape Ecology and<br />
Planning, Department of Ecosystem Studies, Graduate School of Agricultural and<br />
Life Sciences, University of Tokyo, Japan.<br />
REFERENCES<br />
Abdoellah O.S. 1990. <strong>Homegardens</strong> in Java and their future development. In: Landauer K.<br />
and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 69 – 79. United Nations University Press,<br />
Tokyo.<br />
Abdoellah O.S. and Marten G.G. 1986. The complementary roles of homegardens, uplands<br />
fields, and rice fields for meeting nutritional needs in West Java. In: Marten G.G. (ed.),<br />
Traditional agriculture in Southeast Asia: A human ecology perspective, pp 293 – 325.<br />
West view Press, Boulder and London.<br />
Abdoellah O.S., Takeuchi K., Parikesit, Gunawan B. and Hadikusumah H.Y. 2001. Structure<br />
and function of homegarden: revisited. In: Proceedings of First Seminar of JSPS-DGHE<br />
Core University Program in Applied Biosciences “Toward harmonisation between<br />
development and environmental conservation in biological production” (21–23 February<br />
2001), pp 167–185. University of Tokyo, Tokyo.<br />
Blanckaert I., Swennen R.L., Paredes Flores M., Rosas López R. and Lira Saade R. 2004.<br />
Floristic composition, plant uses and management practices in homegardens of San Rafael<br />
Coxcatlan, Valley of Tehuacan-Cuicatlan, Mexico. J Arid Environ 57: 179 – 202.<br />
Brownrigg L. 1985. Home gardening in international development: what literature shows?<br />
The League for International Food Education, Washington, DC, 330p.<br />
Ceccolini L. 2002. The homegardens of Soqotra islands, Yemen: an example of agroforestry<br />
approach to multiple land use in an isolated location. Agroforest Syst 56: 107 – 115.<br />
Christanty L., Abdoellah O.S., Marten G. and Iskandar J. 1986. Traditional agroforestry in<br />
West Java: the pekarangan (homegarden) and kebun-talun (perennial/annual rotation)
250<br />
O.S. ABDOELLAH ET AL .<br />
cropping systems. In: Marten G.G. (ed.), Traditional agriculture in Southeast Asia: A<br />
human ecology perspective, pp 132 – 156. Westview Press, Boulder and London.<br />
De Clerck, F.A.J. and Negreros-Castillo P. 2000. Plant species of traditional Mayan<br />
homegardens of Mexico as analogs for multistrata agroforests. Agroforest Syst 48:<br />
303 – 317.<br />
Karyono 1990. Home gardens in Java: their structure and function. In: Landauer K. and Brazil<br />
M. (eds), <strong>Tropical</strong> home gardens, pp 138 – 146. United Nations University Press, Tokyo.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Lynch F., Hollnsteiner M.R. and Corvar L.C. 1974. Data gathering by social survey. Trial<br />
edition. Social Science Council Inc., the Philippines, Quezon City, 227p.<br />
Magurran E.A. 1988. Ecological diversity and its measurement. Princeton Univ. Press, New<br />
Jersey, 179p.<br />
Mendez V.E., Lok R. and Sommarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Michon G. and Mary F. 1994. Conversion of traditional village gardens and new economic<br />
strategies of rural households in the area of Bogor, Indonesia. Agroforest Syst 25:<br />
31 – 58.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Numata M. 1966. Ecological judgment of grassland condition and trend. II: Judgment by<br />
floristic composition (in Japanese with English abstract). J Jpn Soc Grassland Sci 12:<br />
29 – 36.<br />
Shanavas A. and Kumar B.M. 2003. Fuelwood characteristics of tree species in homegardens<br />
of Kerala, India. Agroforest Syst 58: 11 – 24.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: a traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development,<br />
pp 157 – 170. ICRAF, Nairobi.<br />
Soemarwoto O. and Conway G.R. 1991. The Javanese homegarden. J Farming Syst Res Extn<br />
2: 95 – 118.<br />
Torquebiau E. 1992. Are tropical homegardens sustainable? Agric Ecosyst Environ 41:<br />
189 – 207.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
Wiersum K.F. 1982. Tree gardening and taungya in Java: examples of agroforestry<br />
techniques in the humid tropics. Agroforest Syst 1: 53 – 70.
CHAPTER 14<br />
TRANSPIRATION CHARACTERISTICS<br />
OF SOME HOMEGARDEN TREE SPECIES<br />
IN CENTRAL SRI LANKA<br />
W.A.J.M. DE COSTA*, K.S.P. AMARATUNGA,<br />
AND R.S. UDUMULLAGE<br />
Department of Crop Science, Faculty of Agriculture, University of Peradeniya,<br />
Peredeniya 20400, Sri Lanka; *E-mail: <br />
Keywords: Artocarpus heterophyllus, Cedrela toona, Radiation interception, Soil water<br />
deficit, Swietenia macrophylla, Vapor pressure deficit.<br />
Abstract. Deep-rooted trees that dominate the multilayered homegardens (MHG) in Central Sri<br />
Lanka might adversely impact the catchment water yield because of their high transpiration rates.<br />
Our objectives were to quantify the water use of three representative tree species in an MHG and to<br />
identify the major determinants of transpiration. The species were Artocarpus heterophyllus<br />
(diameter at breast height, DBH = 40.5 cm), Cedrela toona (DBH = 9 cm) and Swietenia<br />
macrophylla (DBH = 3 cm) representing the upper, middle and lower canopy layers respectively of<br />
an MHG in the high-rainfall zone of central Sri Lanka. Transpiration was measured as trunk sap<br />
flow rate using thermal dissipation probes in Artocarpus s and Cedrela and sap flow gauges in<br />
Swietenia. Measurements during a 72 h period, when soil moisture was not limiting, showed that<br />
sap flow of Artocarpus s was significantly greater than those of Cedrela and Swietenia. Daily<br />
transpiration of Cedrela and Swietenia ranged from 19% to 27% of that in Artocarpus and it<br />
increased linearly with incident solar radiation and saturation vapor pressure deficit (VPD).<br />
Measurements during a 54-day rainless period also showed that Artocarpus s and Cedrela had high<br />
transpiration rates despite reduced water availability in the top 1 m soil layer, indicating water<br />
extraction by roots from deeper horizons. Transpiration rate increased with increasing irradiance up<br />
to 13 MJ m –2 2 –1<br />
m d and with increasing VPD up to 0.8 kPa. Decreases in transpiration at irradiances<br />
and VPDs greater than the above values indicated that stomata had begun to exert significant<br />
control over water loss from the foliage canopy.<br />
1. INTRODUCTION<br />
Multi-layered homegardens (MHG) are a common agroforestry system, which<br />
covers a considerable part of the Central Province of Sri Lanka. It is the<br />
251<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 251–267.<br />
© 2006 Springer. Printed in the Netherlands.
252<br />
W.A.J.M. DE COSTA ET AL .<br />
predominant land use system in the Kandy district of Central Sri Lanka, where this<br />
traditional system of perennial cropping has been practiced for several centuries, and<br />
hence known as the ‘Kandyan forest gardens’ (Jacob and Alles, 1987). The MHGs<br />
consist of a mixture of trees of varying heights, canopy spreads, life cycle durations,<br />
and uses. The high diversity of plants in a Kandyan MHG offers economic stability<br />
to farmers because of the wide range of economically important products obtained<br />
from them such as food, beverage and fruits, spices and timber 1 .<br />
The predominantly perennial vegetation in MHGs could absorb a significant<br />
amount of water from the soil through their deep root systems. This water is<br />
ultimately transpired from the foliage after it is translocated through the xylem<br />
system. Therefore, water use by MHGs forms an important consideration in the<br />
catchment water balance of the Central Province of Sri Lanka, which is the most<br />
important rainfall catchment on which much of the Sri Lankan agriculture and<br />
power generation depend on. Catchment water balance of this region thus exerts a<br />
critical influence on the national economy. Strong evidence available from different<br />
parts of the world suggests that high rates of transpiration by forests and other taller,<br />
perennial vegetation could lead to decreased water yields in rivers and reservoirs<br />
(Calder, 1996).<br />
Following an analysis of 94 forest catchment experiments in different parts of<br />
the world across a range of climates, Bosch and Hewlett (1982) concluded that a<br />
10% increase in forest cover would decrease annual water yield by 40 mm for Pinus<br />
and Eucalytpus and by 25 mm for deciduous hardwood forests. Although such<br />
reports generally predict reduced streamflow and ground water availability in areas<br />
planted with Pinus and Eucalytpus, it could be argued that such reductions may not<br />
occur in the humid tropics because of their high rainfall. This question has been<br />
frequently raised in Sri Lanka and based on limited and incomplete data 2,3 , it was<br />
concluded that water use of forests and taller perennial vegetation in the Central<br />
Province was greater than that of grasslands and annual cropping systems.<br />
Quantification of water use of MHGs and determination of its impact on catchment<br />
water balance are difficult because of the complex and diverse vegetation structure<br />
of MHGs. The total water use of an MHG would be the sum of transpiration rates of<br />
different tree species whose canopies are arranged into different vertical layers<br />
(Jacob and Alles, 1987). As a first step in quantifying the total water use of MHGs,<br />
transpiration rates of a limited number of trees representing different vertical layers<br />
in the canopy structure was measured. Our specific objective was to relate the<br />
measured transpiration rates to the microenvironmental factors in the relevant<br />
canopy layers. Developing mechanistic relationships between transpiration and its<br />
driving environmental factors could pave the way to estimate water use of MHGs<br />
through an adequately comprehensive modeling approach.<br />
Transpiration rates depend on soil water availability (Monteith, 1986; Jarvis and<br />
McNaughton, 1986; Wallace, 1996); and in the absence of soil water deficits it is<br />
primarily driven by microenvironmental factors such as incident solar radiation and<br />
vapour pressure deficit. With increasing soil water deficits, stomata may exert a<br />
significant control over transpiration by partial closure. Therefore, a secondary<br />
objective of our study was to see whether stomata could exert an appreciable control<br />
over water use of the MHGs during rainless periods of gradually increasing soil
253<br />
water deficits. Considering the widely predicted reductions in rainfall and water<br />
availability (McCarthy et al., 2001), this information would be relevant to the<br />
discussion on sustainability of MHGs.<br />
2.1. Experimental site<br />
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
2. MATERIALS AND METHODS<br />
The study was conducted from June 2001 to February 2002 in a typical multilayered<br />
homegarden in Kandy (7 o 17’N, 80 o 40’E), in the central highlands of Sri Lanka. The<br />
site is in the mid-elevational (500 m above sea level), high rainfall (2000 mm yr –1 )<br />
zone, which is known as the Mid-country Wet Zone, WM2, according to the<br />
classification of agro-ecological zones of Sri Lanka by Panabokke (1996), with a<br />
mean daily temperature in the range of 25 to 27 o C and relative humidity between 70<br />
to 90%.<br />
This agro-ecological zone contains gently to steeply undulating terrain consisting<br />
of hills and valleys. Multilayered homegardens are located in the hills while the<br />
valleys are cultivated with lowland rice (Oryza sativa). Soil in the hills is deep and<br />
well-drained, dark reddish brown with a sandy clay loam texture; it is classified as<br />
‘Reddish Brown Latosolic Soils’ according to the local classification (Panabokke,<br />
1996), Dystric Cambisols by FAO/UNESCO and Typic Troporthents by USDA<br />
(Senarath and Dassanayake, 1999). Bulk density ranges from 1.50 x 10 3 to 1.60 x<br />
10 3 kg m –3<br />
m and pH from 5.0 to 5.5 throughout the soil profile. Available water, i.e.<br />
soil water content between field capacity (–0.01 MPa of soil water potential) and<br />
permanent wilting point (–1.5 MPa) is 111.4 mm m –1<br />
m .<br />
2.2. Composition of the homegarden<br />
The area of the MHG selected for this study, including the house within, was 0.15<br />
ha. The house was surrounded by vegetation consisting mainly of trees varying in<br />
heights, canopy spread and depth, trunk diameter and age. The woody perennials at<br />
the study site were enumerated by measuring their height and diameter at breast<br />
height (DBH). The probable age of trees was ascertained from the garden owner.<br />
The MHG had 56 trees belonging to 14 species (Table 1), most of which were of<br />
economic value providing food (Artocarpus ( heterophyllus and Cocos nucifera),<br />
beverages (Coffea arabica), fruits (Mangifera indica and Persea americana), spices<br />
(Syzygium aromaticum and Myristica fragrans), timber (Swietenia macrophylla and<br />
Artocarpus heterophyllus) and variety of other products and services.<br />
The trees were categorized into three groups based on their canopy position: top<br />
layer (10 m or taller), middle layer (5 to 10 m), and bottom layer (less than 5 m).<br />
Three representative trees occupying these three vertical layers were selected for<br />
measuring transpiration rates. The species were Artocarpus heterophyllus, Cedrela<br />
toona, and Swietenia macrophylla representing the top, middle, and bottom layers,<br />
respectively. Main attributes of the three species are given in Table 1. The trees were<br />
located within a 15 m radial zone to avoid using excessively long wires conducting
254<br />
voltage signals from sapflow sensors and to have the data-logger in a central<br />
location. The trees were selected based on their characters at the time of<br />
transpiration measurement. For example, a sapling of Swietenia was selected to<br />
represent the bottom canopy layer because of its lower height. However, this does<br />
not imply that Swietenia always would occupy the lower canopy layer of MHGs.<br />
Among the several tree species occupying the top layer, Artocarpus heterophyllus,<br />
which is the most abundant species in Kandyan MHGs 4 , was selected.<br />
Table 1. List of tree species in a multilayered homegarden in central Sri Lanka and their<br />
structural attributes.<br />
Sl.<br />
No<br />
Tree species Height<br />
(m)<br />
W.A.J.M. DE COSTA ET AL .<br />
Canopy<br />
depth<br />
(m)<br />
Canopy<br />
spread<br />
(m)<br />
DBH<br />
(cm)<br />
1 Persea americana 7.5 6 3.5 16 10<br />
2 Artocarpus heterophyllus 15.5 14 14 55 40<br />
3 Gliricidia sepium 13 11 3 14 27<br />
4 Persea americana 11 6 5 18.5 10<br />
5 Mangifera indica 12.5 8.5 5.5 17 10<br />
6 Cedrela toona 8 4 1.5 8.5 8<br />
7 Persea americana 13 10 4.5 25.5 11<br />
8 Swietenia macrophylla 15 6 5 37 15<br />
9 Swietenia macrophylla 15.5 9.5 6.5 46 15<br />
10 Cocos nucifera 15 6 9.5 30 10<br />
11 Swietenia macrophylla 1.9 1.3 0.9 1 3<br />
12 Cocos nucifera 15 6 9 24.5 10<br />
13 Cocos nucifera 14 5.75 9 28.5 10<br />
14 Cocos nucifera 16 7 9 32 10<br />
15 Swietenia macrophylla 4 1.5 0.6 3.5 3<br />
16 Alstonia macrophylla 4 1.5 2 3 5<br />
17 Alstonia macrophylla 9.5 7.5 4 9.5 2<br />
18 Persea americana 9 7 4 15.5 5<br />
19 Artocarpus heterophyllus 15 13 10 40.5 25<br />
20 Persea americana 8.5 4 2 17 8<br />
21 Cedrela toona 8 1.9 2.8 9 8<br />
22 Cedrela toona 8.75 4.5 2.3 9.5 8<br />
23 Alstonia macrophylla 8 3 4.2 7.5 5<br />
24 Michelia champaca 13 8.5 5 18 10<br />
25 Swietenia macrop o hylla 6.5 2.5 2 8 8<br />
26 Swietenia macrophylla 6.5 2.5 3.5 10.5 8<br />
27 Swietenia macrophylla 7 2.5 2.5 10 8<br />
28 Cedrela toona 4.3 0.25 1.5 3 8<br />
Probable<br />
age (yr)
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
29 Cedrela toona 4.3 0.25 1 3.2 8<br />
30 Cedrela toona 4 0.25 0.5 3 8<br />
31 Myristica fragrans 4.3 1.7 1.4 3 8<br />
32 Michelia champaca 13.5 7.5 6 30 8<br />
33 Alstonia macrophylla 8 4.5 5 11 15<br />
34 Swietenia macrophylla 7 4.3 2 7 8<br />
35 Swietenia macrophylla 7 4 1.8 6.5 8<br />
36 Cedrela toona 5 0.75 2 4 8<br />
37 Swietenia macrophylla 7.5 5 3 11 8<br />
38 Swietenia macrophylla 7.5 5 1.5 7.5 8<br />
39 Litsea iteadaphna 7.5 6 5.5 22.2 15<br />
40 Michelia champaca 14 6 4.7 16.5 10<br />
41 Cedrela toona 10 * * 7 8<br />
42 Caryota urens 13 6.5 6 33.5 8<br />
43 Cocos nucifera 14.5 5 9 30 10<br />
44 Cedrela toona 13.5 0.5 2.8 3 8<br />
45 Syzygium aromaticum 13 9.5 6 21.5 25<br />
46 Coffea arabica 7.5 6.5 3.5 5.25 5<br />
47 Cocos nucifera 14 6.5 9.5 27.5 10<br />
48 Persea americana 8 5 5.5 15 6<br />
49 Gliricidia sepium 7 6.5 3 6.5 15<br />
50 Duria zibethinus 22 7 10.2 40 28<br />
51 Gliricidia sepium 8 5 2 12 14<br />
52 Cocos nucifera 13.5 5 4.5 22 28<br />
53 Alstonia macrophylla 17 7 1.5 15 8<br />
54 Psidium guajava 1.9 1.5 1.5 2 10<br />
55 Swietenia macrophylla 1.9 1 1.2 1 3<br />
56 Cassia roxburghii 23 1.2 7 45 40<br />
Note: The trees selected for transpiration measurement are indicated in bold; DBH = diameter<br />
at breast height.<br />
2.3. Measurement of transpiration rates of different trees<br />
255<br />
Among a variety of techniques available for measuring transpiration rates in trees,<br />
thermal methods (Swanson, 1994) involve measurement of the rate of heat flow in<br />
the xylem transpiration stream. Under steady-state conditions, the rate of xylem sap<br />
flow would be equal to the rate of transpiration of a tree (Van den Honert, 1948;<br />
Cowan, 1965). An added advantage of thermal methods is their ability to measure<br />
transpiration rates of individual trees, which is essential in a species mixture such as<br />
the MHG. In the present study, we used two thermal methods, thermal dissipation<br />
probes and stem sap flow gauges (Dynamax Inc, USA/Delta-T, UK), the former for<br />
Artocarpus (DBH = 40.5 cm) and Cedrela (DBH = 9 cm), and the latter for
256<br />
W.A.J.M. DE COSTA ET AL .<br />
Swietenia. Both instruments measure the rate of sap flow in the xylem, but the<br />
thermal dissipation probe (TDP) relates the rate of heat dissipation to sap flow rate<br />
through an empirical relationship, while the sap flow gauge directly calculates the<br />
rate of sap flow as the residual of an energy balance equation.<br />
The TDP consisted of two ‘needles’, each 30 mm long and 1.2 mm in outer<br />
diameter, spaced 40 mm apart; these were inserted horizontally to the trunk at a<br />
height of 1.2 m above the ground. Both needles contained thermocouples to measure<br />
the average sap temperature. In addition, the upper needle contained a heating<br />
element, which was supplied with a constant input of energy (0.15 Js –1 ) by a 12 V<br />
car battery through a voltage regulator (AVRD Dual Regulator, Delta-T Devices).<br />
As the lower un-heated needle measured the reference sap temperature, temperature<br />
difference (dT) T between the two needles was determined by rate of heat dissipation<br />
due to sap flow. As such, dT T was inversely related to sap velocity. It was measured<br />
and recorded as a voltage signal every 30 seconds, averaged every 5 minutes and<br />
stored in a DL2e data logger (Delta-T Devices). Equation 1 (Granier, 1985; 1987)<br />
was used to compute a dimensionless ‘flow index’ K as follows,<br />
K = (dTm – dT)/dT (1)<br />
Where dTm is the maximum recorded value of dT T at times of zero sap flow. In the<br />
present experiment, the maximum dTm values were recorded between midnight and<br />
0400 h. Mean dTm during this four-hour period each day was taken as dTm for the<br />
–1<br />
respective day. K was related to the average sap velocity, V, V in cm s , by an<br />
empirical relationship of Granier (1985; 1987) as,<br />
V = 0.0119 K 1.231<br />
K (2)<br />
This relationship, which did not differ significantly among tree species, was used in<br />
the present study. V was converted to sap flow rate, F , as,<br />
V Fs<br />
Fs F = AsV (3)<br />
Where As is sap wood cross-sectional area, which was measured by taking several<br />
core samples using an increment borer on Artocarpus and Cedrela trees that were<br />
not being used for sap flow measurements. As recommended by the manufacturer<br />
(Dynamax), three thermal dissipation probes were used at different locations around<br />
the trunk of Artocarpus while two probes were used on Cedrela. Signals from<br />
different probes were averaged before sap flow calculations.<br />
The Dynagage (SGB25-ws) consisted of a heating plate, 110 mm high and 28<br />
mm in diameter, which was wrapped around the stem of sapling Swietenia at a<br />
height of 1.2 m above the ground. A constant, regulated power input (Pin) of 0.5 Js –1<br />
was supplied to the heating plate. A set of precise electronic sensors attached to the<br />
gauge measured the radial heat transfer from the gauge to the ambient air (Qr) and
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
257<br />
axial heat fluxes through the trunk (Qv). The variable amount of heat carried by the<br />
sap flow (Qf ) was calculated from an energy balance equation (Sakuratani, 1981;<br />
Baker and Van Bavel, 1987) as,<br />
Qf = Pin – Qr – Qv (4)<br />
Qf f can be converted to the sap flow rate ( F) by dividing it with specific heat capacity<br />
of water (Cp) and sap temperature increase (dT ) as,<br />
F = (Pin – Qr – Qu – Qd)/(Cp dT) (5)<br />
Where Qu and Qd d are the upward and downward components of the axial heat<br />
transfer (Qv). Voltage signals from sensors were recorded every 30 seconds,<br />
averaged every 5 minutes and stored in the data logger.<br />
2.4. Measurement of environmental variables<br />
Solar radiation incident on the canopies was measured by tube solarimeters installed<br />
at appropriate heights above and within the MHG. Relative humidity and air<br />
temperature at the top of the three selected trees were measured by solid-state<br />
sensors (TDK Inc., Hiroshima, Japan) installed at the respective heights. Output<br />
signals from all these sensors were recorded every 30 seconds, averaged every 5<br />
minutes and stored in the data logger. Relative humidity (h) and air temperature (Ta T )<br />
data were used to compute the saturation vapor pressure deficit (VPD) of air as,<br />
VPD = es(Ta) – e (6)<br />
Where es(Ta) is the saturation vapor pressure at air temperature Ta T and e the actual<br />
vapor pressure. es(Ta) was computed by an empirical equation developed by Tetens<br />
(1930) and adopted by Murray (1967) as,<br />
[17.27 [(T – 273)/(T – 36)]<br />
es(Ta) = 0.611 e<br />
The actual vapor pressure (e) was calculated from measured relative humidity and<br />
es(Ta) as,<br />
(7)<br />
e = (h es(Ta))/100 (8)<br />
Soil moisture content was measured gravimetrically at weekly intervals by taking<br />
samples at 20 cm depth intervals down to a maximum depth of 1 m. Soil moisture<br />
was measured at a point approximately equidistant from the three trees on which<br />
transpiration was being measured.
258<br />
2.5. Computation of canopy conductance<br />
Canopy conductance (gc) is the overall stomatal conductance of the entire foliage<br />
canopy. It was computed by inverting a simplified version of the Penman-Monteith<br />
equation adopted for tall vegetation by Granier et al. (1996) as,<br />
gc = (F )/( ( Cp D) (9)<br />
–2 –1<br />
Where F is mean sap flow rate (kg m s ), the latent heat of evaporation of water<br />
(J kg –1 ), the psychrometer constant (kPa K –1 ), the density of air (kg m –3<br />
m ), Cp the<br />
specific heat capacity of air (J kg –1 K –1 ) and D the vapor pressure deficit of air (kPa).<br />
The measured daily total sap flow values obtained in kg tree –1 d –1 were divided by<br />
–2<br />
tree leaf area and day length to convert them to F in kg m s –1 . Therefore, gc<br />
obtained from equation 9 were daily mean canopy conductance values.<br />
2.6. Measurement of canopy area<br />
Approximate leaf area of all three trees selected for sap flow measurements was<br />
computed by measuring area of a sample of 50 leaves representing a range of sizes<br />
and counting the number of branches, leaf cohorts, and leaf number per cohort as<br />
follows:<br />
Approximate canopy area per tree = No. of branches per tree x mean no. of leaf<br />
cohorts per branch x mean no. of leaves per cohort x mean area per leaf (10)<br />
Projected leaf area index (LAI) I was computed as the ratio between approximate<br />
canopy area per tree and projected horizontal ground area covered by the tree<br />
canopy, assuming a circular canopy having a diameter equal to the measured canopy<br />
spread (Table 1).<br />
2.7. Data analysis<br />
W.A.J.M. DE COSTA ET AL .<br />
Data on transpiration and environmental variables recorded at 5-minute intervals<br />
were plotted against time of the day to obtain their diurnal variation patterns.<br />
Regression analysis was used to obtain relationships between transpiration and<br />
environmental variables.<br />
3. RESULTS<br />
3.1. Short-term variations in tree transpiration rates and its determinants in the<br />
absence of soil water deficits<br />
The data presented in Fig. 1a show the diurnal variation of sap flow rate over a 72-h<br />
period (from 23 to 25 June 2001) when the soil was fully saturated. All three species<br />
showed a similar pattern of sap flow, with the maximum rate being achieved from<br />
1300 to 1500 h. Minimum sap flow rates were observed from midnight to 0400 h.
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
259<br />
The very small rates of sap flow that were observed during the night were probably<br />
to replace the water that was lost during the daytime from water storage tissues (i.e.,<br />
capacitors) surrounding the xylem vessels. Artocarpus, which occupied the upper<br />
canopy layer of the MHG, showed the highest sap flow rate. There was, however, no<br />
significant difference in sap flow rate between Cedrela (middle canopy) and<br />
Swietenia (sapling in the lower canopy). Furthermore, the highest sap velocity<br />
Sap Flow (g/300 s)<br />
Sap Velocity (mm/min)<br />
140<br />
120<br />
100<br />
8<br />
7<br />
6<br />
5<br />
4<br />
3<br />
2<br />
1<br />
80<br />
60<br />
40<br />
20<br />
(a)<br />
0<br />
0 4 8 12 16 20 24 28 32 36 40 44 48 52 56 60 64 68 72<br />
(b)<br />
Time (hours)<br />
0<br />
0 4 8 12 16 20 24 28 32 36 40 44 48 52 56 60 64 68 72<br />
Time (hours)<br />
Figure 1. Diurnal variation of sap flow (a) and sap velocity (b) of three tree species<br />
representing different vertical layers in a multi-layered homegarden in central Sri Lanka<br />
during a 72 h measurement period of adequate soil water availability. Art. – Artocarpus<br />
heterophyllus; Ced. – Cedrela toona; Mah. – Swietenia macrophylla.<br />
Art.<br />
Ced.<br />
Mah.<br />
Art.<br />
Ced.<br />
Mah.
260<br />
was observed in Swietenia (Fig. 1b) while the lowest was noted in Artocarpus, with<br />
Cedrela having intermediate values. The respective sapwood cross-sectional areas<br />
for Artocarpus, Cedrela and Swietenia were 605, 51 and 9.6 cm 2 . Hence, an inverse<br />
relationship between sap velocity and sapwood cross-sectional area can be deduced.<br />
This is because sap has to be translocated at a higher velocity to provide enough<br />
water to compensate for the transpirational losses in individuals having a lower<br />
sapwood cross-sectional area.<br />
The upper canopy Artocarpus trees also received the highest irradiance (Fig. 2)<br />
during most of the day. However, irradiance on the upper canopy showed substantial<br />
short-term fluctuations because of partially cloudy weather. Middle canopy Cedrela<br />
Irradiance (W m -2 )<br />
800<br />
700<br />
600<br />
500<br />
400<br />
300<br />
200<br />
100<br />
0<br />
W.A.J.M. DE COSTA ET AL .<br />
0 4 8 12 16 20 24 28 32 36 40 44 48 52 56 60 64 68 72<br />
Time (hours)<br />
Art.<br />
Ced.<br />
Mah.<br />
Figure 2. Diurnal variation of incident radiation on canopies of three selected tree species in<br />
a multi-layered homegarden in central Sri Lanka during a selected 72 h measurement period.<br />
Artocarpus heterophyllus (Art.), Cedrela toona a (Ced.), and Swietenia macrophylla (Mah.)<br />
represented the upper, middle, and lower layers of the homegarden.<br />
and lower canopy Swietenia received radiation, which was approximately similar to<br />
each other and less fluctuating compared to the upper canopy. Even the lower<br />
canopy Swietenia was exposed to direct sunlight during part of the day (from the<br />
direction of the house), which explains the similarity in irradiance level of the<br />
middle and lower canopies. When the data for the different canopy layers were<br />
pooled, there was, however, a clear linear relationship between transpiration and<br />
incident radiation on a daily basis (Fig. 3). Daily total transpiration of middle and<br />
lower canopy trees were 15 to 27% of that of upper canopy trees (Table 2).<br />
Vapor pressure deficit above the MHG (i.e., near the upper canopy) was greater<br />
than that within it (data not shown), especially during daytime. However, the diurnal
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
261<br />
patterns of both were similar with maximum values around 1200 to 1400 h.<br />
Transpiration rates of trees representing all canopy layers increased with increasing<br />
VPD (Fig. 4). However, above a threshold VPD of about 0.8 kPa the increase of<br />
transpiration slowed down, because of decreasing stomatal conductance at higher<br />
VPD (data shown elsewhere).<br />
Sap Flow (kg d −1 tree −1 )<br />
14<br />
12<br />
10<br />
8<br />
6<br />
4<br />
2<br />
0<br />
y = 1.2105 x + 0.8196<br />
R 2 = 0.9941<br />
0 2 4 6 8 10<br />
Irradiance (MJ m −2 d −1 )<br />
Figure 3. Relationship between daily total sap flow of the three selected tree species and the<br />
daily total solar radiation incident on their respective foliage canopies during a 72 h<br />
measurement period.<br />
Table 2. Daily totals of transpiration and incident radiation during a three-day period of<br />
adequate soil water availability (june 2001) in a multi-layered homegarden in central Sri<br />
Lanka.<br />
Transpiration (kg d –1 tree –1 ) Incident radiation (MJ m –2 d<br />
2 –1 Species<br />
)<br />
23 Jun ‘01 24 Jun ‘01 25 Jun ‘01 23 Jun ‘01 24 Jun ‘01 25 Jun ‘01<br />
Artocarpus<br />
heterophyllus<br />
9.46 11.80 10.34 6.73 9.44 7.77<br />
Cedrela toona 2.06 2.64 2.00 1.04 1.37 1.16<br />
(0.22) (0.22) (0.19) (0.16) (0.15) (0.15)<br />
Swietenia<br />
2.04 3.10 2.77 1.38 1.71 1.49<br />
macrophylla (0.22) (0.26) (0.27) (0.20) (0.18) (0.19)<br />
Note: Fraction of the respective upper canopy value is given in parenthesis.<br />
3.2. Medium-term variation of transpiration during a period of increasing soil water<br />
deficits<br />
Daily transpiration rates of the upper canopy Artocarpus and middle canopy Cedrela<br />
were monitored during a two-month period of very little rainfall (25 Dec. 2001 to 20
262<br />
Feb. 2002). Despite the gradual decrease of soil water content in the top 1 m of the<br />
soil, both trees continued to have high rates of transpiration (Fig. 5). This probably<br />
indicated that root systems of both tree species absorbed water from deeper layers of<br />
the soil profile during periods when the top soil was dry. Total transpiration of<br />
Artocarpus during the 54-day experimental period was 3881 kg tree –1 , with the daily<br />
transpiration ranging from 17.84 to 95.87 kg day –1 tree –1 . The corresponding values<br />
for Cedrela were 463 kg tree –1 and 0.64 to 21.60 kg day –1 tree –1 .<br />
Sap Flow Rate (g / 300 s)<br />
140<br />
120<br />
100<br />
80<br />
60<br />
40<br />
20<br />
0<br />
(a)<br />
0 0.3 0.6 0.9 1.2 1.5<br />
Vapour Pressure Deficit above MHG (kPa)<br />
Sap Flow Rate (g / 300 s)<br />
30<br />
25<br />
20<br />
15<br />
10<br />
5<br />
0<br />
(b)<br />
0 0.2 0.4 0.6 0.8 1<br />
Vapour Pressure Deficit within MHG (kPa)<br />
Sap Flow Rate (g / 300s)<br />
35<br />
30<br />
25<br />
20<br />
15<br />
10<br />
5<br />
0<br />
(c)<br />
0 0.3 0.6 0.9 1.2<br />
Vapour Pressure Deficit within MHG<br />
Figure 4. Variation of sap flow rate with vapour pressure deficit in Artocarpous<br />
heterophyllus (a), Cedrela toona a (b) and Swietenia macrophylla (c) in a central Sri Lankan<br />
homegarden during a selected 72 h measurement period.<br />
Daily Sap Flow (kg/tree/d)<br />
120<br />
100<br />
80<br />
60<br />
40<br />
20<br />
0<br />
25-Dec-01<br />
01-Jan-02<br />
W.A.J.M. DE COSTA ET AL .<br />
08-Jan-02<br />
15-Jan-02<br />
22-Jan-02<br />
29-Jan-02<br />
Calendar Date<br />
05-Feb-02<br />
y = -0.5641x + 21071<br />
R 2 = 0.9701<br />
12-Feb-02<br />
19-Feb-02<br />
Artocarpus Cedrella Soil Water Linear (Soil Water)<br />
Figure 5. Medium-term variation of daily sap flow of two selected upper- (Artocarpus<br />
heterophyllus) and middle canopy (Cedrela toona) trees in multi-layered homegarden in<br />
central Sri Lanka during a selected 54-day experimental period. Variation of soil water<br />
content in the top 1 m of the soil profile is also shown along with a fitted linear regression.<br />
70<br />
60<br />
50<br />
40<br />
30<br />
20<br />
10<br />
0<br />
Soil Water Content (cm/m)
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
263<br />
The clear increase of daily transpiration in Cedrela was due to the significant<br />
increase of its projected LAI from 1.4 to 3.0 during the experimental period. In<br />
contrast, the projected LAI of Artocarpus was around 5.7 throughout. Medium-term<br />
fluctuations in sap flow of Artocarpus were closely related to fluctuations of daily<br />
total irradiance (Fig. 6a) and daily mean VPD (Fig. 6b). Transpiration of Artocarpus<br />
increased with increasing irradiance up to about 13 MJ m -2 d -1 and with increasing<br />
VPD up to about 0.8 kPa. The subsequent leveling-off and reduction was probably<br />
due to partial stomatal closure induced by higher VPD. This was confirmed by<br />
observed reduction of canopy conductance (gc) with increasing vapor pressure<br />
deficits both on a daily basis (Fig. 7) and on a diurnal basis (data not shown).<br />
Sap Flow (kg tree −1 d −1 )<br />
100<br />
80<br />
60<br />
40<br />
20<br />
(a)<br />
y = -0.513x 2 + 17.433x - 67.407<br />
R 2 = 0.6746<br />
0<br />
0 5 10 15 20<br />
Incident Radiation (MJ m −2 d −1 )<br />
Figure 6. Relationships between daily sap flow of upper canopy Artocarpus heterophyllus<br />
with daily total irradiance (a) and daily mean vapour pressure deficit (b) in a multi-layered<br />
homegarden in central Sri Lanka during a selected 54-day experimental period.<br />
Canopy Conductance (cm s −1 )<br />
5<br />
4<br />
3<br />
2<br />
1<br />
0<br />
Figure 7. Relationship between canopy conductance during the day time in upper canopy<br />
Artocarpus heterophyllus and vapour pressure deficit in a multi-layered homegarden in<br />
central Sri Lanka during a selected 54-day experimental period. The canopy conductance was<br />
estimated by inverting a simplified version of the Penman-Monteith equation.<br />
Sap Flow (kg/tree/d)<br />
120<br />
100<br />
80<br />
60<br />
40<br />
20<br />
(b)<br />
y = -47.386x 2 + 124.71x + 2.063<br />
R 2 = 0.511<br />
0<br />
0 0.5 1 1.52 2.5<br />
y = -1.0738x + 2.9371<br />
R 2 = 0.438<br />
Daily Mean VPD (kPa)<br />
0 0.5 1 1.5 2 2.5<br />
Vapour Pressure Deficit (kPa)
264<br />
W.A.J.M. DE COSTA ET AL .<br />
4. DISCUSSION<br />
4.1. Water use of multi-layered homegardens and its determinants<br />
Results of the present study clearly show that the taller trees occupying the upper<br />
canopy, which experience higher levels of incident radiation and vapour pressure<br />
deficits, dominate the total water use of MHGs. However, the contribution of trees<br />
occupying the middle and lower strata cannot be ignored. For instance, at the<br />
individual tree level, the combined transpiration of these two strata accounted for 30<br />
to 33% of the total water use of the three strata (based on data in Table 2).<br />
A linear increase in sap flow with incident solar radiation (Fig. 3) and vapour<br />
pressure deficit (Fig. 4) implies that these are the main drivers of transpiration in the<br />
MHG trees, irrespective of the level of water availability in the top 1 m of the soil.<br />
Furthermore, the linear relationship between transpiration and irradiance on a daily<br />
basis can be used to predict the daily transpiration rates of trees during periods of<br />
adequate soil water availability. The dependence of tree transpiration on VPD and<br />
irradiance is consistent with several other studies on a range of tree species (Granier<br />
and Loustau, 1994; Granier et al., 1996).<br />
Decreasing sensitivity of transpiration rate to increasing VPD and irradiance, as<br />
shown by decreasing slopes of the relevant relationships above 0.8 kPa (Fig. 4a and<br />
Fig. 6b) and above 13 MJ m –2 d –1 (Fig. 6a), indicates some degree of stomatal<br />
control of transpiration. These observations suggest that canopy conductance<br />
decreases with increased VPD above 0.8 kPa, which was confirmed by the observed<br />
reduction in canopy conductance with increasing VPD (Fig. 7). This is in agreement<br />
with the findings of several other studies (Roberts et al., 1990; Granier et al., 1996;<br />
Hogg and Hurdle, 1997; Meinzer et al., 1997) on several tree species in different<br />
forest types. However, the high levels of daily transpiration observed during the<br />
prolonged rainless period (Fig. 5) show that the level of stomatal control observed<br />
was not strong enough to reduce transpiration substantially. It is probable that both<br />
Artocarpus and Cedrela had root systems that were deep enough to extract water<br />
from soil depths below 1 m.<br />
4.2. Implications on sustainability of multi-layered homegardens in central<br />
Sri Lanka<br />
The MHGs in the Central Province of Sri Lanka are generally found on deep soils<br />
with high potential for water storage. Presence of deep-rooted trees capable of<br />
absorbing water from the lower soil layers is, however, a matter of concern under<br />
certain circumstances—especially during the rainless periods. Although the Sri<br />
Lankan MHGs generally predominate the humid tropical climatic zone having welldistributed<br />
rainfall (~2000 mm yr –1 ), the predicted drop in total rainfall and its<br />
increasingly non-uniform distribution in a future climate change scenario (McCarthy<br />
et al., 2001), is becoming a matter of concern. Perhaps this may be an overcautious<br />
scenario considering that the MHGs have sustained themselves for several centuries<br />
in areas with shallow soils and limited ground water resources. For example, the
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
265<br />
Maya homegardens of the Yucatan Peninsula of Mexico (Benjamin et al., 2001)<br />
have provided sustainable livelihoods under rather harsh environmental conditions<br />
with limited water resources and soil nutrients. This could be yet another aspect of<br />
the ‘mysteries’ or the ‘enigma’ of tropical homegardens that defy the conventional<br />
scientific wisdom developed based on single-species systems (Nair, 2001; Kumar<br />
and Nair, 2004).<br />
5. CONCLUSIONS AND DIRECTIONS FOR FURTHER RESEARCH<br />
It is acknowledged that this study is based on a set of data, which is limited in<br />
several aspects. First, the transpiration measurements are not replicated and are<br />
based on only three trees out of 56 present in the 0.15 ha extent of the MHG. This<br />
was because of the practical difficulties involved in installing TDPs or sap flow<br />
gauges on an adequate number of trees and saplings and recording their output<br />
signals. Moreover, the highly uneven nature of tree distribution made replications<br />
difficult. For example, the two trees of Artocarpus heterophyllus were situated in the<br />
opposite parts of the MHG. We acknowledge that adequately replicated measurements<br />
of several tree species have to be done before making firm conclusions on the<br />
dynamics of transpiration in a highly complex vegetation system such as the MHGs.<br />
Subject to the above limitations, the study suggests that water use of multilayered<br />
homegardens of Central Sri Lanka is dominated by the upper canopy trees,<br />
with appreciable contributions from middle and lower canopy trees. Transpiration<br />
rates of MHGs are driven by incident radiation and vapour pressure deficit during<br />
periods of both adequate soil water availability and significant soil water deficits.<br />
Upper and middle canopy trees of MHGs maintain high rates of transpiration even<br />
during prolonged rainless periods by absorbing water from deeper soil layers. These<br />
findings should prompt concern on the impacts of high transpiration rates of MHGs<br />
on catchment water yield in a predicted future climate of reduced rainfall. We have<br />
not investigated the extent and depth of ground water availability. Despite the<br />
enormous practical difficulties involved, further in-depth studies are needed to<br />
quantify this impact at the catchment scale and to understand the ability of MHGs to<br />
sustain the livelihoods and ecosystem stability.<br />
ACKNOWLEDGEMENTS<br />
We thank the National Research Council of Sri Lanka for financial support<br />
(Research grant no. NRC-99-23) and Ms. M.R.H.L. Karunasinghe and Mr. P.<br />
Surenthran for help in the fieldwork. A special word of thanks should go to Mr.<br />
Tennakoon, the land owner of the homegarden in which this research was done for<br />
all support and <strong>coop</strong>eration.
266<br />
W.A.J.M. DE COSTA ET AL .<br />
ENDNOTES<br />
1. McConnell D.G. and Dharmapala K.A.B. 1973. The economic structure of<br />
Kandyan Forest Farms. The Management Report No. 7, UNDP/FAO,<br />
Agricultural Diversification Project, Peradeniya, Sri Lanka.<br />
2. Finlayson W. 1998. Effects of deforestation and of tree planting on the<br />
hydrology of the Upper Mahaweli catchment: A review of the published<br />
evidence. Environment and Forest Conservation Division, Mahaweli Authority<br />
of Sri Lanka, Polgolla.<br />
3. Gunawardena E.R.N. 1998. Overview of the hydrology project. In: Gunasena<br />
H.P.M. (ed.), Proceedings of the final workshop: University of Peradeniya -<br />
Oxford Forestry Institute Link Project, July 1998, pp. 20 – 29. UP-OFI Link<br />
Project, Peradeniya.<br />
4. Hitinayake H.M.G.S.B., De Costa W.A.J.M. and Jayaweera K.G.D. 1996. Food<br />
trees in multi-layered homegardens in different agro-ecological regions of<br />
Kandy district. In: Gunasena H.P.M. (ed.), Multipurpose trees for food security.<br />
Proceedings of the seventh regional workshop on multipurpose tree species,<br />
Kandy, Sri Lanka (24 – 26 October 1996), pp. 252 – 264. UP-OFI Link Project,<br />
Peradeniya.<br />
REFERENCES<br />
Baker J.M. and Van Bavel C.H.M. 1987. Measurement of mass flow of water in stems of<br />
herbaceous plants. Plant Cell Environ 10: 777 – 782.<br />
Benjamin T.J., Montanez P.I., Jimenez J.J.M. and Gillespie A.R. 2001. Carbon, water and<br />
nutrient flux in Maya homegardens in the Yucatan peninsula of Mexico. Agroforest Syst<br />
53: 103 – 111.<br />
Bosch J.M. and Hewlett J.D. 1982. A review of catchment experiments to determine the<br />
effects of vegetation changes on water yield and evapotranspiration. J Hydrol 55: 3 – 23.<br />
Calder I.R .1996. Water use by forests at the plot and catchment scale. Commonwealth For<br />
Rev 75: 19 – 30.<br />
Cowan I.R. 1965. Transport of water in the soil-plant-atmosphere system. J Appl Ecol 2:<br />
221 – 239.<br />
Granier A. 1985. Une nouvelle methode pour la mesure du flux de seve brute dans le tronc<br />
des arbres. Ann For Sci 42: 81 – 88.<br />
Granier A. 1987. Evaluation of transpiration in a Douglas-fir stand by means of sap flow<br />
measurements. Tree Physiol 3: 309 – 320.<br />
Granier A. and Loustau D. 1994. Measuring and modeling the transpiration of a maritime<br />
pine canopy from sap-flow data. Agric For Meteorol 71: 61 – 81.<br />
Granier A., Huc R. and Barigah S.T. 1996. Transpiration of natural rain forest and its<br />
dependence on climatic factors. Agric For Meteorol 78: 19 – 29.<br />
Hogg E.H. and Hurdle P.A. 1997. Sap flow in trembling aspen: implications for stomatal<br />
responses to vapour pressure deficit. Tree Physiol 17: 501 – 509.<br />
Jacob V.J. and Alles W.S. 1987. Kandyan gardens of Sri Lanka. Agroforest Syst 5: 123 – 137.<br />
Jarvis P.G. and McNaughton K.G. 1986. Stomatal control of transpiration: scaling up from<br />
leaf to region. Adv Ecol Res 15: 1 – 49.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst<br />
61/62: 135 – 152.
TRANSPIRATION<br />
T CHARACTERISTICS C OF HOMEGARDEN TREES T<br />
267<br />
McCarthy J.J., Canziani O.F., Leary N.A., Dokken D.J. and White K.S. 2001. Climate change<br />
2001: Impacts, adaptation and vulnerability. Contribution of Working Group II to the<br />
Third Assessment Report of the Intergovernmental Panel on Climate Change (IPCC).<br />
Cambridge University Press, Cambridge, 1032p.<br />
Meinzer F.C., Andrade J.L., Goldstein G., Holbrook N.M., Cavelier J. and Jackson P. 1997.<br />
Control of transpiration from the upper canopy of a tropical forest: the role of stomatal,<br />
boundary layer and hydraulic architecture components. Plant Cell Environ 20:<br />
1242 – 1252.<br />
Monteith J.L. 1986. How do crops manipulate water supply and demand? Phil Trans Royal<br />
Soc London, Ser A 316: 245 – 259.<br />
Murray F.W. 1967. On the computation of saturation vapour pressure. J Appl Meteorol 6: 203<br />
– 204.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Panabokke C.R. 1996. Soils and Agro-ecological Environments of Sri Lanka. Natural<br />
Resources, Energy and Science Authority of Sri Lanka, Colombo, 220p.<br />
Roberts J., Cabral O.M.R. and Aguiar L. de F. 1990. Stomatal and boundary layer<br />
conductances in Amazonian Terra firme rain forest. J Appl Ecol 27: 336 – 353.<br />
Sakuratani T. 1981. A heat balance method for measuring water flux in the stem of intact<br />
plants. J Agric Meteorol 37: 9 – 17.<br />
Senarath A. and Dassanayake A.R. 1999. Soils of the mid-country wet zone. In: Mapa R.B.,<br />
Somasiri S., and Nagarajah S. (eds), Soils of the wet zone of Sri Lanka: Morphology,<br />
characterization and classification, pp. 92 – 121. Soil Science Society of Sri Lanka,<br />
Peradeniya.<br />
Swanson R.H. 1994. Significant historical developments in thermal methods for measuring<br />
sap flow in trees. Agric For Meteorol 72: 113 – 132.<br />
Tetens O. 1930. Uber einige meteorologische Begriffe. Zeitschrift Geophysic 6: 297 – 309.<br />
Van den Honert T.H. 1948. Water transport in plants as a catenary process. Discuss Faraday<br />
Soc 3: 146 – 153.<br />
Wallace J.S. 1996. The water balance of mixed tree-crop systems. In: Ong C.K. and Huxley<br />
P.A. (eds), Tree-crop interactions: A physiological approach, pp. 189 – 233. CAB<br />
International, Wallingford.
CHAPTER 15<br />
ECOLOGY VERSUS ECONOMICS<br />
IN TROPICAL MULTISTRATA<br />
AGROFORESTS<br />
E. TORQUEBIAU* AND E. PENOT<br />
CIRAD TERA, TA 60/15 – 34398, Montpellier CX5, France; *E-mail:<br />
<br />
Keywords: Environmental services, Externalities, Modeling, Risk buffering, Rubber.<br />
Abstract. <strong>Homegardens</strong> and other multistrata agroforests are often described as ecologically<br />
sound, economically viable, and socially equitable land use activities. As in a majority of<br />
sustainable management situations, there are no widely accepted norms for a “perfect”<br />
combination of these attributes; what is often envisaged is a compromise among them. We<br />
argue that the development of ecological features of homegardens can be fostered by an<br />
“innovative” economic analysis. Performance of homegardens cannot be fully assessed by<br />
using conventional economic criteria and approaches such as yield, cost-benefit analysis, and<br />
net present value. Alternatively, if micro- and meso-level economic analyses (farming<br />
systems and upper level systems) are applied, the internalization of externalities such as<br />
agrobiodiversity management, carbon sink value, improved nutrient cycling or integrated pest<br />
management may turn homegardens into highly profitable ventures. Economic analysis<br />
methods should integrate risk buffering, outputs of mixtures of plants with different cycles,<br />
and allow to take a into account farming strategies with long-term objectives as well as the<br />
patrimonial (asset inheritance) components. Additionally, the merits of homegardens in terms<br />
of subsistence food for families, flexibility in production, reduced external-input<br />
requirements, enhanced aesthetic-, landscape-, and societal values, should also be<br />
incorporated into such an analysis.<br />
1. INTRODUCTION<br />
In the realm of agroforestry, homegardens and other multistrata, multispecies<br />
associations occupy an odd place. They are the most elaborate manmade, tree-cropanimal<br />
associations, and as such the only agroforestry system which can claim a<br />
resemblance to natural forests; hence their alternative name “agroforests.” Although<br />
269<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 269–282.<br />
© 2006 Springer. Printed in the Netherlands.
270 E. TTORQUEBIAU AND E. PENOT<br />
these systems have been studied in several countries (Indonesia, Brazil, India, and<br />
Sri Lanka: see chapters in this volume), the fact remains that they are seldom<br />
advocated as a land use option in agricultural or forestry development paradigms.<br />
Before pursuing further, a clarification on the use of the term homegardens<br />
versus other multistrata agroforestry systems is relevant. Both are multistrata<br />
combinations of trees and crops (sometimes with a livestock component).<br />
<strong>Homegardens</strong> are located next to human dwellings, managed for the production of<br />
subsistence items, and sometimes includes a cash objective. They are practiced on<br />
small parcels of land and are usually intensively managed. Not all multistrata<br />
agroforestry systems may, however, qualify as homegardens. Examples are the<br />
village-forests (village-forest-gardens), which are multistrata agroforestry systems<br />
developed on larger areas (at least a few ha per family) and managed mainly for<br />
cash income generation through the production of resins, jungle rubber, wood, fruits,<br />
etc. These are often considered as ‘intermediates’ between natural forests and treecrop<br />
plantations (Wiersum, 2004). Neither can all agroforestry systems be called<br />
“agroforests.” For example, the term does not cover agroforestry systems such as<br />
scattered trees on croplands, windbreaks, or woody hedgerows. As the term<br />
suggests, agroforests resemble forests and mimic their ecology (Michon and de<br />
Foresta, 1999; Wiersum, 2004). This resemblance is important in the context of the<br />
present chapter, as ecological and economic analyses applied to multistrata systems<br />
partly draw on their forest equivalents. Agroforest is thus the term used to represent<br />
both homegardens and other multistrata agroforestry systems and will hereafter be<br />
used in this chapter (instead of homegardens).<br />
1.1. Attributes and spread of agroforests<br />
Because of their resemblance to forests both physiognomically and ecologically,<br />
agroforests have a “good reputation.” Most statements recognizing the quality of<br />
agroforests, including in recent papers, refer to their ecological attributes, in<br />
particular biodiversity conservation and the long-term benefits of soil fertility<br />
maintenance and water conservation (Gajaseni and Gajaseni, 1999; Kaya et al.,<br />
2002; Penot, 2001), even under harsh environments (e.g., the Soqotra Island of<br />
Yemen; Ceccolini, 2002). In some studies, the socioeconomic variables are taken<br />
into account (e.g., Mendez et al., 2001; Penot, 2003; Wezel and Bender, 2003) for<br />
analyzing the system’s functions but most do not describe the socioeconomic<br />
attributes with the same rigor as that of the ecological variables. Some studies<br />
dealing with bio-economic modeling of agroforests are also restricted to the<br />
cropping system-level (e.g., Purnamasari et al., 2002). Issues such as labor needs<br />
and returns, investments and return-on-investments in the mid- and long-term,<br />
product benefits, and income generation might be described, but they are seldom<br />
presented as arguments for adoption, or even taken into account in the innovation<br />
process behind the adoption of agroforests. In other words, the overall advantages as<br />
well as positive externalities of agroforests are widely recognized but not properly<br />
valued. Direct benefits of agroforests at farm-level are generally underrated and<br />
more so at the community and landscape levels.
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
271<br />
The only two economic variables which seem to provide convincing arguments<br />
are: (1) diversification linked with the spreading of risk, income and labor, and (2)<br />
income generation as a whole (e.g., Torquebiau, 1992; Mendez et t al., 2001; Penot,<br />
2003; Wezel and Bender, 2003; Wiersum, 2004). The large number of products of<br />
agroforests and their uses may explain the difficulty to go beyond mere description<br />
and quantify these in economic terms. Similarly, the links between diversification,<br />
risk buffering capacity and long-term economic and ecological sustainability have<br />
not been sufficiently taken into account so far. The role of risk and uncertainty has<br />
been studied in agroforestry adoption (Mercer, 2004) but not as an innovation<br />
process in itself.<br />
Yet, tree homegardens cover significant land areas. For example, they occupy<br />
20% of arable land of Java, Indonesia (Jensen, 1993). It has been shown that the<br />
economic functions of these “pekarangans<br />
“ ” (see Wiersum, 2006) contribute to<br />
social equilibrium (Mary and Dury, 1997). A study of their patrimonial value<br />
demonstrated that durians (Durio zibethinus, a popular fruit tree) in these Javanese<br />
homegardens have a significant economic importance, both as a source of income<br />
and as an insurance mechanism in informal financial systems (Dury et al., 1997).<br />
There are more than 5 million homegardens in Kerala State, India (Kumar and Nair,<br />
2004) – another homegarden ‘hotspot’. Three million hectares of jungle rubber<br />
(Hevea brasiliensis-based agroforest) provide more than 50% of the total rubber<br />
production of Indonesia and there are another 2 million ha of various agroforests in<br />
Indonesia (Penot, 2001). Multistrata agroforests are also known in Brazil,<br />
Cameroon, Ghana, Nigeria, Tanzania, Sri Lanka, and other countries (Kumar and<br />
Nair, 2004). Agroforestry homegardens can also be observed in many tropical<br />
countries, both on agricultural frontiers and in stabilized agricultural landscapes.<br />
Although a worldwide estimation of the contribution of these cropping systems to<br />
agricultural production has not been made, it is now accepted that their contribution<br />
is far from negligible, be it in terms of traded products, fuelwood, subsistence crops,<br />
nutritional value, medicinal plants, timber, etc. If farmers worldwide have developed<br />
such systems, it is certainly not because they mimic the forests or foster biodiversity<br />
conservation—there must be something else as discussed hereunder.<br />
1.2. Need for a specific economic analysis<br />
We argue that there is an economic rationale explaining the importance of<br />
agroforests worldwide, but that this rationale is relatively complex to identify and<br />
measure. First, there is a well-known complementarity between direct sales of<br />
agroforestry products (timber, fruits, legumes, resins, nuts, rattan, medicinal<br />
products, etc.) and self-consumption by the garden owner, which leads to significant<br />
savings in the households’ day-to-day expenses. Secondly, it has been shown that<br />
long-term patrimonial strategies are of utmost importance to farmers growing<br />
agroforests (Mary and Dury, 1997); yet, conventional economic analyses based on<br />
discounting rates hardly serve for such perennial, multi-component and multi-cycle<br />
systems, where future discounted values of tree products are difficult to anticipate<br />
and as such seldom taken into account by farmers in their planting choices<br />
(Torquebiau et al., 2002), unless the harvested products are easily marketable and
272<br />
they generate a net margin which covers replanting costs (e.g., clonal rubber).<br />
Finally, farmers also plant and tend agroforests for their social functions (land<br />
tenure, social status and living environment). So, while scientists have repeatedly<br />
said that agroforests are environmentally sound, that alone is probably not a major<br />
motivation for farmers.<br />
The objective of this chapter is to try and show that the reason behind the<br />
“enigma of tropical homegardens” (Kumar and Nair, 2004) lies in elements of<br />
positive externalities, which are not accounted for in standard economic analyses,<br />
yet matter to the farmers and perhaps to other stakeholders (e.g., timber for<br />
sawmills). If agroforestry scientists want to convince farmers and policy makers that<br />
agroforests are more than just relics of the past and are worthy to be considered as<br />
land use options, then appropriate economic analyses of agroforests need to be done<br />
covering the ecological services (e.g., watershed protection, nutrient-cycling, carbon<br />
sink, bio-habitat functions, and biodiversity maintenance) as well as social, cultural<br />
and aesthetic values.<br />
Following Coase (1960)’s analysis of social cost, we make a difference between<br />
“giving a value to a service” (potentially, but not automatically tradable) and<br />
“paying for a service” (which leads to the “who is going to pay” question). Taking<br />
into account (assigning a value to a service) or internalizing positive externalities<br />
(paying for a service) relate to resources or services that cannot be included in<br />
private accounting because they are public goods (e.g., landscape beauty, pollinating<br />
insects) or because they are preserved for future generations (e.g., biodiversity, soil<br />
resources). We argue here that such “global goods,” considered as services to the<br />
community, need be taken into account not only by international negotiations such<br />
as discussions on climate change or biodiversity, but also in agricultural policies and<br />
incentives, and, as a consequence in the farmers’ day-to-day decision-making<br />
processes.<br />
One of the services that are likely to be taken into account in the future is the<br />
carbon sink function of the Clean Development Mechanism (CDM), as scheduled in<br />
the Kyoto Protocol. Rubber being the only tree crop (beside timber trees) eligible for<br />
CDMs, rubber-based and timber-based agroforests will theoretically be eligible. In<br />
such a case, their carbon sink service can be valued and considered in the trade or<br />
exchange of pollution rights (O.J. Cacho, pers. comm., 2002).<br />
2. AGROFORESTS AS CROPPING SYSTEMS PROVIDING MISCELLANEOUS<br />
GOODS AND SERVICES<br />
2.1. Multiple roles<br />
E. TTORQUEBIAU AND E. PENOT<br />
Farmers worldwide, but especially those in the developing countries, do not focus<br />
only on agricultural production. They are concerned first and foremost about their<br />
family priorities and are seldom sensitive to global issues such as biodiversity<br />
conservation or carbon sequestration; they nevertheless contribute to a series of<br />
goods and services that are not always marketed or even recognized. This multifunctional<br />
role of agriculture is now recognized and promoted in some regions (e.g.,
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
273<br />
Europe) in contrast to merely “production-oriented” agriculture. This has also led to<br />
the reduction in direct subsidies to production but subsidizing the environmental<br />
functions of farms.<br />
Agroforests can fulfill this multi-functional role better than other farming<br />
systems because they have more positive externalities than other monocultures or<br />
simpler agroforestry systems. So they deserve a specific economic analysis taking<br />
into account both goods and environmental services as well as short- and long-term<br />
issues. Agroforests, homegardens in particular, combine perennial-based production<br />
with long-term strategies (e.g., resin, nuts, fruits, and timber production) and<br />
shorter-duration food crops (e.g., legumes, cassava – Manihot esculenta, and banana<br />
– Musa sp.) with a short-term perspective. Farming systems models can include<br />
components on externalities or services to analyze this multifunctional feature. It<br />
might, however, be easier to handle the benefits of some services such as<br />
biodiversity conservation at regional- or macro-level. While priority has so far been<br />
on plant biodiversity, some studies have shown the role of agroforests as wildlife<br />
buffer zones (Nyhus and Tilson, 2004).<br />
Another important role is the generation of a “forest rent” as defined by Ruf<br />
(1987), i.e., the reduction of costs and risks of perennial plantation establishment –<br />
thanks to the forest’s positive externalities such as soil quality, weed and pest<br />
control. This concept has been extended to agroforests by Penot (2001), who showed<br />
that agroforests did maintain (sometimes improve) the forest rent while conventional<br />
monoculture plantation crops such as cacao (Theobroma cacao), coffee (Coffea<br />
spp.), and oil palm (Elaeis guineensis) generally consumed (part of) it.<br />
Agroforests have some constraints too, however. Since crop mixtures are the<br />
rule, some crops are favored while others are not; and agroforests may provide small<br />
quantities of a given crop that are not always saleable, except locally. Crops may<br />
also change with time; e.g., rice, maize or cassava may be initially intercropped with<br />
young trees but will not yield optimally under an increasing intensity of shade,<br />
which necessitates their replacement with shade tolerant crops (e.g., beans, some<br />
banana varieties). Similarly, rattan vines intercropped in rubber agroforests will not<br />
be harvested during peak rubber production but rather at the end of rubber trees’<br />
lifespan because rattan harvesting tends to damage tree canopies.<br />
High reliance on manual labor and limited markets for specific products are<br />
other significant features in this respect. Delayed production (from large-sized trees)<br />
delays return on investment. Most farmers use non-improved plants and the quality<br />
can be variable, a potential problem for export of fruits, although there can also be a<br />
niche market for “organically grown” local varieties. However, some agroforests<br />
(e.g., rubber agroforestry systems) also rely on fertilizers and improved planting<br />
materials (e.g., rubber clones and grafted fruit trees).<br />
Overall, agroforests are specific cropping systems, which display a range of<br />
specifications making them more difficult to analyze than the monocropping or even<br />
multiple cropping systems with annual crop associations. It can be argued that it is<br />
this lack of analysis that has hampered the efforts of agronomists and extension<br />
agents to promote agroforests and hindered research to reach beyond the descriptive<br />
studies and into the stage of analytical research.
274<br />
3. SUSTAINABILITY OF AGROFORESTS<br />
Sustainability of agroforests can be explained based on different factors and criteria.<br />
Ecological sustainability stems principally from biodiversity conservation, natural<br />
resource management (soil and water), the control of pollution (little or no use of<br />
agrochemicals) and phytoremediation. Against today’s global change challenge,<br />
agroforests represent an important carbon sequestration potential (Kumar, 2006).<br />
Economic sustainability is based on the consideration that agroforests are able to<br />
provide in the long-run a stable and diversified source of income and are viewed as<br />
patrimonial assets (i.e., contributing to the long-term wealth and inheritance of the<br />
family; Mary and Dury, 1997). A large proportion of the local, traditional farming<br />
knowledge is related to agroforests. The risk buffering capacity of agroforests<br />
contributes to both ecological and economic sustainability. Social sustainability<br />
might be achieved through land tenure security linked to tree growing and<br />
preservation of community values. Institutional sustainability might be seen through<br />
the fact that agroforests can be individually or commonly managed. Table 1<br />
summarizes some arguments that link agroforests with sustainability.<br />
Table 1. A summary of sustainability attributes of agroforests.<br />
Ecological Economic Social and institutional<br />
- reduced soil erosion<br />
- high soil organic matter<br />
content<br />
- buffered soil moisture and<br />
temperature<br />
- closed nutrient cycling<br />
- improved soil physicochemical<br />
properties<br />
- efficient use of light and<br />
water<br />
- high wild plant and animal<br />
biodiversity<br />
- use of endogenous resources<br />
- contribution to on-farm<br />
production of wood and fuel<br />
wood<br />
- high soil biotic activity<br />
- better scope for evolution<br />
and diversification of<br />
economic plants<br />
- differentiated vertical and<br />
horizontal management<br />
zones and related ecological<br />
niches<br />
- potential for organically<br />
grown products<br />
E. TORQUEBIAU T AND E. PENOT<br />
- significant use of<br />
endogenous resources<br />
- high safety factor against<br />
marketing and<br />
seasonality hazards<br />
- reduced cash needs<br />
- high and diversified biophysical<br />
outputs (plant<br />
and animal food,<br />
medicines, fibers, etc.)<br />
- socio-economic outputs<br />
diversified and<br />
distributed over time<br />
- balance between<br />
subsistence and cash<br />
income<br />
- building up of capital<br />
- boosting rural industries<br />
and employment<br />
- adjustment to varied<br />
contexts<br />
- yield stability<br />
- management flexibility<br />
(intensive vs. extensive)<br />
- economic resilience<br />
(value as “land reserve”)<br />
- reduced and flexible<br />
labor needs<br />
- contribution to<br />
nutritional security<br />
- contribution to<br />
community<br />
socialization<br />
- preservation of<br />
traditional knowledge<br />
- biodiversity linked to<br />
traditions and practices<br />
- key role of women<br />
- equitable distribution<br />
of products<br />
- land reserve function<br />
(for alternative<br />
landuses)<br />
- maintenance of access<br />
rights to common<br />
goods (e.g., fruits)<br />
- flexibility of ownership<br />
(private vs. communal)<br />
Source: Adapted from Torquebiau (1992), Penot (2003), and Kumar and Nair (2004).
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
275<br />
Kumar and Nair (2004) rightly point out that homegardens (i.e., not all<br />
agroforests) may be on the verge of extinction due to new trends in agrarian<br />
structure, high market-orientation, demographic pressure, land fragmentation, and<br />
cultural dynamics. In the face of such constraints, the ecological foundations of<br />
homegardens may not be sufficient to warrant their survival. However, Javanese<br />
homegardens keep their place and role with an average population density of more<br />
than 800 persons km –2<br />
m , and a strong market-orientation of agriculture (Wiersum,<br />
2006). Presence of some high value crops (e.g., durian) may probably explain this.<br />
Interestingly, Java is not the only place where a positive correlation is observed<br />
between number of trees per unit area and human population density. Other<br />
examples include Kenya (Tiffen et al., 1994), Kerala (India) and Sri Lanka.<br />
Often multistrata agroforests are also under the influence of changing economic<br />
factors. For instance, jungle rubber and damar (Shorea javanica) gardens of<br />
Indonesia are facing international price fluctuations (e.g., rubber price moving from<br />
2 US $ kg –1 in 1996, to 0.6 in 2001, and then back to 1.2 in 2004). Furthermore,<br />
diversification of local farming may be at the expense of traditional agroforests, e.g.,<br />
massive investments in industrial crops such as oil palm. The recent push toward<br />
globalization impacts the traditional farming practices in a myriad of ways among<br />
which access to market and marketing procedures rank high. In Asia, for instance,<br />
most export products have long been linked with international prices (rubber, oil<br />
palm, coffee and cocoa). The commodity boards established in Africa in the 1970s<br />
to protect farmers from price volatility have failed to deliver the expected results and<br />
their relevance is now being questioned. Thus, globalization has a stronger impact<br />
on African farmers than their Asian counterparts, who used to adapt better to the<br />
international markets and price cycles. We suggest that agroforests play a role in this<br />
adaptability; yet new policies of decentralization and local governance, new rules for<br />
access to credit, projects or information may impact it. It is, however, speculative as<br />
to whether agroforests will be able to react to such changes more efficiently than<br />
conventional monocropping.<br />
4. CHALLENGING THE REAL ECONOMIC IMPACT OF AGROFORESTS<br />
The sustainability advantages of agroforests come from a trade-off between<br />
ecological and socioeconomic attributes. Conventional economic approaches may be<br />
inadequate for integrating these attributes in a comprehensive manner, because (1)<br />
farmers manage agroforests for a variety of objectives, (2) the ecological benefits<br />
are not internalized in existing analyses, and (3) some ecological attributes have no<br />
current market value.<br />
Furthermore, if neoclassical economics are used to assess the performance of<br />
agroforests, the criteria of yield, cost-benefit analysis and net present value may end<br />
up giving agroforests poor ratings compared to conventional monocropping<br />
activities, because the analysis will exclude a series of agroforests’ outputs, which<br />
are not traded in the market or insufficiently taken into account in farm economics;<br />
Indonesia’s jungle rubber is a case in point. While it has been a major opportunity<br />
for poor farmers at the agricultural frontier for years, it is now becoming obsolete<br />
compared to clonal rubber monoculture, in terms of yields and labor productivity
276<br />
(Penot, 2001). However, it is difficult to measure or assign economic values to<br />
intangible services and positive externalities. For instance, carbon sink values of tree<br />
crops and forests are currently available but no one can choose among various prices<br />
suggested by experts as long as carbon markets are not functional. Risk-buffering<br />
potential of agroforests, as in situations of climatic variations and commodity price<br />
volatility, also deserves to be measured. The key question behind this is: how to<br />
make a measurement of the agricultural sustainability of agroforests? Perhaps farmsystem<br />
models used in farming system research could be a useful tool for such<br />
comparative assessments.<br />
4.1. Farming system level approach<br />
E. TTORQUEBIAU AND E. PENOT<br />
A pragmatic approach could be first to analyze at the household-level the cost saved<br />
by using products provided by agroforests for items that would otherwise need to<br />
have been purchased (e.g., building and fencing materials, food, medicines and raw<br />
materials for handicraft). Next, the accounting for environmental benefits might be<br />
performed at the household-level by compiling data over at least a year. Farming<br />
system modeling (e.g., with a software like “Olympe”) 1 is useful to process data on<br />
production, value, cost of production and labor, in order to be able to compare<br />
returns to labor and gross margin per cropping systems at the farm-level. Olympe<br />
performs whole-farm analysis in terms of resources, land, labor and other<br />
opportunities. It is a simulation tool for farm management advice which includes a<br />
“hazard” module that takes into account uncertainties, externalities (both positive<br />
and negative), as well as scenario definition according to risks. It can also be run at<br />
the regional level and with farmers’ groups. An analysis can be made in terms of<br />
income source, return to labor or investment, and linkage between strategic choices<br />
and production factor allocation, in order to assess the relative importance and real<br />
impact of cropping systems within the farming system. The combination of farm<br />
modeling with economic quantification, a historical perspective and the “contextualization”<br />
of farmers’ decisions according to political, socioeconomic, non-market<br />
(ecological)- and market factors provide the explanatory factors of a given farming<br />
system. Typically, the software allows re-interpreting the role of agroforests, as<br />
cropping systems within a farming or regional system.<br />
Under this approach, farmers’ strategies on labor, capital and land use are<br />
analyzed holistically (i.e., at the level of all enterprises of a farm, and not only at the<br />
level of one or the other cropping system). This is crucial to detect the place of<br />
agroforests in the overall farming strategy, because agroforests seldom produce the<br />
main staple food (Michon and de Foresta, 1999), and are invariably one cropping<br />
system among others on a farm. This approach, developed for the rubber farmers of<br />
Indonesia 2 , allows analyzing the diversification of opportunities for farmers facing<br />
an economic crisis and a political change that, in turn, can trigger significant<br />
changes in the social framework.
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
4.2. A social-ecological perspective<br />
277<br />
While a farming system approach can pave the way for a better understanding of<br />
agroforests’ roles, there is also a need for a renewed approach to agroforest analysis<br />
which can deal with higher levels of complexity and translate their “socialecological<br />
3 ” performance into economic performance. An apparently non-rational<br />
behavior that has been observed in Indonesia is the maintenance of old rubber<br />
agroforests along with economically very profitable oil palm plantations. One<br />
hypothesis was that agroforests would gradually leave the way for oil palm<br />
plantations. Social value (land control), possibilities of agroforest improvement<br />
(clonal rubber), and diversification strategies eventually may lead to a new<br />
development of improved rubber agroforests, which remain within the financial<br />
possibilities of local farmers with no access to credit, or even insufficient capital<br />
building capability. Meanwhile, whatever the important gains in return to labor and<br />
net margin provided by oil palm, agroforests have never disappeared – a proof of the<br />
value of such systems in a social perspective. Agroforests as “reserve land factor” or<br />
“long-term land control factor” might not have a direct value but do have an indirect<br />
value as a capital reproduction factor or as a potential expanding factor.<br />
Patrimonial analysis based on the evolution of capital building and asset<br />
transmission could be used for agroforests considered as reserves of land which can<br />
be traded, and since large-sized trees constitute a strategy for the build-up of capital<br />
for further investment. Long-term multi-cycle analyses may provide a framework to<br />
understand the farmers’ behavior and strategies. Economic analyses of mixtures of<br />
plants with different cycles can also be done through farming system modeling.<br />
Smoothening of long-term and patrimonial strategies (Mary and Dury, 1997;<br />
Torquebiau et al., 2002) may help taking into account the time factor and historical<br />
perspective (e.g., capital accumulation and building capacity). A multi-criteria<br />
analysis at both farm and community level is far more powerful than simple<br />
conventional cost-benefit analysis at cropping system level. Again, linking crucial<br />
social aspects (and their consequences in terms of use of production factors) with the<br />
economic analysis may provide a reliable framework that can take into account all<br />
cultural and non-merchantable aspects. Unfortunately, since methods for valuation<br />
of non-tangible social and cultural benefits of agroforestry are practically nonexistent<br />
(Kumar and Nair, 2004), it is difficult to substantiate the above (Penot and<br />
Deheuvels, 2006); rather, it is a plea for future research on these issues.<br />
4.3. Subsistence versus cash income generation<br />
The merits of agroforests in terms of subsistence for families, flexibility in crop<br />
production or reduced external input requirements also need to be taken into<br />
account. The comparison between farms with and without agroforests may show the<br />
savings and impact on household’s income. However, not all agroforests are food<br />
crop-based. Some agroforests are totally cash-oriented, e.g., rubber (jungle rubber),<br />
resin (damar agroforest), spices (e.g., cinnamon: Cinnamomum zeylanicum), fruits<br />
(durian) and timber-based agroforests.
278<br />
E. TTORQUEBIAU AND E. PENOT<br />
The flexibility in crop and tree production in agroforests relates to the different<br />
phases with mature and immature periods of trees or crops. Therefore, it is essential<br />
to take into account the life cycle of plants to implement an economic analysis in the<br />
long run. Specific discounting rates may be necessary as cycles may extend up to 40<br />
or 50 years. Different scenarios are necessary, as this may introduce bias in valuing<br />
products according to the discounting rates chosen. For instance, in tree crop-based<br />
agroforests, rubber or resin is produced for more than 30 years when annual and<br />
biennial crops are generally produced only in the first 3 to 6 years. Timber can be<br />
harvested only at the end of the agroforest’s life-span. Therefore, if detailed data are<br />
available to obtain a reliable assessment of real income (including self-<br />
consumption), system comparison will be more valuable than absolute data (Penot,<br />
2001).<br />
4.4. Landscape amenity and social conviviality<br />
The role of agroforests in providing services such as landscape beauty and aesthetics<br />
or social interaction or social status improvement has also to be incorporated in the<br />
assessment. It seems clear that in many situations, agroforests, and in particular, the<br />
non-private agroforests managed by local communities, and as such considered as<br />
public goods with limited and shared access (for fruits, timber etc.), have a social<br />
importance. The “Tembawang” of the Dayak people in Kalimantan (Indonesia) is a<br />
typical example. Besides being a reserve of forest products through “extractivism,”<br />
when original forests will have disappeared, such agroforests generally include<br />
important social components such as graveyards or may play a role of protection<br />
through the maintenance of a “green belt” around the village. Even if there is no<br />
economic value to this service, its social value will be a compelling reason for the<br />
maintenance of such agroforests and generally prevent its destruction.<br />
5. THE MICRO-ECONOMIC APPROACH<br />
Obviously, many specific features of agroforests might not be purely valued as<br />
goods. Social values, long-term strategic value of land, and risk buffering are<br />
examples; yet they provide powerful incentives to advocate agroforest development.<br />
With farming system modeling and a prospective approach, it is possible to assess<br />
the effects on risks. A prospective analysis with scenarios can lead to identification<br />
of economic thresholds and boundaries 1,2 and enables the definition of an economic<br />
feasibility domain (or expected economic outputs), i.e., the range within which the<br />
system is economically viable.<br />
If agroforests’ benefits can be analyzed through market values of their products<br />
and services, then neo-classical environmental economics can be used and<br />
externalities can be included (or re-internalized) into the process of income<br />
generation. Growth or pollution costs and delay may be taken into account as<br />
negative externalities or constraints to further development. Environmental services<br />
(for example, carbon sequestration potential: Albrecht and Kandji, 2003;<br />
Montagnini and Nair, 2004; Kumar, 2006) can be valued according to a “system of
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
279<br />
values” recognized locally as relevant at a higher, community or provincial level.<br />
The real problem is, therefore, to see whether farmers can potentially or do really<br />
take benefit of externalities and positive advantages of agroforestry. The payment of<br />
environmental services as promoted by the RUPES project (South-Sumatra and<br />
Lampung provinces, Indonesia) 4 provides some evidence in this respect. Other<br />
examples include the potential of agroforestry to reach the millennium development<br />
goals (Garrity, 2004) and the application of the Kyoto mechanisms to rubber trees<br />
(Hamel and Eschbach, 2001). Research on rubber agroforestry in Indonesia<br />
(Lawrence, 1996) provides an important data-set on these issues.<br />
In the context of most developing countries, huge income gaps due to strong<br />
social stratification, information asymmetry, high transaction costs and institutional<br />
failures have strong implications on local economies. Microeconomics allows<br />
accounting for environmental assets, complexity, and uncertainty, and involves<br />
stakeholder participation. When dealing with agroforests, benefits that relate to<br />
public goods or goods that cannot be given a market price because they are for<br />
future generations (e.g., biodiversity, landscape amenity, carbon sink and cultural<br />
and aesthetic values) need to be assessed through a new perspective. A multifunctional<br />
approach, similar to that developed by the Common Agricultural Policy<br />
for European farmers (Dévé, 2004), can provide ideas to take these externalities into<br />
account. New mechanisms such as the CDMs could be explored, in particular for<br />
global issues such as biodiversity conservation.<br />
Agroforest attributes should also be considered in national accounting. Policy<br />
makers should acknowledge the fact that if resource depletion is taken into account<br />
through an environmental economics approach; agroforests will rank very high<br />
among land use options because they generate an “agroforest rent” which is much<br />
higher than the rent from conventional agriculture or other forms of resource<br />
exploitation (e.g., logging, mining the soil through excessive harvests). Farmers<br />
contributing to this resource rent could hence be given direct or, better, indirect<br />
incentives (e.g., tax exemption) to stimulate land use options, which contribute to<br />
such public goods for current and/or future generations.<br />
To reach a status where agroforests could be recommended among other land use<br />
options, they need a reference framework, which takes into account these alternative<br />
economic analyses. Unfortunately, such analyses are lacking at present. In the<br />
meantime, multistrata agroforestry systems will continue being rejected or<br />
marginalized by conventional literature as not fitting into the mainstream economics<br />
and hence in development objectives. Be it for commercially oriented agroforests or<br />
subsistence oriented homegardens, a long-term perspective must be part of farmers’<br />
strategy. However, there is obviously a biased debate between short-term<br />
(economics) vs. long-term (ecology) issues. In both cases, farmers have developed<br />
long-term farming practices through a long haul innovation process that eventually<br />
takes into account economics through the risk buffering capacity of agroforests. In<br />
most cases, social organization is deeply linked with technical constraints in<br />
production, food reliance, income securing and, eventually, land control. There is a<br />
strong coherence between technical pathways and social systems (Penot, 2003).<br />
Customary laws take into account this important point and are generally able to<br />
adapt to changes. There is an economic strategy behind maintaining agroforestry
280<br />
E. TTORQUEBIAU AND E. PENOT<br />
practices that have proved to be able to secure production and maintain control on<br />
land. In other words, long-term economics are totally associated with ecology and<br />
sustainability. An appropriate economic analysis should actually take care of the<br />
long-term aspects. One main challenge for the immediate future, however, is to take<br />
further steps towards the internalization of externalities, providing a value to<br />
services through a multifunctional approach and giving value-added objectives to<br />
ecological criteria.<br />
6. CONCLUSIONS<br />
If an economic perspective with emphasis at local and regional level is applied to<br />
integrate positive externalities such as agrobiodiversity management, improved<br />
nutrient cycling, integrated pest management, ecological sustainability and services,<br />
decision-makers may be convinced that homegardens and agroforests are highly<br />
profitable ventures. If an “agroforest rent” approach is adopted, policy makers and<br />
development officers will see a long-term profitable investment in agroforests.<br />
Hopefully, this will lead to agroforests being given better consideration than at<br />
present in research and development programs worldwide. Furthermore, if<br />
agroforests are still a success-story with many farmers, it is obviously not because of<br />
biodiversity conservation. Other values such as social values, security, diversity,<br />
land control and reserve (including land and tree tenure) are probably important.<br />
There is also a need for a mechanism for the societal or community payment of<br />
those external and social benefits. A micro-economic analysis at the farming system<br />
level including all sources of income, cost-benefit per activity and return to labor,<br />
can explain such long-term strategies, provided they take into account the dynamics<br />
(“time effect”) of perennial crops in homegardens and other agroforests.<br />
Economic analysis methods using farming system modeling which integrate the<br />
outputs of mixtures of plants with different cycles and allow for the smoothening of<br />
long-term and patrimonial strategies are required to explain with accuracy what the<br />
farmers do and why they do so. Agroforests, despite their positive externalities and<br />
advantages are not a “panacea” but seem to be an ideal compromise between<br />
sustainability and risk spreading.<br />
ACKNOWLEDGEMENTS<br />
We acknowledge the useful comments by Stefano Farolfi, Anne Marie Izac, Patrice<br />
Levang, and three anonymous referees on an earlier version of the manuscript.<br />
ENDNOTES<br />
1. Penot E., Le Bars M., Deheuvels O., Le Grusse Ph. and Attonaty J.M. 2004.<br />
Farming systems modelling in tropical agriculture using the software<br />
“Olympe.” ECOMOD Workshop, June 2004, Paris.<br />
2. Penot E. and Hébraud C. 2003. Modélisation et analyse prospective des<br />
exploitations hévéicoles en Indonésie: Utilisation du logiciel Olympe pour la
ECOLOGY VERSUS ECONOMICS IN MULTISTRATA AGROFORESTS<br />
281<br />
définition de scénarios d'évolution en fonction de choix techniques et des aléas.<br />
Modélisation des exploitation agricoles: les multiples usages du logiciel<br />
Olympe. CIRAD Workshop, September 2003, Montpellier.<br />
3. The term “social-ecological” implies an interactive system of equally important<br />
social and ecological parts, while the conventional “socio-ecological” has the<br />
simple connotation of an ecological system with some social aspects (Sayer and<br />
Campbell, 2004).<br />
4. Van Noordwijk M., Chandler F.J. and Tomich T.P. 2004. An introduction to the<br />
conceptual basis of RUPES: rewarding upland poor for the environmental<br />
services they provide. ICRAF Southeast Asia, Bogor, 46p.<br />
REFERENCES<br />
Albrecht A. and Kandji S.T. 2003. Carbon sequestration in tropical agroforestry systems.<br />
Agric Ecosyst Environ 99: 15 – 27.<br />
Ceccolini L. 2002. The homegardens of Soqotra island, Yemen: an example of agroforestry<br />
approach to multiple land use in an isolated location. Agroforest Syst 56: 107 – 115.<br />
Coase R.H. 1960. The problem of social cost. J Law Econ 3: 1 – 44.<br />
Dévé F. 2004. Major findings and conclusions on the role of agriculture. RAO Project of<br />
FAO, Phase 1. FAO, Rome, 18p.<br />
Dury L., Vilcosqui L. and Mary F. 1997. Durian trees in Javanese homegardens: their<br />
importance in informal financial systems. Agroforest Syst 33: 215 – 230.<br />
Gajaseni J. and Gajaseni N. 1999. Ecological rationalities of the traditional homegarden<br />
system in the Chao Phraya Basin, Thailand. Agroforest Syst 46: 3 – 23.<br />
Garrity D.P. 2004. Agroforestry and the achievement of the millennium development goals.<br />
Agroforest Syst 61: 5 – 17.<br />
Hamel O. and Eschbach J.M. 2001. Impact potentiel du MDP dans l’avenir des cultures<br />
pérennes: état des négociations internationales et analyse prospective à travers l'exemple<br />
de la filière de production du caoutchouc naturel. Oléagineux Corps Gras Lipides 8:<br />
599 – 610.<br />
Jensen M. 1993. Productivity and nutrient cycling of a Javanese homegarden. Agroforest Syst<br />
24: 187 – 201.<br />
Kaya M., Kammesheidt L. and Weidelt H.J. 2002. The forest garden system of Saparua<br />
island, Central Maluku, Indonesia, and its role in maintaining tree species diversity.<br />
Agroforest Syst 54: 225 – 234.<br />
Kumar B.M. 2006. Carbon sequestration potential of tropical homegardens. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 185 – 204. Springer Science, Dordrecht.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Lawrence D.C. 1996. Trade-offs between rubber production and maintenance of diversity: the<br />
structure of rubber gardens in West Kalimantan, Indonesia. Agroforest Syst 34: 83 – 100.<br />
Mary F. and Dury S. 1997. Les fonctions économiques méconnues des jardins villageois à<br />
Java-Ouest. Fruits 49: 141 – 150.<br />
Mendez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: mico-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Mercer D.E. 2004. Adoption of agroforestry innovations in the tropics: a review. Agroforest<br />
Syst 61: 311 – 328.
282<br />
E. TTORQUEBIAU AND E. PENOT<br />
Michon G. and de Foresta H. 1999. Agro-forests: incorporating a forest vision in agroforestry.<br />
In: Buck L.E., Lassoie J.P., and Fernandes E.C.M. (eds.) Agroforestry in sustainable<br />
agricultural systems, pp 381 – 406. CRC Press, Boca Raton, FL.<br />
Montagnini F. and Nair P.K.R. 2004. Carbon sequestration: an underexploited environmental<br />
benefit of agroforestry systems. Agroforest Syst 61: 281 – 295.<br />
Nyhus P. and Tilson R. 2004 Agroforestry, elephants and tigers: balancing conservation<br />
theory and practice in human dominated landscapes of Southeast Asia. Agric Ecosyst<br />
Environ 104: 87 – 97.<br />
Penot E. 2001. Stratégies paysannes et évolution des savoirs: l’hévéaculture agro-forestière<br />
indonésienne. PhD Thesis, University of Montpellier, Faculty of Economics, France,<br />
360p.<br />
Penot E. 2003. Cohérence entre systèmes techniques et systèmes sociaux et territoires.<br />
Evolution des systèmes de production hévéicoles et gestion de la ressource foncière : le<br />
cas de la province de Ouest-Kalimantan, Indonésie. In: Dugué P. and Jouve P. (eds),<br />
Organisation spatiale et gestion des ressources et territoires ruraux, pp 60 – 68. UMR<br />
SAGERT (CIRAD – CNEARC - ENGREF), Montpellier.<br />
Penot E. and Deheuvels O. (eds) 2006. Du système de culture à la petite région: Modélisation<br />
du fonctionnement de l’exploitation agricole, simulation et aide à la décision avec le<br />
logiciel Olympe. L’Harmattan, Paris (in press).<br />
Purnamasari R., Cacho O. and Simmons P. 2002. Management strategies for Indonesian<br />
rubber production under yield and price uncertainty: a bio-economic analysis. Agroforest<br />
Syst 54: 121 – 135.<br />
Ruf F. 1987. Eléments pour une théorie sur l’agriculture des régions tropicales humides: de la<br />
forêt, rente différentielle au cacaoyer, capital travail. Agron Trop 42: 218 – 232.<br />
Sayer J. and Campbell B. 2004. The science of sustainable development: Local livelihoods<br />
and the global environment. Cambridge University Press, Cambridge, 268p.<br />
Tiffen M., Mortimore M. and Gichuki F. 1994. More people less erosion: Environmental<br />
recovery in Kenya. John Wiley, London, 234p.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Torquebiau E., Mary F. and Sibelet N. 2002. Les associations agroforestières et leurs<br />
multiples enjeux. Bois et Forêts des Tropiques 271: 23 – 36.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
Wiersum K.F. 2004. Forest gardens as an ‘intermediate’ land use system in the nature-culture<br />
continuum: Characteristics and future potential. Agroforest Syst 61: 123 – 134.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.
CHAPTER 16<br />
FINANCIAL ANALYSIS OF HOMEGARDENS:<br />
A CASE STUDY FROM KERALA STATE,<br />
INDIA<br />
S. MOHAN*, J.R.R. ALAVALAPATI, AND P.K.R. NAIR<br />
School of Forest Resources and Conservation, University of Florida, Gainesville,<br />
FL 32611, USA; *Current Address: CREST-RESSACA, Texas A&M University,<br />
Kingsville, MSC 213, 700 University Blvd, Kingsville, TX 78363, USA; E-mail:<br />
<br />
Keywords: Adaptive management, Economic utility, Non-monetary benefits, Resilience,<br />
Sensitivity analysis.<br />
Abstract. <strong>Homegardens</strong> are touted as economically and biologically sustainable systems, but<br />
studies to quantify the economics of these gardens are limited. This study used inventories,<br />
survey information and market data to estimate the productivity of 75 homegardens in<br />
Thrissur district of Kerala state, India, and applied benefit-cost analysis to ascertain the<br />
current financial values of these systems. All homegardens were found to be economically<br />
profitable and also to be of better economic utility to the farmer than selling or leasing the<br />
land. Sensitivity analyses indicated that these systems were easily resilient to 10% shifts in<br />
the prices of hired labor and in the prices of the three most economically important crops:<br />
coconut (Cocos nucifera), arecanut (Areca ( catechu),<br />
and banana (Musa spp.). Profit value of<br />
the gardens tended to increase with holding size and with increasing years of cultivation.<br />
Labor hours (both household and hired) and gender of the decision-maker were not suitable<br />
predictors of profit. Intensity of profit generation was highest in the smaller gardens, thus<br />
perhaps indicating both adaptive management to land constraints, and the presence of other<br />
intangible benefits that might affect land management strategies.<br />
1. INTRODUCTION<br />
<strong>Homegardens</strong> are well developed agroforestry systems consisting of distinct<br />
assemblages of plants with or without livestock, intensively managed within the<br />
residential compound. Economic theories and methodologies relating to agroforestry<br />
283<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 283–296.<br />
© 2006 Springer. Printed in the Netherlands.
284 S.MOHAN ET AL .<br />
systems are well documented (Alavalapati et al., 2004); however, rigorous field<br />
studies that apply these concepts to homegardens are rare (Nair, 2001). One of the<br />
major constraints to implementing some of these concepts stems back to an<br />
observation made by Scherr (1992) regarding the lack of guidelines for data<br />
collection and analysis. Preliminary economic analyses in Central America and the<br />
Caribbean have indicated that many agroforestry systems are profitable even at real<br />
discount rates of 20% or higher (Current et t al., 1995), yet economic studies relating<br />
to homegardens are limited. The economic worth of homegardens is especially<br />
difficult to quantify due to three reasons: these systems have high, yet variable levels<br />
of biodiversity, making data collection time-intensive and error-prone; these systems<br />
provide some benefits that are designed to be of particular use to certain farmers<br />
only; and finally, these are established systems, some of which have existed for<br />
many hundreds of years, and the benefits realized in the past may not be accurately<br />
quantified because of the inadequate availability of data. Different methods of<br />
quantification of intangible benefits, which are outside the scope of this chapter, are<br />
now increasingly studied and might potentially, be used to address these non-market<br />
values.<br />
<strong>Homegardens</strong>, although primarily used for subsistence purposes of the<br />
household, are increasingly being used to generate cash income (Christanty, 1990;<br />
Torquebiau, 1992; Mendez et al., 2001). They are also used to generate non-market<br />
benefits such as aesthetics, ornamentation, improved food quality, and nutritional<br />
security to the farmers (Karyono, 1990; Jose and Shanmugaratnam, 1993; Drescher,<br />
1996). With the overall aim of using a combination of different methodologies to<br />
assess the current tangible financial status of existing homegardens and providing a<br />
set of guidelines for data collection and analysis, a study was carried out in Kerala,<br />
India (Mohan, 2004). This chapter forms a part of that investigation and deals with<br />
the cost-benefit analyses for one year, and sensitivity studies to ascertain the<br />
economic resilience of Kerala homegardens to market fluctuations. The net values of<br />
these gardens were also compared with other available economic alternatives.<br />
2. METHODOLOGY<br />
2.1. Study location, sampling and economic evaluation<br />
The study was conducted in Thrissur District of central Kerala (between 10 o and<br />
10 o 47’ N latitude, and 75 o 55’ and 76 o 54’ E longitude). Kerala is one of the<br />
southernmost states of India, with a coastline of approximately 600 km and a<br />
tropical monsoonal climate. Thrissur experiences an annual precipitation of<br />
approximately 2500 mm. <strong>Homegardens</strong> are the predominant form of agriculture in<br />
the district, along with rice (Oryza sativa) farming and commercial plantations of<br />
coconut (Cocos nucifera), arecanut or betel nut (Areca ( catechu)<br />
and bananas (Musa<br />
spp.). A wide variety of plants are grown in the homegardens including commercial<br />
crops such as coconut and arecanut, starchy foods such as cassava (Manihot<br />
esculenta), and a large number of vegetables and fruits.
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
285<br />
Seventy-five homegardens of Thrissur district were randomly selected, and<br />
systematically (based on location) inventoried during October 2002 – February<br />
2003. These homegardens were located in both rural and semi-urban areas. A<br />
comprehensive survey was administered and the productivity of all homegardens<br />
estimated. The values of the products were determined according to existing market<br />
prices (shadow prices for medicinal plants), and key decision-makers in the selected<br />
homegardens were interviewed.<br />
The study was based on the premise that an analysis encompassing the steps<br />
summarized below would provide an adequate understanding of the economic value<br />
of these gardens in steady state (i.e., no natural calamities or extenuating<br />
circumstances that distinguished the year of study from other years).<br />
• Accounting the costs and benefits for the farmer over a one-year period.<br />
• Assessing the economic resilience of homegardens to market shifts in labor or<br />
crop price patterns by conducting sensitivity analyses.<br />
• Comparing homegardens with other economic alternatives to evaluate the option<br />
that would provide optimal economic utility to the farmer.<br />
Cost and benefit sources were determined based on the farmers’ records, as well<br />
as inventory of the gardens. Plant productivity was based on both yield estimates 1<br />
and farmer records. Market values were determined based on existing prices. Costs<br />
and benefits were assessed at the actual existing prices that the participating farmers<br />
encountered in markets. Many of the costs had already been incurred, such as onetime<br />
costs for building wells and for the initial preparation of land, but they were<br />
added to the total cost involved in maintaining the garden if incurred during the<br />
lifetime of the farmer who owned and farmed the property during the time of the<br />
study. The benefits realized from these costs are usually continuous and stretch over<br />
several years. Therefore, the yearly worth of these benefits was also added to the<br />
annual profits generated from these gardens.<br />
2.2. Opportunity costs of land and household labor<br />
The land tenure and ownership system in Kerala makes land a very valuable<br />
commodity in an increasingly land-deprived social system. Furthermore, the land<br />
occupied by the homegarden almost always houses the residential building, and<br />
these homes are usually inherited by the next generation. Therefore, it is unlikely<br />
that a homegarden will be sold on its own, without the residential building.<br />
However, in order to avoid inflating the financial worth of these systems while<br />
adhering to the observed social and cultural norms of the land, the opportunity costs<br />
of land were assigned values equivalent to the rate at which farmers were able to<br />
lease out all or part of the land. This rent rate was calculated to be an average of<br />
Rupees 12 350 (~ $262) per ha per year (one US $ = ~ Rupees 47, October 2003).<br />
Opportunity cost of household labor (OCHL) was calculated as a function of<br />
time as OCHL= ƒ (t*labor rate), where t is time spent in the garden. If the daily rate<br />
for a hired male laborer in a particular area was Rupees 70, and the owner/farmer<br />
put in an average of four hours work in the garden per day, the household labor costs<br />
were calculated to be Rupees 35(30) = 1050 (~ $22) per month.
286<br />
2.3. Components of the annual financial cycle of Kerala homegardens<br />
Based on farmer surveys and farm inventories, Table 1 presents the inputs and<br />
outputs that are the main components of the annual finances of a typical Kerala<br />
homegarden in steady state. Inputs were determined as any monetary contribution<br />
to the annual economic cycle of the garden and were generally found to comprise of<br />
human labor, seeds, organic and chemical fertilizers, hired labor, one-time costs<br />
such as barn maintenance and equipment (if incurred during the year of study), and<br />
the associated transportation costs. Some of the associated maintenance costs<br />
included transportation of products to markets, de-husking of coconuts, and the<br />
harvesting of coconuts, areca nut, and other market products. Except for<br />
transportation, these tasks were usually performed by hired labor. The farmers<br />
sometimes employed a system called karar r (contract), in which the commercial<br />
produce is leased either in part or full to a buyer, who would undertake all associated<br />
tasks, such as harvesting, transporting and selling, after paying a fixed sum of<br />
money to the owner. Such local barter systems might exist in other geographic<br />
locations around the world, and any financial analysis should take into account these<br />
individual practices and the social and cultural factors that influence these decisions.<br />
The tangible benefits derived from the garden also included products for market<br />
sale, milk and other livestock products, and goods used for household consumption<br />
such as food, firewood and medicinal plants.<br />
Inputs Outputs<br />
Fertilizers<br />
Seeds and seedlings<br />
Animal feed<br />
One-time expenses<br />
Maintenance operations<br />
Land cost<br />
Household labor<br />
S.MOHAN ET AL .<br />
Table 1. Components of the annual finances of a typical homegarden in steady state in<br />
Thrissur district, Kerala, India.<br />
Household products<br />
Market products<br />
Animal products (milk, meat, dairy)<br />
Long-term benefits (timber)<br />
Medicinal plants<br />
Note: Intangible benefits (e.g., shade, aesthetics, and ornamentation) have not been quantified<br />
in this study.<br />
All economically important species were inventoried and the production over the<br />
period of one year was estimated based on farmer reports. The economic inventory<br />
included medicinal plants that might or might not have been used by the farmer<br />
during the course of one-year, but were present in the garden because the gardeners<br />
considered them essential. The values of these medicinal species were included in<br />
those instances where the farmer had occasion to utilize a medicinal plant, by using<br />
a shadow pricing mechanism of estimating the cost involved in obtaining a similar<br />
benefit elsewhere.
287<br />
Economic theory argues that the highest social utility is attained when producers<br />
adopt practices generating the highest rates of return to all available resources,<br />
including all costs and benefits (Scherr, 1992). Economic planners also prefer<br />
investment in those activities yielding the highest rates of return to total resources or<br />
total labor used. However, the adoption decision for farmers is more complicated,<br />
especially in the case of homegardens where they reside within the confines of the<br />
agricultural property. These decisions may be influenced by a desire to maximize<br />
utility of family labor, returns to land, or even nutritional security. Two alternatives<br />
to homegarden cultivation have been considered in this study in order to understand<br />
the extent to which the farmer-needs and desires affect the pure cash flow into the<br />
homegarden system: Option I, entails selling the entire property and the house<br />
(assuming that selling the property without the house might prove to be improbable<br />
in the case of Kerala state) and Option II, in which the homegarden land is leased to<br />
another farmer, while the owner resides in the same house. Both options would<br />
allow the decision-making farmer to seek employment (work as agricultural laborer)<br />
elsewhere, assuming there is a steady demand for labor; yet they would have to pay<br />
to attain all benefits from the homegarden. Option 1 would also require that the<br />
farmer seek out an alternate residence.<br />
2.4. Analysis<br />
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
The collected data were analyzed using the basic economic methods of benefits and<br />
costs comparison, i.e., net financial worth (NFW) = Br r – Cr,<br />
where, B = benefits, C =<br />
costs, and r = year of study. For this, the homegarden products were categorized as<br />
having one of the three levels of economic utility; primary utility: those that are<br />
essential to the household, e.g., cassava, coconuts, and banana; secondary utility:<br />
those that are not absolutely essential but without which the household might suffer<br />
from nutritional deficiencies or other losses, e.g., gourd vegetables, amaranth<br />
(Amaranthus<br />
( spp.), and medicinal plants; and tertiary utility: those that are grown<br />
primarily for personal pleasure, e.g., ornamental plants and flowers, e.g., roses (Rosa<br />
spp.). Some plants are grown for both decorative and medicinal purposes, e.g., the<br />
shoe flower plant (Hibiscus spp.). The value of primary utility plants was quantified,<br />
and the value of the secondary category including medicinals was estimated using<br />
shadow pricing; the tertiary category provides mainly intangible benefits. All tree<br />
and shrub species found in the homegardens are listed in Appendix I.<br />
The sensitivity analyses were conducted by adding a 10% increment to the price<br />
of hired labor, and reduction of 10% in market prices of coconut, arecanut and<br />
banana, which are the main market crops in Kerala. Data were analyzed using the<br />
statistical software, Statistica. Various statistical procedures utilized in the analysis<br />
included analysis of variance (ANOVA) to compare characteristics of different size<br />
categories of homegardens, t-tests for comparison of means assuming unequal<br />
variances, and multivariate regression analyses to determine the predictors of<br />
homegarden profitability.
288<br />
S. MOHAN ET AL .<br />
3. RESULTS<br />
The 75 gardens included in this study had a mean landholding size (excluding the<br />
residential area) of 0.34 ha (± 0.03; median = 0.26 ha). The smallest garden was 0.01<br />
ha in extent, and the largest, 1.0 ha. Although homegardens greater than 1 ha was<br />
initially included in the data collection as part of the random sampling scheme, they<br />
were subsequently excluded from the analysis because they were deemed to be very<br />
large farms that showed more characteristics of sole cropping than that of traditional<br />
homegardening. The gardens included in the study were also subdivided into four<br />
groups (small: 0.26 ha; medium: 0.26 to 0.52 ha; large: 0.52 to 0.78 ha and<br />
commercial: 0.78 to 1.0 ha). Following this, there were 24 small, 14 medium-sized,<br />
10 large, and 27 commercial gardens.<br />
3.1. Economic values of homegardens and annual economic profit<br />
The existing financial worth of all the surveyed gardens, estimated based on the<br />
quantitative values of costs and benefits experienced in the year of study, is<br />
presented in Table 2. All 75 homegardens generated a positive economic value for<br />
the year 2001 – ‘02. Intensity of cultivation as indicated by the generation of profit<br />
per unit area (mean profit/m 2 of homegarden) calculated for the four holding-size<br />
categories was highest for the small gardens. While the commercial gardens yielded<br />
an average profit of Rupees 40.61/m 2 , the small gardens yielded more than double<br />
the average profit at more than Rupees 84/m 2 . Implicit in this is that the intensity of<br />
production is much greater in the smaller gardens, despite net production being<br />
higher in the larger gardens.<br />
Table 2. Mean financial value of homegardens for 2002 – ‘03 based on the benefits and costs<br />
of 75 gardens surveyed in Thrissur district, Kerala, India.<br />
Size of homegarden Mean<br />
financial<br />
value<br />
(Rupees) 1<br />
Mean financial<br />
value including<br />
opportunity<br />
costs of land<br />
and labor<br />
(Rupees)<br />
Intensity of profit<br />
generation 2<br />
Mean<br />
profit/m 2<br />
(Rupees/<br />
year)<br />
Standard<br />
error<br />
Small (≤0.26 ha, n = 24) 62261 46284 84.28 a 10.72<br />
Medium (≤0.52 ha, n = 14) 157524 132759 68.80 b<br />
9.61<br />
Large (≤0.78 ha, n = 10) 256639 225116 76.64 a 11.48<br />
Commercial (≤1.0 ha, n = 27) 275967 214899 40.6 c 4.15<br />
1 Financial worth measured in Rupees (1.00 $US ~ Rs. 47, October 2003).<br />
2 Intensity refers to the mean profit generated per m 2 of cultivated area in the homegarden.<br />
Superscripts (a, b, and c) following a value indicate significant changes in means at = 0.05<br />
in t-tests assuming unequal variances.
3.2. Economic importance of homegarden species<br />
289<br />
The most important contributors to the economic profit generated by homegardens<br />
were coconut, arecanut and banana (both cooking and dessert varieties), but the<br />
distribution of profit varied across garden sizes (Fig. 1). The other economically<br />
important categories in the homegarden were dairy, cashew (Anacardium<br />
(<br />
occidentale), spice trees such as nutmeg (Myristica fragrans), and vanilla (Vanilla<br />
planifolia) (data not presented). Household needs consumed a significant percentage<br />
of the products (more than 50%) in the smaller gardens, while the larger and<br />
commercial gardeners invested most in the commercial production of coconut and<br />
arecanut (Fig. 1).<br />
Figure 1. Contribution of three crop categories and household use to total profit generated<br />
by different size classes of homegardens in Thrissur district, Kerala, India. The holding sizes<br />
are: small: 0.26 ha; medium: 0.26 to 0.52 ha; large: 0.52 to 0.78 ha, and commercial: 0.78<br />
to 1.0 ha.<br />
3.3. Sensitivity analyses<br />
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
Sensitivity analyses are important when evaluating economic benefits, in order to<br />
ascertain the extent to which agricultural systems are susceptible to shifts in the<br />
prices of labor and market products. A majority of the households surveyed (96%)<br />
reported that the prices of hired labor to be the most restrictive aspect of managing<br />
these systems, and coconut, arecanut and banana are the most economically<br />
important crops. A comparison of the data in Table 3 indicates the changes in net<br />
value of the gardens when the labor prices are increased by 10%, and the market<br />
prices of coconut, arecanut, and banana are reduced by 10%. Some of the gardens<br />
that cultivated rubber trees (Hevea brasiliensis) as a component were also very<br />
dependent on it; but rubber was mainly found in the larger gardens, mostly as a sole<br />
crop. Hence, it was excluded from the sensitivity analysis.<br />
The results indicate very low changes in annual profit value across all classes of<br />
homegardens, ranging from 0.24% to 2.46%. The only statistically significant
290<br />
S.MOHAN ET AL .<br />
difference across means was the effect of raised arecanut prices in the commercial<br />
gardens, which ranged from 2.46% for commercial gardens to 0.81% for the small<br />
gardens.<br />
Table 3. Sensitivity analysis to ascertain the economic resilience of homegardens to price<br />
fluctuations in labor and price of three economically significant crops.<br />
Sensitive categories<br />
Percent response in financial worth<br />
(based upon a 10% change in price)<br />
Small Medium Large Commercial<br />
P (hired labor) 0.28 1.12 0.24 0.31<br />
P (coconut) 1.0 2.0 2.8 1.0<br />
P (arecanut) 0.81 1.65 2.21 2.46 *<br />
P (banana) 0.42 0.35 0.74 0.92<br />
P = existing market price; *significant at = 0.05 in comparisons involving small (n = 24),<br />
medium (n = 14), large (n = 10), and commercial (n = 27) using t-test assuming unequal<br />
variances.<br />
3.4. What factors affect the financial value of homegardens?<br />
The multivariate regression model developed to predict the effects of various factors<br />
on the financial values of the surveyed homegardens is as follows and its statistical<br />
parameters are given in Table 4.<br />
Financial worth of homegarden = 4.61 + 0.007(x1) + 0.003(x2)<br />
Where x1 = land area in m 2 and x2 = number of years in cultivation.<br />
Table 4. Coefficients, standard error and probability level of significance of the predictors of<br />
homegarden’s economic worth in Thrissur district in Kerala, India.<br />
Parameter B Standard<br />
error of B<br />
p values<br />
Intercept 4.61 0.073 0.000<br />
Land holding size (m 2 ) 0.007 0.056 < 0.005<br />
Age of garden (years) 0.003 0.001 0.017<br />
Adj. R 2 = 0.447; standard error = 0.319.<br />
The model indicates that the financial value of Kerala homegardens increases<br />
with increasing land holding size and with an increase in the number of years of<br />
cultivation, although both are only modest predictors of profit. The number of hours<br />
of household or hired labor and gender of the decision-maker in the household were,<br />
however, not significant predictors (p 1.00) of net profitability. Biophysical
aspects such as soil quality and availability of water might contribute to the financial<br />
value of these gardens, but such effects need to be investigated further.<br />
3.5. Economic alternatives to homegardens<br />
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
291<br />
Two possible alternatives were considered when comparing the economic rationale<br />
behind homegarden cultivation to other forms of investment. The first assigned<br />
alternative for a farmer was to sell the land, with the house and all associated crops<br />
and benefits, invest the capital in a bank at 6% compound interest rate (average<br />
prevailing rate at the time of study) and to live in a comparable neighborhood with a<br />
similar quality of life. The second option was to lease the land and all associated<br />
benefits to other farmers. Both alternatives and their profit values for all size classes<br />
of homegardens at the end of the investment year are considered in Table 5. The<br />
non-monetary benefits, however, were not quantified.<br />
Table 5. Comparison of homegarden finances to two alternate forms of economic<br />
investment<br />
India.<br />
1 for small, medium, large and commercial holdings in Thrissur district, Kerala,<br />
Variables<br />
Finances from gardens and two<br />
alternate land use options (Rupees)<br />
Garden 2<br />
Lease 3 Bank 4<br />
k<br />
a. Mean ‘small’ homegarden (n = 24)<br />
Land 0 1086 22012<br />
Labor 0 7250 7250<br />
Living expense 0 (20000) (20000)<br />
Rent 0 0 (15000)<br />
Transportation 0 (500) (500)<br />
Incidentals 0 (800) (800)<br />
Homegarden costs (7548) 0 0<br />
Benefits 65519 0 0<br />
Net income 57971 (12964) (7038)<br />
b. Mean ‘medium’ homegarden (n = 14)<br />
Land 0 2552 61329<br />
Labor 0 14914 14914<br />
Living expense 0 (22000) (22000)<br />
Rent 0 0 (15000)<br />
Transportation 0 (500) (500)<br />
Incidentals 0 (800) (800)<br />
Homegarden costs (12399) 0 0<br />
Benefits 174912 0 0<br />
Net income 162513 (5834) 37943<br />
Table 5 (contd.)
292<br />
S.MOHAN ET AL .<br />
Variables<br />
Finances from gardens and two<br />
alternate land use options (Rupees)<br />
Garden 2<br />
Lease 3 Bank 4<br />
k<br />
c. Mean ‘large’ homegarden (n = 10)<br />
Land 0 4240 101760<br />
Labor 0 11880 11880<br />
Living expense 0 (22000) (22000)<br />
Rent (15000)<br />
Transportation 0 (500) (500)<br />
Incidentals 0 (800) (800)<br />
Homegarden costs (12307) 0 0<br />
Benefits 237158 0 0<br />
Net income 224851 (7180) 75340<br />
d. Mean ‘commercial’ homegarden (n = 27)<br />
Land 0 8250 201370<br />
Labor 0 17862 17862<br />
Living expense 0 (24000) (24000)<br />
Rent (15000)<br />
Transportation 0 (500) (500)<br />
Incidentals 0 (800) (800)<br />
Homegarden costs (17302) 0 0<br />
Benefits 275524 0 0<br />
Net income 258222 812 178932<br />
1 2<br />
Estimated for a homegarden of mean size in each size category; maintained as<br />
homegardens; 3 lease option and the lease values were based on existing rent rate of Rupees 12<br />
350 per ha. 4 capital invested in a bank at 6% compound interest rate; parenthetical values are<br />
costs.<br />
Living costs were estimated based on a two-month survey of expenses incurred<br />
by four urban and rural households with no attached homegardens. All household<br />
expenses, not including meat, staple foods such as rice, potato, salt, and other goods<br />
not normally realized from the garden, were estimated to be an average minimum of<br />
Rupees 20 000 per year per household. A comparison of the data in Table 5 also<br />
indicates the financial effectiveness of maintaining homegardens as opposed to<br />
leasing or selling the land. Selling the garden becomes a reasonable yet not<br />
comparable alternative with increases in land area. Small farmers would be best<br />
served if they retained their homegardens. Leasing was not an economically viable<br />
option especially for the small, medium or large gardeners.<br />
4. DISCUSSION AND CONCLUSIONS<br />
All homegardens surveyed in this study generated profits at steady state, thus<br />
justifying the need to consider them by the policy makers as on par with other<br />
mainstream agricultural production systems. The positive financial value, regardless<br />
of the number of years in cultivation, implies the renewable nature of these gardens<br />
year after year. The profit generated per unit area was highest for the small gardens
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
293<br />
(Table 2) and was lowest in the commercial gardens, perhaps implying that the small<br />
farmers are particularly adept at adaptive management techniques. Holding size<br />
being a constraint, farmers intensify cultivation on available land in order to attain<br />
desired goals and objectives. Commercial farmers, however, may devote some part<br />
of their holding for intangible benefits such as aesthetics and ornamentation. Future<br />
studies could assess whether this difference in profit generation equals the<br />
opportunity cost incurred by those commercial farmers who do not intensify<br />
production. Coconut, arecanut and bananas were the three most economically<br />
important crops (Fig. 1). It was noted, however, that although market needs were<br />
extremely important in determining garden use, small gardeners used more than half<br />
their annual produce for household uses, e.g., vegetables, fruits and firewood.<br />
Allocation of garden space also was need-based; i.e., if the farmers possessed liquid<br />
cash at their disposal with which to buy subsistence products, they increased the<br />
acreage under commercial crops such as areca and spice trees. On an average, more<br />
than 75% of the household needs of an average family were met by their<br />
homegardens irrespective of the garden size. The sensitivity analyses (Table 3)<br />
reaffirmed the hypothesis that these systems are economically stable, not dependent<br />
on any one crop or factor, and that the farmers followed an age-old adaptive<br />
approach to farming. Harvests were staggered so as to retain food crops such as<br />
cassava, for times of the year when staple food crops such as rice were not readily<br />
available. No one crop formed a focal point in the garden. For example, the areca<br />
crop had been sustaining high returns during the 1990s, but suffered a crash in<br />
market prices during the past few years (2001 and 2002); many farmers would have<br />
sustained heavy losses had their gardens consisted of sole stands of areca palms<br />
alone. With the existing complexity and diversity of these gardens (Appendix I),<br />
however, the lagging arecanut prices did not substantially affect the overall profit<br />
from the gardens. After considering two potential alternatives to homegardening<br />
(Table 5), it was estimated that retaining the land under homegarden cultivation was<br />
more profitable than leasing or selling the land even without factoring in intangible<br />
benefits such as aesthetics, nutritional security, and improved quality of food.<br />
Plantation farming was not considered as an alternative because many of the gardens<br />
surveyed were deemed too small to be fit for plantation agricultural systems.<br />
The household labor associated with homegardening was an important<br />
component of the alternatives because it was assumed that if the land were no longer<br />
available to farmers, they would earn money by working as laborers in the nearby<br />
farms. This is another debatable point, however, because many of the farmers<br />
reported that they were not equipped to perform any skilled work, nor did they<br />
desire to perform farm labor outside their properties. Furthermore, many of the<br />
farmers were older, and cherished the relative freedom they enjoyed from working<br />
in their own fields, and in their ability to set their own timings.<br />
It needs to be acknowledged that the methodology used for the study had some<br />
constraints. <strong>Homegardens</strong> are so diverse in species richness and composition<br />
(Appendix I) that data collection becomes arduous and error-prone. Data analysis<br />
becomes further complicated because many homegarden species are retained to<br />
fulfill certain specific needs and functions, and these needs vary from farmer to<br />
farmer and from region to region. Intangible benefits of homegardens, such as
294<br />
S. MOHAN ET AL .<br />
aesthetics and ornamentation, nutritional security, food quality, and empowerment<br />
of women also need to be considered in order to obtain a more accurate assessment<br />
of the economic values as articulated also by Torquebiau and Penot (2006).<br />
Furthermore, some of the data presented here, especially the monetary values, are<br />
time-sensitive. Although these constraints set some limits to applicability of the<br />
findings to other regions, we believe that the methodology can be adapted in any<br />
geographic area to estimate the economic value of these multipurpose production<br />
systems.<br />
ENDNOTE<br />
1. The data were gathered during the first author’s field study, which involved<br />
interaction with farmers and discussion with various officials of the Kerala<br />
Agricultural University and local field extension personnel of the government<br />
agricultural and other departments. The authors thank the Kerala Agricultural<br />
University, Thrissur, India for extending support to this project.<br />
REFERENCES<br />
Alavalapati J.R.R., Shrestha R.K., Stainback G.A. and Matta J.R. 2004. Agroforestry<br />
development: An environmental economic perspective. Agroforest Syst 61/62: 299 – 310.<br />
Christanty L. 1990. <strong>Homegardens</strong> in tropical Asia with special reference to Indonesia. In:<br />
Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 9 – 20. United Nations<br />
University Press, Tokyo.<br />
Current D., Lutz E. and Scherr S. (eds). 1995. Costs, benefits, and farmer adoption of<br />
agroforestry. The World Bank environment paper 14: World Bank, Washington, DC.<br />
Drescher A.W. 1996. Management strategies in African homegardens and the need for new<br />
extension approaches. In: Heidhues F. and Fadani A. (eds), Food security and innovations<br />
– Successes and lessons learned. pp 231 – 245. Peter Lang, Frankfurt.<br />
Jose D. and Shanmugaratnam N. 1993. Traditional homegardens of Kerala: a sustainable<br />
human ecosystem. Agroforest Syst 24: 203 – 213.<br />
Karyono 1990. <strong>Homegardens</strong> in Java: their structure and function. In: Landauer K. and Brazil<br />
M. (eds), <strong>Tropical</strong> home gardens, pp 138 – 146. United Nations University Press, Tokyo.<br />
Mendez V.E., Lok R. and Somarriba E. 2001. Interdisciplinary analysis of homegardens in<br />
Nicaragua: micro-zonation, plant use and socioeconomic importance. Agroforest Syst 51:<br />
85 – 96.<br />
Mohan S. 2004. An Assessment of the ecological and socioeconomic benefits provided by the<br />
homegardens: A case study from Kerala, India. PhD Dissertation, University of Florida,<br />
Gainesville, FL.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Scherr S.J. 1992. Financial and economic analyses of agroforestry systems: An overview of<br />
the case studies. In: Sullivan G.M., Huke S.M., and Fox J.M. (eds), Financial and<br />
economic analyses of agroforestry systems. Proceedings of a workshop held in Honolulu,<br />
Hawaii, USA. July 1991, pp 3 – 12. Nitrogen Fixing Tree Association, Paia, Hawaii.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosys<br />
Environ 41: 189 – 207.<br />
Torquebiau E. and Penot E. 2006. Ecology versus economics in tropical multistrata<br />
agroforests. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 269 – 282. Springer Science, Dordrecht.
FINANCIAL F ANALYSIS OF HOMEGARDENS<br />
APPENDIX I<br />
Woody perennials encountered in the sampled homegardens of Thrissur district,<br />
Kerala, India 1 .<br />
295<br />
Scientific name Local/common name Family Uses 2<br />
Tree and shrub species of primary economic utility to farmers<br />
Ailanthus triphysa matti Simaroubaceae b<br />
Anacardium occidentale cashew Anacardiaceae d,b,a<br />
Areca catechu arecanut Palmaceae a, f<br />
Artocarpus heterophyllus jackfruit Moraceae d, b<br />
Artocarpus hirsutus aini Moraceae b,c<br />
Bridelia retusa kaini Euphorbiaceae b<br />
Bombax ceiba poola Bombacaceae b,f<br />
Borassus flabellifer palmyra palm Palmaceae f,e<br />
Calophyllum inophyllum punna Clusiaceae b,c<br />
Caryota urens fish-tail palm Palmaceae f<br />
Cocos nucifera coconut Palmaceae a, c, f<br />
Coffea arabica coffee Rubiaceae d<br />
Corypha umbraculifera talipot palm Palmaceae f<br />
Dalbergia latifolia rosewood Fabaceae b<br />
Delonix regia poomaram Caesalpiniaceae b<br />
Garcinia cambogia kodampuli Clusiaceae d, c<br />
Grewia tiliifolia chadachi Tiliaceae b<br />
Hevea brasiliensis rubber Euphorbiaceae c, f<br />
Mangifera indica mango Anacardiaceae d, b<br />
Manihot esculenta cassava Euphorbiaceae e<br />
Michelia champaca kaatu chembakam Magnoliaceae b,c,g<br />
Morus alba mulberry Moraceae c,f<br />
Myristica fragrans nutmeg/mace Myristicaceae a<br />
Palaquium ellipticum pali Sapotaceae b,g<br />
Piper longum thippili Piperaceae g,b<br />
Pterocarpus marsupium venga Fabaceae b<br />
Santalum album sandalwood Santalaceae f<br />
Saraca indica asoka tree Caesalpiniaceae b,c<br />
Swietenia macrophylla mahogany Meliaceae b<br />
Syzygium aromaticum clove Myrtaceae a<br />
Tamarindus indica tamarind Caesalpiniaceae d<br />
Tectona grandis teak Verbenaceae b<br />
Terminalia tormentosa maruthy Combretaceae b<br />
Xylia xylocarpa irumullu Mimosoideae b,c<br />
Appendix 1 (contd.)
296<br />
S.MOHAN ET AL .<br />
Scientific name Local/common name Family Uses 2<br />
Trees and shrubs species of secondary economic utility to farmers, used<br />
mainly in the household<br />
Annona squamosa custard apple Annonaceae d,b,c<br />
Artocarpus altilis breadfruit Moraceae d<br />
Averrhoa bilimbi irimbampuli Oxalidaceae d,c<br />
Azadirachta indica neem Meliaceae g<br />
Cananga odorata ylang ylang Annonaceae f,g<br />
Carica papaya papaya Caricaceae d<br />
Casuarina equisetifolia kattaadi Casuarinaceae f,h<br />
Cinnamomum camphora camphor Lauraceae f,g<br />
Cinnamomum zeylanicum cinnamon Lauraceae e,c<br />
Citrus limon cherunarakam Rutaceae d,c<br />
Emblica officinalis Indian gooseberry Euphorbiaceae d,g<br />
Flacourtia inermis louvi-louvi Flacourtiaceae b,c,d<br />
Manilkara zapota sapota (sapodilla) Sapotaceae d,b,c<br />
Murraya koenigii curry leaf tree Rutaceae e,c<br />
Pimenta dioica allspice Myrtaceae e,g<br />
Pouteria campechiana eggfruit Sapotaceae d<br />
Psidium guajava guava Myrtaceae b,c,d<br />
Punica granatum pomegranate Punicaceae d<br />
Syzygium jambolana<br />
rose apple Myrtaceae d,b<br />
Terminalia catappa Indian almond Combretaceae e,c<br />
Theobroma cacao cacao Sterculiaceae f,a<br />
1<br />
In addition, 17 herbaceous species were identified under two categories each (having primary<br />
or secondary economic utility to the farmers); for details see Mohan (2004).<br />
2<br />
Uses: a = nuts, b = timber, c = fuelwood, d = fruits, e = leaves, bark and other parts of plant<br />
used as food, f = leaves bark and other parts of plant used for other purposes, g = ornamental<br />
or medicinal purpose, h = shade; Local names appearing in italics are vernacular names<br />
(Malayalam).
SECTION 4<br />
FUTURE OF HOMEGARDENS
CHAPTER 17<br />
THE ROLE OF HOMEGARDENS<br />
IN AGROFORESTRY DEVELOPMENT:<br />
LESSONS FROM TOMÉ-AÇU, A JAPANESE-<br />
BRAZILIAN SETTLEMENT IN THE<br />
AMAZON<br />
M. YAMADA* AND H.M.L. OSAQUI<br />
Division of International Environmental and Agricultural Science, Graduate School<br />
of Agriculture, Tokyo University of Agriculture and Technology, 3-5-8 Saiwaich, <br />
Fuch-shi, TT ky 183-8509, Japan; *E-mail: <br />
Keywords: Adaptive research, Farm <strong>coop</strong>eratives, Farmer innovation, Fruit trees,<br />
Institutional support.<br />
Abstract. Agroforestry systems developed by the Japanese immigrants and their descendants<br />
in the Eastern Amazon region have been the focus of attention as a model for sustainable rural<br />
development in the humid tropics. This paper looks at the role of homegardens in agroforestry<br />
development at the Tomé-Açu Nikkei settlement in Pará, Brazil during the past seven decades.<br />
Potential crop species – native as well as exotic – were gathered and nurtured by the farm<br />
families in these homegardens of size 1 to 3 ha. Although the Tomé-Açu Multipurpose<br />
Agricultural Cooperative (CAMTA) had experimental nurseries and the Japanese public<br />
agencies established local agricultural research stations for supporting emigrant farmers in the<br />
Amazon, the homegardens functioned as individual validation fields where the farmers<br />
‘experimented’ with new crops. <strong>Homegardens</strong> were also used for improvement and<br />
propagation of nursery stock making them on-farm laboratories for adaptive research and<br />
extension. The immigrants with the traditional tokun (master farmer) education of East Asia<br />
analyzed the local environment and ‘experimented’ with various plant associations and<br />
management techniques, which led to the evolution of the exceptionally successful and<br />
popular multistrata agroforestry systems in the Eastern Amazon region.<br />
1. INTRODUCTION<br />
Since the early 1980s, various authors of Amazonian studies have discussed<br />
agroforestry systems developed by the Japanese immigrants and their descendents<br />
299<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 299–316.<br />
© 2006 Springer. Printed in the Netherlands.
300 M. YYAMADA AND H.M.L. OSAQUI<br />
(the Nikkei farmers) as an economically viable and ecologically sustainable rural<br />
development option for the region (Jordan, 1986; Gradwohl and Greenberg, 1988;<br />
Uhl and Subler, 1988; Uhl et al., 1989; 1990; Anderson, 1990; Barrow, 1990; Subler<br />
and Uhl, 1990; Serrão and Homma, 1993; Fearnside, 1995; Serrão, 1995; Homma,<br />
1998). While there are more than thirty rural Japanese-Brazilian (Nikkei)<br />
communities in the Amazon, most authors have focused on the Tomé-Açu<br />
settlement, which was the center of commercial rice (Oryza sativa) and vegetable<br />
production in the Amazon, and the location where first commercial black pepper<br />
(Piper nigrum) production was started in the Americas. The Tomé-Açu settlement<br />
was founded in 1929 ca. 120 km south of Belém, the capital of Pará (2°31’ S and<br />
48°22’ W). At the end of 2002, there were 214 Nikkei farms in Tomé-Açu, covering<br />
77 500 ha, with 7200 ha of agroforestry fields 1 . By the end of 1996, the Nikkei<br />
farmers in Tomé-Açu planted 6500 ha of agroforestry fields, with three perennial<br />
vine species, four shade trees, 33 fruit trees, 68 multipurpose tall trees, and<br />
numerous vegetable-, herb-, grain-, tuber- and green manure plants, forming a<br />
spatial mosaic of different ages and species combinations (Yamada, 1999). The<br />
main farm fields (6100 ha) excluding the homegardens around the housing/barnyard<br />
areas were occupied by ~70 crop species (90% arboreal) and some leguminous<br />
shade trees (“eritrina” = Erythrina spp. and “palheteira” = Clitoria racemosa). Sixty<br />
percent of the area involved polycultures, with approximately 300 different crop<br />
combinations, while the remainder was temporary monocultures based on sequential<br />
intercropping or “successional” agroforestry (Subler, 1993; Tanaka, 1997). For<br />
example, rubber (Hevea brasiliensis) and cupuaçu (Theobroma grandiflorum) fields<br />
were planted with annual crops (grains and vegetables) and perennial vines, but<br />
seedlings of shade tolerant arboreal and herbal species were subsequently<br />
introduced.<br />
2. HOMEGARDENS IN TOMÉ-AÇU AS LABORATORIES FOR SPECIES<br />
INTRODUCTION, SCREENING AND BREEDING<br />
2.1. Data collection<br />
The first author lived in Tomé-Açu from January 1995 to January 1997 conducting<br />
field work on farm histories and crop inventories. He visited all 214 Nikkei farms<br />
and interviewed farm owners on land use (intact and explored primary forests,<br />
secondary forest, fallow, pasture, and area under agroforestry) and crop species<br />
(year planted, number, area, and nature of cropping, i.e., with or without intercrops).<br />
For the inventory of homegardens, the authors first acquired reference information<br />
from the Tomé-Açu Multipurpose Agricultural Cooperative (CAMTA), which listed<br />
about 30 candidates that typified the history and dynamics of plant introduction,<br />
screening and dissemination in the Nikkei settlement. Among them, a dozen gardens<br />
were randomly chosen. With assistance from the owners of these gardens, details on<br />
each species (year, route of introduction, from where it came, etc.) were gathered.<br />
Additional information was gathered by revisiting the site occasionally from May<br />
2002 to March 2005, and reviewing published and unpublished documents.
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
2.2. Extent and diversity of components<br />
301<br />
<strong>Homegardens</strong> are maintained in areas of 1 to 3 ha around the house of almost every<br />
Nikkei farmer. The gardens contain a mixture of fruit trees, vegetables, medicinal<br />
plants, ornamental plants, and tall trees for shade, timber, nuts, fruits, and resin. In<br />
many farms, the housing areas were distinctively noticeable from a distance by the<br />
presence of 35 to 40 m tall trees surrounding the house. <strong>Homegardens</strong> often had a<br />
plant nursery for the main fields and sometimes a henhouse, a pigsty, a tortoise pen,<br />
or a small pond for pisciculture. The nursery was often close to the farmhouse to<br />
facilitate irrigation and close monitoring. The locations of animal sheds and fishponds<br />
depended on the species, source of water, and security against possible attacks<br />
by predators and thieves (guard dogs were also used for protection especially at<br />
night). Each homegarden had a unique appearance due to its history, species<br />
diversity, and physical arrangements. The owner’s family, labor families, and<br />
domestic animals living on-farm consumed the homegarden produce. The native<br />
and/or traditional homegarden fruits such as açai (Euterpe oleracea), mango<br />
(Mangifera indica), jackfruit ( (Artocarpus heterophyllus),<br />
and guava (Psidium<br />
guajava), were often harvested freely for on-farm consumption even without<br />
the permission of the owner. In farms where the young successors took over the<br />
management of main fields, the retired but still active parents took care of the<br />
homegardens, which not only increased the product shipment off-farm but also<br />
provided engagement and recreational avenues to the senior citizens. Homegarden<br />
surpluses were also given to the neighbors, friends and relatives, and sold in<br />
unprocessed or home-processed form at the local markets. Each farm also had<br />
modest facilities for cleaning and packaging vegetables, extracting and freezing fruit<br />
juice, making jam and bonbons from fruit pulp, and baking cookies with fruits and<br />
nuts. The homegarden facilities were also used for processing the off-season main<br />
field produce, especially when there was not enough to ship to the CAMTA’s juice<br />
factory. Marketing the homegarden produce gave income to the garden’s caretakers,<br />
mostly elders, housewives and children, and provided them with an opportunity for<br />
socialization at the marketplaces.<br />
2.3. Source of homegarden components<br />
Most species found in the homegardens had been acquired by the male farmers,<br />
casually or purposefully, during their travels. In addition, the CAMTA used to send<br />
exploratory missions for collection of seeds and vegetative materials, when the<br />
members became interested in certain species/cultivars available at farther locations,<br />
such as the Caribbean and tropical Asia. Such materials were initially brought to the<br />
<strong>coop</strong>erative’s experimental nursery for multiplication and eventual distribution<br />
among the associated farmers. The local research stations established by the<br />
Japanese public agencies for emigrant support, such as the Amazon <strong>Tropical</strong><br />
Agriculture Experiment Institute (INATAM; eventually incorporated by the<br />
Brazilian Agricultural Research Corporation for Eastern Amazon – EMBRAPA<br />
Amazônia Oriental) also introduced potential crop species for the agricultural<br />
<strong>coop</strong>eratives and interested individuals. In addition, some housewives made
Table 1. Inventory of tree species and other useful plants found in two Tomé-Açu Japanese-Brazilian homegardens, with information on their origin, route<br />
of introduction, and date.<br />
a. Sakaguchi Farm (Lot # Açaizal 185; given from father-in-law in 1959; homegarden 2 ha)<br />
Species (Latin name) English name Local name Year and route of introduction Origin<br />
Albizia spp. ? volunteer<br />
Annona muricata L. soursop graviola 1991 friend Tomé-Açu<br />
Araucaria angustifolia (Bertol.) Kuntze. Paraná pine pinheiro do 1988 market Belém<br />
Paraná<br />
Artocarpus incisa L. breadfruit fruta-pão 1992 friend Tomé-Açu<br />
Artocarpus odoratissima Blanco. marang marang 1987 friend Mindanao, Philippines<br />
Averrhoa bilimbi L. bilimbi bilimbi ? ?<br />
Averrhoa carambola L. starfruit carambola 1974 CAMTA survey Kuala Lumpur,<br />
Malaysia<br />
Azadirachta indica A. Juss. neem nim 1984 friend Thailand<br />
Bactris gasipaes H.B.K. peach palm pupunha 1970 IPEAN Belém<br />
Bertholletia excelsa H.B.K. brazilnut castanha do 1932 JPCB Tomé-Açu<br />
Pará<br />
Bischofia javanica Blume. Javanese bishopwood 1995 visitor Ogasawara, Japan<br />
Camellia sinensis (L.) Kuntze. (5 vars.) tea chá 1981 excursion Registro, São Paulo<br />
Carapa guianensis Aubl. andiroba andiroba 1960 friend (from JPCB) Tomé-Açu<br />
Carica papaya L. papaya mamão 1979 CAMTA survey Kao-hsiung, Taiwan<br />
Casuarina equisetifolia L. Australian pine casuarina 1985 friend Philippines<br />
302 M. YAMADA AND<br />
Y H.M.L. OSAQUI
Cedrela odorata L. cedro cedro 1984 farm laborer Bujarú, Pará<br />
Ceiba pentandra L. kapok sumaúma ? volunteer<br />
Cereus jamacaru D.C. mandacaru mandacaru 1976 INATAM excursion Pernambuco<br />
Cinnamomum zeylanicum Blume. cinnamon canela 1978 Takasago Farm Daini Tomé-Açu<br />
Citrus latifolia Tan. Tahiti lime limão Tahiti ? ?<br />
Citrus aurantium L. bitter orange laranja amarga 1977 INATAM Daini Tomé-Açu<br />
Citrus deliciosa Ten. (mexerica) common mandarin mexerica ? ?<br />
Citrus deliciosa Ten. (2 vars.) common mandarin mandarina 1980 Dienberger nursery Limeira, São Paulo<br />
comum<br />
Citrus junos Sieb. ex Tan. yuzu ? relative Japan<br />
Citrus noblis Makino mandarin mandarina 1977 friend Acará, Pará<br />
Citrus noblis Makino mandarin mandarina 1980 Dienberger nursery Limeira, São Paulo<br />
Citrus sinensis (L.) Osbeck. orange laranja 1980 Dienberger nursery Limeira, São Paulo<br />
Citrus unshiu Marc. Satsuma mandarin Satsuma 1986 friend Tomé-Açu (from<br />
unshiu<br />
Japan)<br />
Cocos nucifera L. (praia) coconut coqueiro 1962 relative Tomé-Açu<br />
Cocos nucifera L. (anão) coconut coqueiro 1970 friend Tomé-Açu<br />
Coumarouna odorata (Aubl.) Willd. tonka bean cumarú 1965 friend Tomé-Açu (from<br />
Belém)<br />
Crescentia cujete L. calabash tree cuieira 1970 excursion Igarapé-Açu, Pará<br />
Cryptomeria japonica D.Don. Japanese cedar cedro japonês 1980 Japanese consul Belém (from Yaku,<br />
Japan)<br />
Table 1 (cont.)<br />
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
303
Species (Latin name) English name Local name Year and route of introduction Origin<br />
Cryptomeria japonica D.Don. (obi) Japanese cedar cedro japonês 1981 friend Tomé-Açu (from<br />
Japan)<br />
Cycas revoluta Thunb. sago palm cica, palmeira- ? friend Tomé-Açu<br />
sagu<br />
Dalbergia spruceana Benth. Amazon rosewood jacarandá do 1974 JICA Monte Alegre, Pará<br />
Pará<br />
Delonix regia (Bojer ex Hook.) Raf. flamboyant tree flamboyant m 1966 friend<br />
Belém (from São<br />
Paulo)<br />
Durio zibethinus L. durian durião 1979 CAMTA survey Johor Bharu, Malaysia<br />
Endopleura uxi (Huber) Cuatrec. uxi uxi 1982 friend Daini Tomé-Açu<br />
Euphoria longan (Lour.) Steud. longan longan 1979 CAMTA survey P’ing-tung, Taiwan<br />
Ficus elastica Roxb. ex Hornem. Indian rubber tree figueira-da- 1978 friend Atibaia, São Paulo<br />
borracha<br />
Garcinia dulcis (Roxb.) Kurz. rata mangostão 1960 friend Tomé-Açu (from São<br />
falso<br />
Paulo)<br />
Garcinia mangostana L. mangosteen mangostão 1961 IPEAN Belém<br />
Gliricidia sepium (Jacq.) Steud. mother of cacao gliricidia 1985 friend Philippines<br />
Gmelina arborea Roxb. gmelina gmelina 1970 friend Belém (from Jari, Pará)<br />
Hevea brasiliensis (Willd.) Muell.-Arg. rubber tree seringueira 1959 Goodyear nursery Igarapé-Açu, Pará<br />
Illicium spp. 1973 CAMTA excursion Uruçuca, Bahia<br />
Inga edulis Mart. (long variety) inga inga 1984 excursion Abaetetuba, Pará<br />
Lansium domesticum Corr. langsat langsat 1979 CAMTA survey Kuala Lumpur,<br />
Malaysia<br />
304 M. YAMADA AND<br />
Y H.M.L. OSAQUI
Leucaena leucocephala (Lam.) de Wit. leucaena leucaena 1976 INATAM excursion Juazeiro, Bahia<br />
Licaria puchury-major r (Mart.) Kosterm. pichury bean puxuri 1982 relative (from JPCB) Tomé-Açu<br />
Litchi chinensis Sonn. lychee lichia 1979 CAMTA survey P’ing-tung, Taiwan<br />
Macadamia ternifolia F. Muell. macadamia nut macadâmia 1978 Dienberger nursery Limeira, São Paulo<br />
Moringa oleifera Lam. horseradish tree moringa 1976 friend Tomé-Açu<br />
Murraya paniculata (L.) Jack. orange jasmine murta 1981 JICA survey Dominican Republic<br />
Myrciaria dubia (H.B.K.) Mc Vaugh. camu camu camu mm camu 1987 friend<br />
Belém<br />
Myrciaria cauliflora Berg. jaboticaba jaboticaba 1973 CAMTA excursion Uruçuca, Bahia<br />
Myristica fragans Houtt. nutmeg noz-moscada 1976 INATAM excursion Uruçuca, Bahia<br />
Nephelium lappaceum L. rambutan rambutan 1979 CAMTA survey Johor Bharu, Malaysia<br />
Ocotea cymbarum H.B.K. Brazilian sassafras louro- 1988 Takasago Farm Daini Tomé-Açu<br />
inhamuí<br />
Pachira aquatica Aubl. Guiana chestnut munguba 1974 JICA Belém<br />
Paulownia spp. 1993 friend Belém (from Taiwan)<br />
Persea americana Mill. avocado abacate 1977 friend Tomé-Açu (from São<br />
Paulo)<br />
Pimenta dioica (L.) Merr. allspice pimenta da 1976 INATAM excursion Uruçuca, Bahia<br />
Jamaica<br />
Platonia insignis Mart. bacurí bacurí ? volunteer<br />
Pouteria caimito Radlk. abiu abiu 1976 friend Belém<br />
Pouteria campechiana Baehni. eggfruit canistel 1976 friend Tomé-Açu<br />
Prosopis juliflora (Sw.) DC. mesquite algaroba 1982 CAMTA excursion Ceará<br />
Table 1 (cont.)<br />
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
305
Species (Latin name) English name Local name Year and route of introduction Origin<br />
Quassia amara L. amargo pau amargo 1976 friend Tomé-Açu<br />
Rollinia deliciosa Saff. biriba biriba 1995 volunteer<br />
Spondias tuberosa Arruda. umbú umbú 1976 INATAM excursion Juazeiro, Bahia<br />
Tamarindus indica L. tamarind tamarindo 1977 friend Pakistan<br />
Theobroma subincanum Mart. cupuí cupuí 1990 volunteer<br />
Veronica condensata Baker boldo verde boldo verde 1988 friend Daini Tomé-Açu<br />
Virola surinamensis (Rol.) Warb. virola ucuúba 1995 volunteer<br />
(herbs; a portion)<br />
Capsicum spp. 1988 friend Chili<br />
Curcuma zedoaria (Christm.) Roscoe. zedoary zedoária 1980 friend Campinas, São Paulo<br />
Elettaria cardamomum (L.) Maton. cardamom cardamom 1971 CAMTA Tomé-Açu (fr.<br />
Guatemala)<br />
Manihot aypi Spruce sweet cassava macaxeira ? ? Rio de Janeiro<br />
Pogostemon patchouli Pell. patchouli patchouli 1960s Takasago Farm Daini Tomé-Açu<br />
Spilanthes acmella Murr. jambu, toothache jambu 1959 relative Tomé-Açu<br />
plant<br />
Vanilla planifolia Andr. vanilla baunilha 1960s IPEAN Belém<br />
Vanilla planifolia Andr. vanilla baunilha 1969 JICA Mexico<br />
Vanilla planifolia Andr. vanilla baunilha 1970s Takasago Co. Madagascar<br />
Zingiber officinale Roscoe. ginger gengibre 1971 CAMTA Tomé-Açu (from<br />
Trinidad)<br />
b. Maki Farm (Lot # Cuxiu 2-241; purchased in 1969; homegarden 1 ha)<br />
Achras sapota L. sapodilla sapoti ? CAMTA nursery Tomé-Açu<br />
306 M. YAMADA AND<br />
Y H.M.L. OSAQUI
Aleurites moluccana (L.) Willd. candlenut<br />
1975 San Stepano Farm Daini T-Açu (from<br />
noz da India<br />
Hawaii)<br />
Aniba canelilla (Kunth.) Mez. casca preciosa casca 1980 farm laborer<br />
preciosa<br />
Aniba rosaeodora Ducke. rose wood<br />
1976 INATAM Daini T-Açu (fr.<br />
pau-rosa<br />
Manaus)<br />
Annona muricata L. soursop graviola 1977 friend Daini Tomé-Açu<br />
Annona squamosa L. sugar apple fruta-do-conde 1990 market Belém<br />
Artocarpus heterophyllus Lam. jackfruit jaca 1960s former lot owner<br />
Averrhoa bilimbi L. bilimbi bilimbi 1977 relative Daini Tomé-Açu<br />
Averrhoa carambola L. starfruit carambola 1960s former lot owner<br />
Averrhoa carambola L. starfruit carambola 1990 friend Tomé-Açu<br />
Azadirachta indica A. Juss. neem nim 1987 COPAMASA Tomé-Açu<br />
Caryocar villosum (Aubl.) Pers. piquiá piquiá 1981 forest Daini Tomé-Açu<br />
Cedrela odorata L. cedro cedro 1976 forest Daini Tomé-Açu<br />
Citrus aurantifolia (Christm.) Swingle. lime limão 1960s former lot owner<br />
Galego<br />
Citrus latifolia Tan. Tahiti lime limão Tahiti 1982 friend Daini Tomé-Açu<br />
Citrus deliciosa Ten. (murcote) common mandarin murcote 1970s INATAM Daini Tomé-Açu<br />
Citrus deliciosa Ten. (tangerin) common mandarin tangerina 1980s relative Daini Tomé-Açu<br />
Citrus grandis (L.) Osbeck pomelo toranja 1975 friend Daini Tomé-Açu<br />
Citrus sinensis (L.) Osbeck. orange laranja ? volunteer<br />
Citrus spp. (limãozinho) 1960s former lot owner<br />
Table 1 (cont.)<br />
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
307
Species (Latin name) English name Local name Year and route of introduction Origin<br />
Cordia goeldiana Huber. freijó freijó 1974 forest Daini Tomé-Açu<br />
Dalbergia nigra (Vell.Conc.) Benth. Brazilian rosewood jacarandá da 1976 JAMIC Bahia<br />
Bahia<br />
Dendrocalamus giganteus Wall. ex giant bamboo bambu- 1976 friend Tomé-Açu<br />
Munro.<br />
gigante<br />
Diospyros kaki Thunb. Japanese persimmon caqui 1976 friend Tomé-Açu<br />
Endopleura uxi (Huber) Cuatrec. uxi uxi 1981 forest Daini Tomé-Açu<br />
Eugenia cumini (L.) Druce. Java plum, jambolan jamborão 1992 elementary school Daini Tomé-Açu<br />
Eugenia stipitata McVaugh. araçá-boi araçá-boi 1989 friend Daini T-Açu (fr.<br />
Manaus)<br />
Fortunella japonica (Thunb.) Swingle. round kumquat kumquat 1970 friend Tomé-Açu<br />
Garcinia mangostana L. mangostin mangostão 1990 relative Daini Tomé-Açu<br />
Macadamia ternifolia F. Muell. macadamia nut macadâmia 1985 CAMTA nursery Tomé-Açu<br />
Malpighia glabra L. acerola, Barbados<br />
1980s friend (fr. INATAM) Daini Tomé-Açu<br />
acerola<br />
cherry<br />
Mammea americana L. mammey apple abrico do 1980s friend Tomé-Açu<br />
Pará<br />
Mangifera indica L. (haden) mango manga 1978 friend Tomé-Açu (from São<br />
Paulo)<br />
Mangifera indica L. (haden) mango manga 1978 friend Tomé-Açu (from<br />
Mimosa caesalpiniaefolia Benth. sabiá sabiá 1990 CAMTA survey Ceará<br />
Manaus)<br />
308 M. YAMADA AND<br />
Y H.M.L. OSAQUI
Morus bombycis Koidz. mulberry amora 1978 ? Tomé-Açu<br />
Myrciaria cauliflora Berg. jaboticaba jaboticaba 1976 friend Tomé-Açu<br />
Pachira aquatica Aubl. Guiana chestnut munguba 1970s friend Tomé-Açu<br />
Persea americana Mill. avocado abacate 1974 INATAM Daini Tomé-Açu<br />
Pilocarpus microphyllus Stapf. jaborandi jaborandi 1980s friend (fr. INATAM) Daini Tomé-Açu<br />
Platymiscium ulei Harms. macacauba macacauba 1976 Museu Goeldi Belém<br />
Prosopis juliflora (Sw.) DC. mesquite algaroba 1982 CAMTA survey Ceará<br />
Prunus mume Sieb. et Zucc. Japanese apricot umezeiro 1990 friend Daini Tomé-Açu<br />
Psidium guajava L. guava goiabeira ? volunteer<br />
Rauwolfia serpentina Benth. Indian snakeroot 1980s friend (fr. INATAM) Daini Tomé-Açu<br />
Rheedia macrophylla Planch. et Triana. biribá biribá 1988 CAMTA nursery Tomé-Açu<br />
Schizolobium amazonicum Hub. ex Ducke. paricá paricá 1980 friend Tomé-Açu<br />
Simarouba amara Aubl. marupá marupá 1977 IBDF Santa Isabel do Pará<br />
Spondias tuberosa Arruda. umbú umbú 1976 INATAM excursion Juazeiro, Bahia<br />
Stenocalyx pitanga O. Berg. pitanga pitanga 1980 friend Tomé-Açu<br />
Swietenia macrophylla King. mahogany mogno 1976 INATAM Daini Tomé-Açu<br />
Theobroma speciosum Willd. ex Spreng. cacaui cacaui 1980s volunteer<br />
Veronica condensata Baker boldo verde boldo verde 1992 CAMTA nursery Tomé-Açu<br />
herbs; a portion<br />
Cephaelis ipecacuanha (Brot.) A. Rich. ipeca ipecacuanha 1992 friend Daini Tomé-Açu<br />
Curcuma zedoaria (Christm.) Roscoe. zedoary zedoária 1990 friend Tomé-Açu<br />
Table 1 (cont.)<br />
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
309
Species (Latin name) English name Local name Year and route of introduction Origin<br />
Egletodendron pariri pariri pariri ? farm laborer<br />
Hibiscus sabdariffa L. red sorrel hibiscus ? relative Daini Tomé-Açu<br />
Kaempferia spp. ? CAMTA nursery Tomé-Açu<br />
Luffa operculata (L.) Cogn. luffa luffa ? ?<br />
Spilanthes acmella Murr. jambu, toothache<br />
? volunteer<br />
plant<br />
jambu<br />
CAMTA = Tomé-Açu Multipurpose Agricultural Cooperative; COPAMASA = Pará Cassava Corporation; IBDF = Brazilian Forest Defense Institute<br />
(today’s IBAMA); INATAM = Amazon <strong>Tropical</strong> Agriculture Experiment Institute (today’s EMBRAPA Eastern Amazon Research Station in Tomé-<br />
Açu); IPEAN = Northern Agriculture and Stockbreeding Research Institute (today’s EMBRAPA Eastern Amazon); JAMIC = Japan Migration and<br />
Colonization Corporation (today’s JICA); JICA = Japan International Cooperation Agency; JPCB = Japanese Plantation Company mm of Brazil.<br />
310 M. YAMADA AND<br />
Y H.M.L. OSAQUI
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
311<br />
collections of local medicinal plants from the yards of neighboring Brazilians.<br />
Friends, relatives, neighbors, contract workers, and visitors also brought in plant<br />
species often as gifts, or in exchange. Table 1 lists the species present in two sample<br />
homegardens in Tomé-Açu, along with the year of introduction and from where they<br />
were obtained.<br />
2.4. Innovative approach of farmer-explorers<br />
The Tomé-Açu homegardens became well known in the region since the late 1970s,<br />
thanks mainly to the efforts of two leading farmers: Noboru Sakaguchi (1933 –) and<br />
Takur Maki (1947 –). Some details on these two farms are furnished hereunder.<br />
• Sakaguchi farm: Noboru Sakaguchi is a forest science graduate from the Tokyo<br />
University of Agriculture, who traveled extensively on CAMTA missions in<br />
search of alternatives to black pepper that had been seriously threatened by<br />
diseases. From such expeditions, he brought back several species to the<br />
CAMTA nursery and to his own homegarden. Moreover, after studying the<br />
species composition and structure of rural Brazilian homegardens and writing<br />
accounts on traditional farming systems in the Amazon, Sakaguchi reported to<br />
the CAMTA administrative board that native Theobroma species planted with<br />
native multipurpose tall trees for shading (such as rubber and andiroba =<br />
Carapa guianensis) would be most appropriate for sustainable production in the<br />
region. CAMTA thus introduced the Bahian hybrid cacao (Theobroma cacao)<br />
to Tomé-Açu in 1971.<br />
• Maki farm: Takur Maki was one of the pioneer farmers to plant freijó (Cordia<br />
goeldiana) and macacauba (Platymiscium ulei), two highly appreciated native<br />
timber trees, for shading cupuaçu and cacao. Maki loved to wander around the<br />
forests and collect seedlings of useful trees that he learned about from the rural<br />
Brazilians. Although he did not have frequent chances of travel as Sakaguchi<br />
did, he looked for interesting species in the homegardens of friends and<br />
relatives within the settlement. The species procured from distant sources by<br />
Sakaguchi and others thus spread among the farmers of Tomé-Açu. Maki also<br />
provided seeds and seedlings from his homegarden to other interested farmers.<br />
With support from a Japanese public agency for emigrant support, he even<br />
shipped freijó seeds to other Nikkei settlements in Amazon.<br />
Although the homegarden caretakers (elders, housewives, and children)<br />
evaluated the local performance of new plants, final decision regarding large-scale<br />
planting in the main fields was taken primarily by the male heads of households<br />
after considering the available market information. Moreover, those pioneering<br />
family heads had often received tokun (master farmer) education in Japan that<br />
emphasized diligent practices based on careful observation of nature and taking<br />
pride in the vocation of producing food. The following case studies of key<br />
agroforestry species introductions in Tomé-Açu further illustrate the innovative<br />
approach of these farmer-explorers.<br />
• Sait-Oshikiri farm [specialty crops: cacao, rubber, brazilnut (Bertholletia<br />
excelsa) and black pepper]: According to Aiko Oshikiri (1920 – 2000), who
312<br />
M. YAMADA Y AND H.M.L. OSAQUI<br />
wrote about the period in the 1930s when Tomé-Açu was called the “green hell<br />
of poverty and fatal endemics,” she and her mother took care of the homegarden<br />
plants collected by her father, Enji Sait (1891 – 1958). While acting as the<br />
president of the vegetable producers’ <strong>coop</strong>erative (the predecessor of CAMTA)<br />
for the daily survival of the impoverished Japanese immigrants in the interior<br />
settlements, Mr. Sait searched for seedlings of ‘permanent crop’ species<br />
including brazilnut, cacao, rubber, urucu (Bixa orellana), guaraná (Paullinia<br />
cupana), and black pepper (‘Singapura’ or Kuching variety). He later became<br />
known as the founder of black pepper culture in the Amazon and the Americas<br />
(Oshikiri, 1985). It was his son-in-law Tanio Oshikiri (1911 – 1987), Aiko<br />
Oshikiri’s husband and CAMTA president, who promoted rubber and brazilnut<br />
among the Tomé-Açu farmers during the mid-1960s as substitute crops for<br />
black pepper. Today more than forty 70-year-old brazilnut trees remain in the<br />
Sait-Oshikiri farm, with the largest ones attaining 200 cm diameter at breast<br />
height and a height of about 35 m.<br />
• Shimomaebara farm [specialty crop: passionfruit (Passiflora edulis)]: In the<br />
early 1970s, black pepper fields in Tomé-Açu were severely affected by fungal<br />
blight (Fusarium solani f. sp. piperis) causing great economic hardship to the<br />
Nikkei community, which had made this crop their principal source of income.<br />
Mitsuji Shimomaebara (1914 – 1994), an honors graduate from Matsuda Farmer<br />
School in Matsubase, Kumamoto, Japan, where he received the tokun<br />
education, however, developed a simple system of passionfruit culture through<br />
which the economic hardships of the Nikkei community could be partially<br />
mitigated. In this method, passionfruit, a common local homegarden vine grown<br />
on trellises, vigorously climbed on the abandoned black pepper stakes, taking<br />
advantage of the residual soil fertility and spreading horizontally on a single<br />
wire extended over the stakes. The fruit bearing vines hung from the wire like a<br />
curtain and produced excellent results. With growing demand from juice<br />
factories, passionfruit became a key crop in the black pepper plantations, which<br />
began succumbing to Fusarium five to six years after planting. In 1974, Mr.<br />
Shimomaebara was awarded the Marshal Rondon medal for interior<br />
development by the Brazilian government, as his method of growing passionfruit<br />
became popular nation-wide. In terms of importance to agroforestry, both<br />
black pepper and passionfruit provided temporary shade, wind protection, and<br />
residual soil fertility to the young trees planted between the rows of perennial<br />
vines. Consequently, native fruit trees previously screened in homegardens such<br />
as cacao and cupuaçu, and tall trees including rubber, brazilnut, freijó, and<br />
andiroba established very well in this system (Yamada, 1999).<br />
• Kusano and Yokokura farms (specialty crops: Theobroma spp. under shade<br />
trees): After the introduction of Bahian-hybrid cacao in 1971, it became the<br />
most popular tree crop in the region in the 1970s and early 1980s. However, it<br />
was susceptible to witch’s broom disease caused by the fungus Crinipellis<br />
perniciosa (Stahel) Singer. By the mid-1980s, Hisaharu Kusano (1927 – 2003),<br />
another honors graduate from Matsuda Farmer School, and his son Tsuneo<br />
Kusano (1948 –) developed disease resistant cacao cultivars. They along with
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
313<br />
families toiled for more than a decade in their homegardens and the adjoining<br />
cacao orchards, conducting individual selection, grafting, and cross-pollination<br />
of the tiny cacao flowers 2 . The cacao scions screened were disseminated to<br />
interested farmers along with information on grafting techniques. Their methods<br />
were also applied to cupuaçu, which had been established as a major field crop<br />
by Nobuyoshi Yokokura (1914 – 1997), a farming haiku poet who had learned<br />
tokun discipline in his youth at the Kitami Colonization Training Center,<br />
Hokkaido, Japan. In the early 1970s, Yokokura anticipated the potential fruit<br />
pulp market for this homegarden species, which has a growth habit similar to<br />
that of cacao. He planted the first cupuaçu field at Tomé-Açu with tree shading,<br />
and distributed seeds to his young followers. Due to misplaced worries of<br />
cupuaçu transmitting the witch’s broom disease to cacao, local agricultural<br />
extension authorities warned Yokokura to cut down his cupuaçu trees or lose<br />
institutional financing. However, he never gave up his orchard. In the 1990s,<br />
when cacao and the vine crops faced low prices, Tomé-Açu farmers were<br />
sustained by their cupuaçu pulp sales. The native and shade-tolerant<br />
Theobroma-based systems thus expanded to 3400 ha or 56% of the main field<br />
agroforests in Tomé-Açu and opened up new opportunities for planting cacao<br />
with various useful tall tree species screened in the homegardens (Yamada,<br />
1999). Again, farmers were initially warned by the extension agents to plant<br />
only leguminous shade trees (eritrina and palheteira) with cacao, but they<br />
pursued their own ideas and created productive multistrata/multispecies farms<br />
within three decades, which now serve as officially recommended models for<br />
family farms in the region.<br />
The Nikkei farmers of Tomé-Açu are perpetual innovators. In addition to the<br />
significant cases of local and regional agroforestry development history listed above,<br />
we identified during the farm visits various on-going studies involving promising<br />
crop species, such as acerola (Malpighia glabra), açai, araça-pera (Psidium<br />
acutangulum), avocado (Persea americana), lime and oranges (Citrus spp.), uxi<br />
(Endopleura uchi), bacuri (Platonia insignis), spice trees, and other tall tree species<br />
for timber and non-timber purposes. While each farm became specialized in certain<br />
species or cropping systems, successful results were shared quickly within the<br />
community and beyond, partially because of the easy access for curious visitors to<br />
the homegarden area near the farmhouse. However, it was essentially the<br />
multipurpose agricultural <strong>coop</strong>erative (CAMTA) that prompted the development and<br />
dissemination processes. CAMTA’s Technical Assistance and Extension Division<br />
(ATEA) had experienced agronomists, who regularly visited these farms. Besides,<br />
the <strong>coop</strong>erative received public supports from Japan, such as visiting experts of<br />
various specialties, training and excursion programs for farmers, introduction of new<br />
species and varieties, and financial support for the <strong>coop</strong>erative projects including<br />
construction of the experimental juice factory. Considering that institutional support<br />
is crucial in developing complex agroforestry systems (Follis and Nair, 1994), in<br />
Tomé-Açu the long-lasting and comprehensive collaboration between CAMTA, the<br />
<strong>coop</strong>erative representing immigrant farmers, and the Japanese public agencies for<br />
emigrant support led to the success of agroforestry development. In this scenario, the<br />
Nikkei homegardens functioned as an informal ‘institutions’ run by the networked
314<br />
farmers with tokun orientation, complementing the roles of the <strong>coop</strong>erative and<br />
public institutions and making their initiatives more effective. Tomé-Açu farmers<br />
thus realized the intensive and economically viable production systems that<br />
converted much less forested area to farmlands compared to other prevailing types<br />
of land development models in the Eastern Amazon, and generated rural<br />
employment (Yamada, 1999; Nair, 2001; Yamada and Gholz, 2002).<br />
2.5. Outreach and technology transfer<br />
M. YAMADA Y AND H.M.L. OSAQUI<br />
Since the mid-1990s CAMTA board members became active in transferring agroforestry<br />
techniques to non-Nikkei family farms in the neighborhood. Michinori<br />
Konagano (1958 –), who was in charge of the <strong>coop</strong>erative’s extension division, told<br />
the authors that raising production on numerous small family farms would make the<br />
rural societies peace-loving and the society at large would also be free from criminal<br />
activities. During the weekends, Konagano would, therefore, visit his neighbors,<br />
distribute seedlings, and teach agricultural techniques. Konagano was later<br />
appointed the secretary of agriculture of the municipality of Tomé-Açu.<br />
Since the early 2000s, the Tomé-Açu agroforestry model gained wider attention<br />
as a viable alternative to mass forest destruction in the Amazon and orders to<br />
CAMTA for its products increased from the US, Europe, and Japan. In 2004, the<br />
Japan International Cooperation Agency (JICA) launched a project in Tomé-Açu, in<br />
collaboration with the local municipal office, CAMTA, EMBRAPA Amazônia<br />
Oriental, and POEMA (Poverty and the Environment in Amazonia – a local NGO),<br />
to establish an agroforestry training center for the young owners of the small family<br />
farms. In 2005, SAMBAZON, a US-based customer of CAMTA facilitated organic<br />
certification of açai products, which in turn led to doubling the capacity of the<br />
<strong>coop</strong>erative’s fruit juice factory to 2400 Mg month –1 . It encouraged CAMTA to<br />
disseminate agroforestry among small family farmers of the region, teach them how<br />
to organize marketing <strong>coop</strong>eratives, and buy products from these <strong>coop</strong>eratives for<br />
processing at the CAMTA juice factory.<br />
3. CONCLUSIONS<br />
The individual, collective, and public efforts at Tomé-Açu over the past 75 years<br />
have led to the development of successful multistrata agroforestry systems that have<br />
attracted worldwide attention. The homegarden as the locus of individual experimentation<br />
with a variety of crops and their mixtures has been at the center of this<br />
historical process. Through this developmental process, immigrant farmers overcame<br />
difficulties in the unfamiliar climate, and established commercial crops such as<br />
vegetables, black pepper, passionfruit, fruit trees, and other products. This case<br />
implies that stimulating and supporting farmer initiatives in agroforestry<br />
homegardens is an effective approach to the development of sustainable rural<br />
development projects – perhaps more effective than the ‘conventional’ strategy of<br />
providing farmers with supposedly proven agroforestry modules for their main farm<br />
fields.
HOMEGARDENS IN AGROFORESTRY DEVELOPMENT: TOMÉ-AÇU T<br />
ENDNOTES<br />
315<br />
1. Nagasaki Y. 2003. Tomé-Açu ni okeru Nikkei Nka no Sakumotsu Uetsuke<br />
Jky ni kansuru Tsuiseki Chsa. ACTA, Tomé-Açu, Brazil,19p.<br />
2. The elder Kusano recounted his tokun philosophy to the authors that farm crops<br />
grow by listening to the owner’s footsteps (i.e., the owner needs frequent visits<br />
and careful observation of his field) and that a farm is established only after the<br />
pioneer’s wooden house has been returned to the soil (i.e., it takes long-term<br />
efforts to make good soil for sustainable production). Thus, even after the crash<br />
in international market of cacao, or the sudden drop in cacao bean prices in the<br />
international markets in the early 1980s, the Kusanos continued their on-farm<br />
research. However, the traditional tokun farmers sometimes overemphasized<br />
diligence over rationality and preferred clean culture rather than green mulch or<br />
grass cover methods in the tropical climate.<br />
REFERENCES<br />
Anderson A.B. 1990. Deforestation in Amazonia: Dynamics, causes, and alternatives. In:<br />
Anderson A.B. (ed.), Alternatives to deforestation: Steps toward sustainable use of the<br />
Amazon rain forest, pp 3 – 23. Columbia University Press, New York.<br />
Barrow C. 1990. Environmentally appropriate, sustainable small-farm strategies for<br />
Amazonia. In: Goodman D. and Hall A. (eds), The Future of Amazonia: Destruction or<br />
sustainable development? pp 360 – 382. Belhaven Press, London.<br />
Fearnside P.M. 1995. Agroforestry in Brazil’s Amazonian development policy: The role and<br />
limits of a potential use for degraded lands. In: Clüsener-Godt M. and Sachs I. (eds),<br />
Brazilian perspectives on sustainable development of the Amazon region, pp 125 – 148.<br />
UNESCO Man and the Biosphere Series 15, Parthenon Publishing Group, Carnforth.<br />
Follis M. and Nair P.K.R. 1994. Policy and institutional support for agroforestry: An analysis<br />
of two Ecuadorian case studies. Agroforest Syst 27: 223 – 240.<br />
Gradwohl J. and Greenberg R. 1988. Sustainable agriculture. In: Saving the tropical forests,<br />
pp 102 – 137. Earthscan Publications, London.<br />
Homma A.K.O. (ed.). 1998. Amazônia – Meio Ambiente e Desenvolvimento Agrícola.<br />
EMBRAPA, Brasília, 386p.<br />
Jordan C.F. (ed.). 1986. Permanent plots for agriculture and forestry. In: Amazonian<br />
rainforests: Ecosystem disturbance and recovery, pp 58–75. Springer-Verlag, New York.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science or, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Oshikiri A. 1985. Nyshoku tji no omoide. In: Hensh<br />
Iinkai (ed.), Tomé-Açu Kaitaku<br />
Gojusshnenshi, pp 74 – 75. Midori no Daichi, Tomé-Açu Cultural Association, Tomé-<br />
Açu.<br />
Serrão E.A.S. 1995. Possibilities for sustainable agriculture development in the Brazilian<br />
Amazon: An EMBRAPA proposal. In: Clüsener-Godt M. and Sachs I. (eds), Brazilian<br />
perspectives on sustainable development of the Amazon region, pp 259 – 285. UNESCO<br />
Man and the Biosphere Series 15, Parthenon Publishing Group, Carnforth.<br />
Serrão E.A.S. and Homma A.K.O. 1993. Sustainable agriculture in the humid tropics: Brazil.<br />
In: Committee on Sustainable Agriculture and the Environment in the Humid Tropics,<br />
Board on Agriculture and Board on Science and Technology for International<br />
Development of the National Research Council (ed.), Sustainable agriculture and the
316<br />
M. YAMADA Y AND H.M.L. OSAQUI<br />
environment in the humid tropics, pp 265 – 351. National Academy Press, Washington<br />
D.C.<br />
Subler S. 1993. Mechanisms of nutrient retention and recycling in a chronosequence of<br />
Amazonian agroforestry systems: Comparisons with natural forest ecosystems. PhD<br />
Dissertation. The Pennsylvania State University, State College, 203p.<br />
Subler S. and Uhl C. 1990. Japanese agroforestry in Amazonia: A case study in Tomé-Açu,<br />
Brazil. In: Anderson A.B. (ed.), Alternatives to deforestation: Steps toward sustainable<br />
use of the Amazon rain forest, pp 152 – 166. Columbia University Press, New York.<br />
Tanaka N. 1997. Nettai ngy ni okeru takakuka no tenkai to agroforestry ni kansuru ichi<br />
ksatsu: Amazon chiiki Tomé-Açu mura wo jirei to shite. Hokkaid Daigaku Nkei<br />
Rons 53: 151 – 163.<br />
Uhl C. and Subler S. 1988. Asian farmers: Stewards of Amazonia. Garden 12: 16 – 20, 31.<br />
Uhl C., Nepstad D., Buschbacher R., Clark K., Kauffman B. and Subler S. 1989. Disturbance<br />
and regeneration in Amazonia: Lessons for sustainable land use. The Ecologist 19:<br />
235 – 240.<br />
Uhl C., Nepstad D., Buschbacher R., Clark K., Kauffman B. and Subler S. 1990. Studies of<br />
ecosystem response to natural and anthropogenic disturbances provide guidelines for<br />
designing sustainable land use systems in Amazonia. In: Anderson A.B. (ed.),<br />
Alternatives to deforestation: Steps toward sustainable use of the Amazon rain forest, pp<br />
24 – 42. Columbia University Press, New York.<br />
Yamada M. 1999. Japanese immigrant agroforestry in the Brazilian Amazon: A case study of<br />
sustainable rural development in the tropics. PhD Dissertation, University of Florida,<br />
Gainesville, 821p.<br />
Yamada M. and Gholz H.L. 2002. An evaluation of agroforestry systems as a rural<br />
development option for the Brazilian Amazon. Agroforest Syst 55: 81–87.
CHAPTER 18<br />
URBAN HOMEGARDENS AND ALLOTMENT<br />
GARDENS FOR SUSTAINABLE LIVELI-<br />
HOODS: MANAGEMENT STRATEGIES<br />
AND INSTITUTIONAL ENVIRONMENTS<br />
A.W. DRESCHER 1 , R.J. HOLMER 2 , AND D.L. IAQUINTA 3<br />
1 Albert-Ludwigs-Universität, Freiburg, Germany; E-mail:<br />
. 2 Xavier University College of Agriculture,<br />
Cagayan de Oro, The Philippines. 3 Nebraska Wesleyan University, Lincoln,<br />
Nebraska, USA<br />
Keywords: Food security, Households, Species diversity, Sustainable development, Urban<br />
agriculture, Urban planning.<br />
Abstract. Diversity of food and income resources is one of the main buffers against<br />
vulnerability of the urban poor. Based on the authors’ field experience in the Philippines,<br />
Latin America, and southern Africa, and involvement with various other project evaluations,<br />
this chapter discusses the major differences between individual homegardens and allotment<br />
gardens and their respective roles in urban livelihood support programs. Major differences<br />
between these two systems of gardening are in their respective decision-making processes and<br />
impacts—in terms of both quantitative and qualitative outcomes. Current land use planning,<br />
multistory housing, and land use competition from different sectors limit both open space and<br />
space for gardening in the urban centers, necessitating lobbying and public advocacy to<br />
support such garden systems. While homegardens need public advocacy and extension<br />
services, allotment gardens additionally require significant political intervention to secure<br />
land, organize access, and support development. Implicit in this is the need for identifying the<br />
institutional barriers as well as gathering support for gardening projects in urban and<br />
periurban environments, prior to promoting the urban-gardening programs.<br />
1. INTRODUCTION<br />
Urban agriculture is the general term used to refer to a wide variety of food<br />
production practices in and around cities. Together with periurban agriculture, it<br />
317<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 317–338.<br />
© 2006 Springer. Printed in the Netherlands.
318 A.W. DRESCHER ET AL .<br />
represents a continually growing activity and consequently an emerging research<br />
area. Urban gardening is perhaps the most significant component of urban<br />
agriculture from the perspective of individual practitioners. It includes three types of<br />
practices: homegardens, allotment gardens and community gardens. While there is<br />
no universal agreement on the precise meaning of these terms, we adopt the<br />
following definitions. <strong>Homegardens</strong> are maintained – typically, but not always, near<br />
the homes – by individuals or households who have some access to land (either<br />
customary or legal), which they have arranged for themselves. Allotment gardens<br />
are separate parcels of land allocated to individuals or households for personal use.<br />
While contiguous, each household works on the parcels independently and the land<br />
is made available through either government action or private enterprises. The<br />
individual households are organized into self-governing associations. Community<br />
gardens are maintained by a group of individuals or households who produce<br />
agricultural goods collectively on a piece of land primarily for self-consumption.<br />
The extent and significance of urban gardening have been discussed elsewhere<br />
(Mougeot, 2005). Suffice to say that, it is widespread and that all three forms of<br />
gardening are growing throughout the world, particularly in response to income<br />
deprivation and the crises involving economic recession, natural disasters, and civil<br />
disorder (Jacobi et al., 2000). This is especially significant for the large segments of<br />
urban poor that continue to grow.<br />
In an extensive treatment of urbanization, urban and periurban agriculture, and<br />
urban poverty, Drescher and Iaquinta (2003) addressed an array of issues relevant to<br />
this chapter. In particular, they identified the characteristics of urban and rural poor,<br />
the very different socioeconomic conditions of urban poor in the developed and<br />
developing economies (see also UNCHS, 2001), and the gender-related aspects of<br />
urban gardens in different social and cultural systems. Another important<br />
consideration is that people practice urban gardening – whether home or allotment –<br />
for more varied reasons in the developed countries than in developing countries,<br />
principally because of the size of the economically stable population. Food<br />
production and income generation are important in both places, but the objectives of<br />
middle-income urban gardeners in developed countries (who grow flowers, create<br />
leisure environments, build kids’ playground, promote outdoor meeting places, etc.)<br />
are often different from those of their low-income counterparts.<br />
This chapter discusses the importance of urban gardens, highlights the<br />
constraints faced by urban gardeners, and addresses possible resolution of such<br />
constraints. Central to this discussion is the extension of the homegarden model to<br />
allotment gardens and the elaboration of the institutional contexts within which<br />
household livelihood strategies operate.<br />
2. DIVERSITY AND THE INVENTIVE SPIRIT OF URBAN GARDENING<br />
A common problem for urban gardens is the increased demographic pressure on<br />
available land, which we call spatial densification. This is caused or exacerbated by<br />
planning regulations intended to avoid urban sprawl and the desire of homeowners<br />
and users to create more living space. Because spatial densification means adding<br />
more people to the same area, it involves increased housing construction, which
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
319<br />
competes directly with the land available for gardening. This is a major problem for<br />
the poor, urban squatters and for the residents in spontaneous, periurban land<br />
occupations. Despite this apparent space constraint, homegardens are common in<br />
many urban environments.<br />
A probable solution to the constraints imposed by the overall urban situation is<br />
the development of alternative production systems adapted to lack of space, water,<br />
and other inputs. Soil-less cultures such as hydroponics, substrate cultures, and<br />
container gardens are just a few examples. Rooftop gardening is increasing in many<br />
densely populated cities. Even poultry farming and pig rearing take place on the<br />
rooftops or within houses, and are sometimes promoted by the local nongovernmental<br />
organizations (NGO) or international organizations such as the Food and<br />
Agriculture Organization (FAO, 1998; Drescher and Iaquinta, 2003). Since 1985, a<br />
<strong>coop</strong>erative of 100 poor women in Bogotá, Colombia, have used rooftops to grow<br />
hydroponic vegetables for city supermarkets. Unmarketable crops are either fed to<br />
livestock or used for home consumption.<br />
In Lima, Peru during the past two decades, the Ministry of Agriculture, FAO and<br />
United Nations International Children’s Emergency Fund (UNICEF) have promoted<br />
household and community kitchen gardens to avert widespread hunger. The Center<br />
for Education and Technology in Santiago, Chile, promotes 20 m 2 gardens, where<br />
plants are raised in containers stacked up in pyramids and walls are used for trailing<br />
vines (FAO, 1998). In Sri Lanka, “edible air-scapes” are promoted by the island<br />
nation’s department of agriculture as a strategy for rebuilding in the aftermath of the<br />
tsunami. Walls, bottles, bags, and fences are used for raising plants as part of this<br />
(Ranasinghe, 2005). The Cuban example of “organoponics” (Cruz and Medina,<br />
2003; Pinderhughes, 2004) is a well-established system incorporating both efficient<br />
water conservation and the use of compost and manure for fertilization in the urban<br />
context.<br />
In other parts of Latin America also, a particular form of organized homegardens<br />
exists (the so-called microgranjas), which involves the production of vegetables,<br />
fruits, other products, and small animals (e.g., chickens, pigs, guinea pigs, or rabbits)<br />
(Arias, 2000). The distinguishing characteristic of the microgranjas is that with<br />
governmental support, the homegardeners have been organized into groups. It thus,<br />
provides a platform for exchange of information and knowledge, despite the spatial<br />
dispersion of cultivated plots. Thus, while there are several forms of urban<br />
gardening, they can broadly be classified as homegardens, allotment gardens, and<br />
community gardens.<br />
3. URBAN HOMEGARDENS<br />
An urban homegarden, a multispecies production system on the area of land around<br />
the house to meet different physical, social, and economic needs and functions, is<br />
traditionally an important land use activity for individual households. Although its<br />
functions are similar throughout the world, focusing principally on subsistence or<br />
income generation, their structure and size vary considerably. For instance, in Papua<br />
New Guinea, Vasey (1985) reported that house plots generally range between 300 to<br />
400 m 2 but are often too small to meet the household demands. Consequently, many
320<br />
A.W. DRESCHER ET AL .<br />
households establish second gardens away from the house. Christanty (1990)<br />
reported that the size of homegardens in Bangladesh ranged between 30 and 700 m 2 ,<br />
with an average of 200 m 2 . Hoogerbrugge and Fresco (1993) also found wide<br />
variations in the size of homegardens even within a given country. For example,<br />
they reported size estimates ranging from 10 to 120 m 2 and 5000 to 20 000 m 2 in<br />
two separate studies in Zambia and ranges of 172 to 500 m 2 and 200 to 1700 m 2 in<br />
two Javanese studies. Drescher (1998) also observed large variations in this respect<br />
(17 to 865 m 2 in Lusaka, Zambia), but indicated that the majority of urban<br />
homegardens were less than 300 m 2 and that on average, women’s gardens were<br />
more than double the size that of men’s. Prosterman and Mitchell (2002) suggested<br />
that a great majority of homegarden plots in Java were less than 200 m 2 . Christanty<br />
(1990) earlier showed that on the less densely populated Indonesian islands,<br />
homegarden plots averaged 2500 m 2 , sometimes reaching a size of three hectares. In<br />
Lima, Peru, Hetterschijt (2004) also found that the size of urban organic<br />
homegardens varied between 25 and 900 m 2 , with an average of 110 m 2 (n = 109).<br />
In several studies, however, the failure to designate the study areas as rural, urban,<br />
or periurban, and the lack of universally accepted definitions for these classifications<br />
complicate the matter. For example, in thickly populated regions such as Java<br />
(Indonesia) and Kerala (India), the distinction between rural and urban settings is<br />
rather blurred. Iaquinta and Drescher (2000) have shown that this is even more<br />
pronounced in periurban areas.<br />
Nonetheless, four points emanate from the discussion above. First, the size of<br />
homegardens varies considerably across cultures and even within them. Second, the<br />
size of the majority of homegardens tends toward the low end of the range<br />
(positively skewed distribution pattern). Third, households sometimes make<br />
managerial decisions to locate some or all homegardens geographically distant from<br />
the house due to space or other constraints. Fourth, households make numerous<br />
management decisions, which, along with environmental constraints, determine both<br />
the physical structure and the outputs of the homegarden.<br />
Urban homegardens integrate a variety of physical, social, and economic<br />
functions. Typical homegardens include (1) physical areas for living, storage, and<br />
waste disposal, (2) social areas for meetings, children’s playgrounds, and display,<br />
and (3) economic areas for raising animals and for growing food, medicinal plants,<br />
and fruit trees. Overall, the homegarden is a place for people to live but also a place<br />
to produce a variety of foods and products for home consumption and income<br />
generation (Landon-Lane, 2004).<br />
<strong>Homegardens</strong> also play an important role in the conservation of indigenous<br />
crops, thus enhancing biodiversity in rural, periurban and urban environments.<br />
Drescher (1998) and Boncodin et al. (2000) found a variety of indigenous vegetables<br />
in the homegardens and in the local markets of Lusaka, Zambia. In particular,<br />
Amaranthus sp. grows semi-cultivated in the gardens. Other examples include<br />
Bidens pilosa, Brassica sp., Corchorus sp., Solanum macrocarpum, Hibiscus sp.,<br />
Cleome sp., Ipomoea batatas (sweet potato), and Cucumeropsis edulis (squash).<br />
Indigenous tree species providing multiple products such as firewood, food,<br />
fruits, and medicines also abound in the homegardens, yet they are often overlooked<br />
when talking about urban homegardens. For example, the leaves of the horseradish
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
321<br />
tree (Moringa oleifera) grown in the homegardens are the most frequently consumed<br />
vegetable among the households of Cagayan de Oro City, the Philippines (Agbayani<br />
et al., 2001).<br />
Urban homegardens compensate to a certain extent for the gardener’s restricted<br />
access to natural resources. While gathering wild vegetables and roots is still<br />
prevalent in periurban and rural areas, such options are limited in the urban context.<br />
For example, only 39% of households included in a study in Lusaka gathered wild<br />
fruits and vegetables, compared to 76% in periurban and 86% in the rural areas.<br />
Thus, in the urban context, homegarden produce provides an economical and<br />
nutritious substitute for wild vegetables and roots (Drescher, 1998). As seen in Fig.<br />
1, trees [e.g., peach (Prunus persica), papaya (Carica papaya), mango (Mangifera<br />
indica), and Morus alba] are an integral part of the urban homegardens in Lusaka.<br />
Figure 1. Map of an urban homegarden in Lusaka (Zambia) (Source: Drescher, 1998).<br />
While homegardening provides subsistence and supplementary household food<br />
supply, Boncodin et al. (2000) showed that it concurrently makes a significant<br />
contribution to the amount of nutrients and variety in the household food intake. In a<br />
study of the rural homegardens in the Philippines, they identified 33 different food<br />
crops, including green, leafy, and yellow vegetables; starchy roots and tubers; and<br />
legumes, beans, nuts, and spices. <strong>Homegardens</strong> thus provide year-round food<br />
supplements to households not only in terms of quantity but also in terms of food<br />
diversity and variation, and play an important role in providing Vitamin A and<br />
Vitamin C as well as supplying one-third or more of calcium and iron needs (Kumar<br />
and Nair 2004). This is consistent with the findings of a study on urban<br />
homegardens in the Philippines 1 .
322<br />
3.1. Community and allotment gardens<br />
A.W. DRESCHER ET AL .<br />
Community gardens are defined as gardens where people share the basic resources<br />
of land, water, and sunlight (MacNair, 2002). Allotment gardens, a special type of<br />
community garden, were first developed in Germany. Introduced as Schrebergärten<br />
in the mid-1800s, they flourished over a century and a half (Kasch, 2001). Allotment<br />
gardens are characterized by a concentration in one place of several small land<br />
parcels (usually 200 to 400 m 2 each). Individual families are organized into an<br />
association, which assigns the land parcels. In allotment gardens, the parcels are<br />
cultivated individually, as compared to community gardens where the entire area is<br />
tended collectively by a group of people (Holmer et al., 2003). Community gardens<br />
are often organized around a particular institution such as school, workplace, faith<br />
organization, hospital, etc. They may also be organized around social characteristics<br />
such as ethnicity, age, or religious orientation.<br />
In the Philippines, the production practices for vegetables in urban allotment<br />
gardens are similar to those in the rural areas; however, they differ particularly in the<br />
choice of cultivars and in the reduced application of agrochemicals due to the<br />
proximity to populated areas (Guanzon and Holmer, 2003). Although allotment<br />
gardeners are not excessively environment-oriented, nor are there many government<br />
restrictions on the use of agrochemicals, they are usually market-oriented. That is,<br />
about 70% of the produce is marketed directly within the garden itself—mostly to<br />
close neighbors; the consumers are generally well aware of the production practices<br />
and do not accept produce that has been heavily sprayed with chemicals. This<br />
situation differs greatly from the general system where vegetables are anonymously<br />
produced in far away locations and the customers mostly make assumptions<br />
regarding the production practices. This contrast is particularly true in the<br />
developing countries where government food safety controls are lax and quality<br />
labeling is either non-existent or unreliable.<br />
A preliminary study 2 in Cagayan de Oro, the Philippines indicated that the local<br />
people perceive the multiple benefits of allotment gardens. While 25% of the<br />
vegetables produced were consumed by the family or shared among friends, 75%<br />
were sold to neighbors or walk-in clients who come directly to the garden and who<br />
appreciate the freshness of the produce, the convenience of proximity, and the<br />
relatively lower price than the public markets. The gardening activities, a secondary<br />
occupation for all association members, thus augmented their incomes by about<br />
20%, while vegetable consumption also increased by about 75%. This is especially<br />
notable since the average vegetable consumption in Cagayan de Oro is only 36 kg<br />
per capita per year, about one-half of the minimum recommended intake suggested<br />
by FAO (Agbayani et al., 2001). In addition to these direct effects, the gardeners<br />
appreciate the strengthening of the community values brought about by allotment<br />
gardening.<br />
The gardens are also essential for the successful implementation of the city’s<br />
integrated solid waste management program. Segregated biodegradable wastes from<br />
neighboring households are delivered to the allotment gardens where they are<br />
converted into compost. The amount of residual waste delivered to the landfill site<br />
from these areas could theoretically be reduced by more than one-third, if all
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
323<br />
biodegradable wastes are channeled to a system of composting by allotment gardens<br />
in the city 3 . The city government of Cagayan de Oro is presently mainstreaming this<br />
concept into its overall city planning and development, using participatory GISbased<br />
approaches to identify suitable areas for further expansion of allotment<br />
gardens (Emmanuel Abejuela, pers. comm., August 2005). The advantages of such<br />
an approach are manifold. For example, mineral fertilizer application can be<br />
drastically reduced by using enriched compost, m thus reducing the danger of ground<br />
water pollution. The difference between urban and rural gardens is that the former<br />
uses biodegradable household wastes from many nearby households organized by<br />
the local government, and not only the bio-waste generated within the garden. Thus,<br />
urban gardens have a comparative advantage in their access to organic inputs<br />
generated by urban households, which is important in view of projected increase in<br />
demand for organically grown food.<br />
3.2. Urban homegardens, allotment gardens, and community gardens: a comparison<br />
The most important feature of allotment gardens is that they are institutionally<br />
administered and organized, and they serve as a community facility and a place of<br />
social interaction. In the German context, each gardener in an allotment garden<br />
needs to be a member of the respective Kleingartenverein (allotment garden<br />
association). In developing countries, however, gardeners are often not members of<br />
any associations, but are part of the community. The gardens are not necessarily<br />
near the homes, but rather located where sufficient space is available, and sometimes<br />
where favorable soil and water conditions exist. Obviously, transportation issues<br />
arise as the distance between homes and gardens increases. In other cases, the<br />
gardens are located in areas unsuitable for buildings or they are established as buffer<br />
zones along rail corridors and highways. They may even be located in protected<br />
areas necessary to balance the urban microclimate, as is the case in some German<br />
cities. Here allotment gardens are used on green belts to facilitate cold drainage, thus<br />
reducing urban heat island effects and the demand for air conditioning<br />
(Landeshauptstadt, 1998; Innenministerium, 2004).<br />
In Lusaka, Zambia, community gardens can be found on the edges of densely<br />
populated compounds that do not allow people to grow food within the housing area.<br />
A similar situation existed formerly in Harare, the capital of Zimbabwe. In Port<br />
Elizabeth, South Africa, some allotment gardens are situated on common areas and<br />
on the grounds outside churches, schools and hospitals (Jarlöv, 2000). Unlike<br />
Germany where allotment gardens are located on public lands owned by the city or<br />
railway, all allotment gardens of Cagayan de Oro, the Philippines, were established<br />
on private lands, due to the lack of publicly owned open spaces (Holmer et al.,<br />
2003). In Cagayan de Oro, the chairpersons of the barangay (city district) simply<br />
asked the local private landowners if poor residents of the barangay could use their<br />
vacant land for food production. To preclude residential occupation, however, the<br />
gardeners are only permitted to construct a small shed for tools and other garden<br />
implements, and not allowed to establish residential structures. Conditions for land<br />
use are then formalized into a memorandum of agreement jointly signed by all
324<br />
A.W. DRESCHER ET AL .<br />
stakeholders to legitimize access to the land for horticultural purposes (Vélez-<br />
Guerra, 2004).<br />
Urban homegardens have several characteristics that are similar to those of nonurban<br />
homegardens: (1) their location adjacent to homes (2) close association with<br />
joint family activities and (3) wide diversity of crop and livestock species used to<br />
meet family needs (Landon-Lane, 2004). Thus, homegardening is not simply a<br />
spatial integration but is characterized by a social and socioeconomic integration of<br />
the families involved. Importantly, overemphasis on the first characteristic<br />
oversimplifies the realities faced by residents in places such as the seasonal tropics<br />
of Africa, where homegardens are not necessarily near the homes; rather, they are<br />
located near water sources due to long dry seasons (Fig. 2). This could be a major<br />
reason why these production systems have often been overlooked in the past<br />
(Drescher, 1998).<br />
Figure 2. <strong>Homegardens</strong> and adjacent community gardens in Southern Zimbabwe. Some<br />
gardens are near the houses (upper left) some others are distant to the houses near the water<br />
source.<br />
The choice homegardeners make in locating their plots represents a clear<br />
environmentally informed management decision, and not a decision to engage in<br />
something fundamentally different from homegardening, or one made by the local<br />
authorities. Such gardens are household-based small-production entities and it is<br />
best that our definition be concerned less with the location than the nature and aim<br />
of the activity.<br />
The geographic separation of homegardens and dwellings is not unique to<br />
Africa. During the first half of the twentieth century in urban United States,<br />
immigrant groups such as Southern Italians practiced substantial levels of
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
325<br />
homegardening for both cultural and economic reasons similar to the poor around<br />
the world today. Evidence on the extent of such practices in the US is found in<br />
historical collections of folklore and ethnographic narratives. A particularly good<br />
example is from Pennsylvania 4 , where the Southern Italians practiced urban<br />
gardening to produce subsistence items (direct consumables) and preserved food<br />
stock, highlighting the centrality of food to Italian culture, family, and gender roles.<br />
More recent examples such as MacNair (2002) demonstrate that the same groups<br />
such as Italians continue to practice urban gardening often under conditions of land<br />
scarcity. In this example from Montreal, it resulted in policy interventions to<br />
regularize the practice under municipal authority. Often the gardens were at some<br />
distance away from the urban dwellings due to the nature of the land market.<br />
Multiple tenancy dwellings, however, made no provisions for land use by occupants.<br />
Available land was scattered and had to be purchased or leased. This meant that<br />
many households maintained multiple plots scattered around the neighborhood or<br />
further afield. As in the case of the African gardens in the semiarid tropics discussed<br />
above, we view these gardens as clear examples of homegardens because of their<br />
centrality to family activities and the absence of municipal facilitation. The<br />
difference in this case is that the management decisions on the part of the<br />
practitioners were conditioned primarily by land availability rather than the labor<br />
costs associated with transporting water. Nonetheless, the majority of homegardens<br />
are located adjacent to the houses.<br />
4. ALLOTMENT GARDENS: A SPECIAL CASE OF HOMEGARDENS<br />
OR AN ‘INDEPENDENT’ SYSTEM?<br />
4.1. Secured access to space: an important question<br />
Stakeholders themselves differ in important ways, mostly in terms of economics and<br />
land tenure. Generally, homegardeners are not the “poorest” residents on the<br />
socioeconomic scale since they already have access to land. Their challenge is<br />
primarily political, mobilizing a fragmented group of individual households with<br />
shared but unrecognized common interests.<br />
The establishment of allotment gardens also requires space within the city<br />
boundaries. This is important only partially to minimize transportation issues.<br />
Women with children need to be near the house because they are generally involved<br />
in multiple household tasks such as cooking, firewood collection, cleaning, and<br />
childcare. However, the reservation and allocation of land for allotment gardens is a<br />
major problem in most urban settings. City authorities rather tend to create public<br />
parks or golf courses, which they consider more in line with the urban disposition.<br />
Generally, gardening does not fit into the conceptualization of urbanization or the<br />
philosophy of urban planners, and this makes it difficult to convince urban<br />
authorities that agriculture in the city is not inherently a problem, but a solution to<br />
various other urban problems (Drescher, 2001).<br />
Securedd access to land is especially relevant when considering the role of trees in<br />
urban gardens of all kinds. To justify capital and labor inputs, the urban gardener
326<br />
A.W. DRESCHER ET AL .<br />
must realize a return on investment. For some crops such as sweet potatoes grown<br />
along roadsides, this occurs in a single growing cycle. However, where soil building,<br />
tree planting, pond construction, or landscaping, for example, are involved, the time<br />
span of guaranteed d access increases, and a number of years is required to recover<br />
labor and capital inputs. For allotment gardeners this means sufficiently long sitespecific<br />
‘lease’ arrangements for the allotment as a whole combined with binding<br />
association ‘rules of access’ for individual participants. For homegardeners this is an<br />
issue of legally formalized land tenure rights, meaning either a deed/title system or<br />
the legal enfranchisement of usufruct rights for individuals in communal ownership<br />
contexts or in long-term spontaneous land occupations.<br />
4.2. Lobbying or political mobilization is particularly relevant to allotment gardens<br />
Overcoming limited vision, economic constraints, and political resistance requires<br />
effective lobbying. Yet, allotment gardeners typically represent some of the most<br />
politically alienated and economically impoverished residents, requiring external<br />
expertise to facilitate participatory lobbying and support for allotment gardens.<br />
Often nongovernmental organizations (NGOs), educational institutions, or<br />
sympathetic municipal agencies play this role. However, effective lobbying also<br />
requires solutions tailored to local conditions. Urban and periurban environments<br />
represent a “lumpy continuum” of human settlement with important and varied<br />
institutional capacities (Iaquinta and Drescher, 2000; 2001; 2005). The respective<br />
roles of home and allotment gardens differ dramatically across this continuum, as do<br />
the lobbying strategies necessary, even within a single municipality.<br />
4.3. Who makes the key management decisions that directly affect outputs?<br />
Observations made in Zambia and Zimbabwe show that community gardens are<br />
often located in unsuitable locations, distant from water sources and/or with bad soil<br />
conditions. Further, as compared to homegardens, trees are generally lacking in the<br />
observed community gardens (Drescher, 1998). These facts suggest that some of the<br />
limitations experienced in community gardens are due more to poor administrative<br />
planning, constrained extension support and gaps in data collection related to the<br />
importance of trees than to problems inherent to community gardens per se.<br />
Bilateral and reciprocal transfer of knowledge between gardeners, extension<br />
officers, and local officials is required to properly understand the smallholders’ land<br />
use system and management strategies (Drescher, 1996).<br />
Development projects tend to over-regulate the maintenance and management of<br />
allotment gardens. Typically, “the authorities” select and provide the seeds and<br />
fertilizers and this minimizes growing traditional vegetables, mitigates the role of<br />
the garden as a site for household experimentation, and marginalizes the gardener as<br />
innovator. Further, attitudes of municipal authorities also reflect the general<br />
underestimation of the multifunctional role of trees in cities, including food security.
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
5. THE HOMEGARDEN MODEL<br />
5.1. Relationship to household livelihood strategy<br />
327<br />
The homegarden model (Fig. 3) is based on the assumption that homegardening is a<br />
process that forms part of the household livelihood strategy. Household livelihood<br />
security is defined as “adequate and sustainable access to income and resources to<br />
meet basic needs, including adequate access to food, potable water, health facilities,<br />
educational opportunities, housing, time for community participation and social<br />
integration” (Frankenberger and McCaston, 1998). For the present discussion, our<br />
emphasis is on the access to food, directly, or by means of access to income and<br />
resources.<br />
The model in Fig. 3 was originally designed for homegardens, but has been<br />
modified to incorporate allotment gardens. The major differences between the two<br />
systems of gardening are in the decision-making process and their differing impacts,<br />
in terms of both quantitative and qualitative results. Allocation of assets in the model<br />
follows the treatment of Swift (1989). Collective assets (e.g., tools, stores and<br />
buildings) are of particular importance for allotment gardens.<br />
The model implies that the household decision to get involved in gardening<br />
depends on factors such as the existence of a supportive general environment, access<br />
to land and water resources, and the availability of specific inputs (seeds,<br />
knowledge, work and time). The model also identifies household vulnerability<br />
factors that either stimulate or inhibit household involvement in subsistence and<br />
market-oriented food production. The risk of livelihood failure determines the level<br />
of vulnerability of a household to income, food, health, and nutritional insecurity.<br />
Therefore, livelihoods are secure when households have secured ownership of—or<br />
access to—resources and income earning activities, including reserves and assets, to<br />
offset risks, ease shocks, and meet contingencies (Chambers, 1989).<br />
The model in Fig. 3 implicitly depicts homegardening as embedded in the<br />
livelihood system, interacting with the socioeconomic and environmental conditions<br />
of the larger system. It helps to identify factors that promote or inhibit household<br />
gardening and assists in the development of scenarios for different contexts<br />
regarding climate, space, politics, institutional framework, culture, and economics.<br />
<strong>Homegardens</strong> support important farm-development activities; some farm inputs<br />
come from homegarden activities such as plant propagation, raising and housing<br />
draught animals, and making and repairing tools. New crops and farming techniques<br />
are often first tried out in the homegarden, which is also an area for drying,<br />
processing, and storing farm products (Landon-Lane, 2004; Yamada and Osaqui,<br />
2006). In small compound homegardens in Zambia, sweet potato seedlings are<br />
planted and later transferred into the fields during the rainy season. Sweet potato<br />
leaves are also used as a vegetable both in dry and rainy season. Nearly 40% of the<br />
compound residents of Lusaka are engaged in staple crop production during the<br />
rainy season, while about 25% are engaged in homegardening and community<br />
gardening (Drescher, 1998).
328<br />
A.W. DRESCHER ET AL .<br />
Figure 3. The Homegarden Model (adapted from Drescher, 1998).<br />
5.2. Why so important in the urban context?<br />
Urban environments differ considerably from rural environments. Urban poor<br />
dwellers are more likely to report food insecurity and heavy dependency on urban<br />
markets (Zalilah and Khor, 2004). At the same time, there are clear signs that poverty<br />
and malnutrition in cities are increasing, especially in slum areas and high-density
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
329<br />
compounds (FAO, 2001; Iaquinta and Drescher, 2002). Low purchasing power and<br />
declining access to food are major problems in many rapidly growing cities in the<br />
developing world (Drescher and Iaquinta, 2003).<br />
Both home- and allotment gardening can partly compensate for the deficiencies<br />
of urban poor households and alleviate food insecurity. Thus, secured access to<br />
resources such as land, water, seeds, and tools is key to increasing food security in<br />
cities. Further, food production is only one dimension, albeit t an important one, of<br />
the benefits to be derived from urban and periurban production. With proper<br />
assistance and management techniques, these environments are positively impacted<br />
through provision of shade, microclimate modification, waste recycling, soil<br />
stabilization, and soil building.<br />
5.3. Applying the model to allotment gardens<br />
If we apply the homegarden model (Fig. 3) to allotment gardens, it is evident, that<br />
the two systems are similar regarding access to resources, assets, activities, and<br />
outcomes. However, the arena of decision-making becomes more complex.<br />
Essentially, we need to “nest” the individual family as a decision-maker within the<br />
allotment association as the decision-broker. The decision module appears as a<br />
shaded pentagon in the center of the model and represents the decision-making<br />
process (e.g., what to grow, when to grow, where to grow, with whom and how to<br />
interact and <strong>coop</strong>erate, the balance between short and long-term investments, etc.).<br />
For homegardens, it is simply a single module (pentagon). For allotment gardens,<br />
however, it appears as a series of household pentagons nested within a larger<br />
“association” module. In practice, the entire decision-making process becomes more<br />
transparent and probably more standardized in the case of allotment gardens because<br />
individual family decisions are now directly guided and influenced by the<br />
association in a more public forum. Assets and activities can be better shared in<br />
allotment gardens and the transcendent outcomes are eventually more visible,<br />
mainly regarding community empowerment, social peace and status, and economic<br />
power. In this sense, we do not consider individual parcels in allotment gardens as<br />
independent farming systems in the way that homegardens are. However, an<br />
allotment garden association is an institution and it may conflict with preexisting<br />
institutions—particularly but not exclusively—those of a traditional form. That is<br />
why it is so important to understand the relationship between the type of urban or<br />
periurban environment and the respective institutions (Iaquinta and Drescher, 2000;<br />
2001; 2005).<br />
5.4. Contrasting roles of homegardens vs. allotment gardens within the general household<br />
gardening model<br />
Allotment gardens, properly institutionalized and integrated into urban planning have<br />
their biggest influence on the level of the (non-) supporting structures and can<br />
regularize better entitlement and access to resources. Thus, allotment gardeners have
330<br />
A.W. DRESCHER ET AL .<br />
an enhanced voice through the association, while homegardeners typically remain<br />
isolated, or “just “ gardeners!”<br />
Because homegardening is seen primarily as a private activity, there is little<br />
public support for these gardens. Homegardening is only done when the specific<br />
circumstances permit it; for the most part space availability is the major determinant.<br />
Public support is more likely to occur in relation to allotment gardens, because of<br />
their greater visibility.<br />
With respect to the environmental impact and output of both activities, the<br />
differences depend primarily on the management strategies employed. For both<br />
systems, space is restricted. Allotment gardens allow gardening for those who do not<br />
have access to land near their residences.<br />
Thus, in terms of access, both homegardens and allotment gardens have a clear<br />
political dimension. On the one hand, The World Food Summit 2002 (FAO, 2002)<br />
reaffirmed the right of everyone to have access to safe, nutritious, and culturally<br />
relevant food. The ability to grow food is one important dimension of this access<br />
relevant to both types of gardens. On the other hand, community empowerment,<br />
which is especially relevant to allotment gardens, is inherently a process of<br />
advocacy and political negotiation among municipal authorities, local residents, and<br />
various interest groups.<br />
6. HOW DO MANAGEMENT STRATEGIES FOR HOMEGARDENS<br />
AND ALLOTMENT GARDENS DIFFERENTIALLY IMPACT OUTPUTS?<br />
6.1. The example of species diversity: observations from Africa<br />
Species diversity or “garden biodiversity” provides an excellent example of the way<br />
management strategies and their outcomes differ between the two systems. Species<br />
diversity is determined by two factors: the number of species per garden and the<br />
abundance of each species within the community in a given area. Which species are<br />
planted and how much of each species gets planted represent fundamental<br />
management decisions.<br />
Important inferences can be drawn from a study of rural homegardens and rural<br />
community gardens in southern Zimbabwe (Drescher et al., 1999), where, clear<br />
differences were found in the diversity of plant species in community and individual<br />
gardens. Individual gardens (average 8.6; range 5 to 12 species) showed a higher<br />
species diversity than community gardens. Community gardens averaged only four<br />
species (Drescher et al., 1999). In a related study, garden species-diversity was<br />
shown to be positively correlated with the prevalence of biological antagonists of<br />
crop pests (Drescher, 1998). Together, these results point to the benefits of<br />
promoting individual gardens and the need for extension strategies adapted<br />
specifically for community gardens.<br />
Sweet potato, grown for both its nutritious leaves and starchy tubers, further<br />
illustrates the different approaches to management in the two types of gardens.<br />
Sweet potatoes were cultivated in all individual gardens but in only one community<br />
garden (Drescher et al., 1999). In homegardens, sweet potatoes serve as early patch
331<br />
leafy greens and are transplanted to fields later during the rainy season. This is a<br />
household decision that contributes to a more balanced diet, provides good early<br />
ground cover, and releases land for subsequent cultivation. Authorities decided,<br />
however, that the sweet potato would not be recommended for planting in<br />
community gardens, hence distributed no sweet potato seeds to gardeners. These<br />
two management systems could be brought into greater alignment with appropriate<br />
research and support. One opportunity for this is to transfer greater management<br />
authority to allotment associations, which typically function more interactively with<br />
households than do formal institutions and authorities.<br />
6.2. Other outputs<br />
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
In private homegardens, output in terms of yield can suffer due to the lack of labor<br />
and time. This can be more easily compensated for in allotment gardens, first by<br />
more <strong>coop</strong>eration between families and second by economies of scale relative to<br />
water supply and other capital investments and crop management techniques.<br />
6.3. Marketing of surplus easier in allotment gardens?<br />
Allotment gardens can produce marketable surplus produce too. Greater<br />
concentration of output creates economies of scale wherein both direct marketing<br />
and production are facilitated. In Cape Town, South Africa, for instance, the<br />
Siyazama community allotment garden produced both for the market as well as for<br />
home consumption of 15 dependent families. Public support for allotment gardens<br />
might help urban poor to get better access to markets 5,6 .<br />
The lack of public support for homegardens, however, reduces the number of<br />
such gardens in cities. Current land use planning, multistory housing and land use<br />
competition from different sectors often limit the open space and space for<br />
gardening. Nonetheless, in most cities, open space, unused sites, and idle land are<br />
still widely available. Fig. 4 shows the use of open spaces for gardening near Manila<br />
International Airport. Elsewhere in the Philippines, tax policy has been used to<br />
stimulate changes in land use. For example, in Cagayan de Oro, the municipal<br />
authorities taxed unused open lands motivating property owners to make the sites<br />
available to poor city dwellers for crop production.<br />
In homegardens, important information flows through informal channels whereas<br />
the allotment gardens have enhanced information access through associations and<br />
extension support. This includes information on pest abatement strategies,<br />
management, and technologies. Effective systems combine the delivery and<br />
scientific advantages of extension with the firsthand user knowledge of gardeners in<br />
a reciprocal fashion.<br />
Social interaction is higher in allotment gardens because of the joint activities of<br />
different families in close spatial proximity. Social interaction is even higher if there<br />
is a corporate identity like membership in an allotment garden association. Exchange<br />
of information, joint activities, and family-based participatory learning is more
332<br />
A.W. DRESCHER ET AL .<br />
likely to happen in allotment gardens while homegardening is in most cases a purely<br />
family-based activity.<br />
Figure 4. Gardens near Manila International Airport with densely populated compound<br />
at the top.<br />
In contrast to homegardens, allotment gardens enable users to learn democratic<br />
rules more efficiently because they have to resolve many problems within the<br />
association. Usually, problems arise regarding the use of land, equitable distribution<br />
of land and water, and joint community work. Where democratic rules and civil<br />
society associations differ significantly from customary practices, participants need<br />
guidance and support in acquiring the necessary skills. New cultural forms and<br />
institutions may be strongly resisted since they can upset existing political and social<br />
arrangements. In such situations participatory process planning can be combined<br />
with knowledge of the institutional context to direct the structure and functioning of<br />
the association.
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
7. INSTITUTIONAL CONTEXT<br />
333<br />
In Table 1, we present the institutional context surrounding homegardens and<br />
allotment gardens. While it does not survey the complete range of possible relevant<br />
institutions, it gives a good idea of the complex ways that homegardens and<br />
allotment gardens are differentially affected. A more complete accounting of the<br />
context would classify institutions along at least two important dimensions: formal<br />
versus informal and traditional versus modern. Importantly, these two dimensions<br />
are neither collinear nor orthogonal. The utility of such a classification goes beyond<br />
the simple question of support or lack of support for one or the other type of<br />
gardening. The exercise points to the linkages between urban gardening and other<br />
social problems in the community, fostering the possibility for constituency building<br />
through integrated problem-solving. Nonetheless, even without such synergistic<br />
system gains, elaboration of the institutional context provides valuable insights into<br />
the gardening model presented and into the processes by which urban gardening –<br />
whether homegardening or allotment gardening – is facilitated or hindered.<br />
8. RECOMMENDATIONS<br />
• Allotment gardens need to be institutionalized. Ideally, they should be part of the<br />
concept of urbanization wherein land is specifically set aside for such activities<br />
in the planning process.<br />
• Lobbying or public advocacy is required to support both garden systems.<br />
However, the nature of such lobbying efforts differs significantly between the<br />
two systems. <strong>Homegardens</strong> need public advocacy and extension services while<br />
allotment gardens additionally require political intervention to secure land,<br />
organize access, and support development.<br />
• Allotment gardens should be developed as a package of services, including for<br />
example extension outreach, community and infrastructure building, delivery of<br />
health care, etc. Allotment gardens will be more protected and access to them<br />
will be better coordinated in such a configuration. For example, allotment<br />
gardens are well adapted to periurban environments when authorities are willing<br />
to regularize spontaneous occupations. Small- and medium-sized towns provide<br />
ideal conditions for such early intervention and land preservation when<br />
combined with proper waste management and extension services.<br />
• Housing design and planning for backyards should facilitate homegardens.<br />
• Provision of adequate water is a problem in many cities, and public water use<br />
often restricted. Solutions include urban rainwater harvesting and the use of grey<br />
water (i.e., non-septic household wastewater), but their effectiveness depends on<br />
developing cost-effective locally adapted designs/technologies. Other solutions<br />
such as modern irrigation techniques can be implemented rather cheaply in<br />
conjunction with the preceding but depend more on extension information<br />
services to be effective.<br />
• The urban-periurban continuum is not uniform. Participatory process planning<br />
should be framed within the components of the periurban-urban typology and<br />
their corresponding institutions.
Institutional<br />
Type of urban gardens<br />
(non-)<br />
<strong>Homegardens</strong> Allotment gardens<br />
supporting<br />
structures 1 Characteristics Impact Characteristics Impact<br />
Allotment<br />
garden<br />
associations<br />
Not applicable Not applicable Sometimes existent Supporting<br />
City councils Mostly neglecting Strongly inhibiting Some recognition Sometimes<br />
supporting<br />
Town Planning Generally not aware of the importance<br />
of homegardens<br />
Educational<br />
system<br />
Health<br />
Authorities<br />
Table 1. Institutional contexts of urban homegardens and allotment gardens.<br />
Strongly inhibiting Generally not aware of the importance<br />
of urban food production<br />
Strongly<br />
inhibiting<br />
Ignorant in many countries Little support Ignorant in many countries Little support<br />
Generally not involved and not aware Inhibiting but<br />
probably no effect<br />
Generally not involved Inhibiting<br />
334<br />
A.W. DRESCHER ET AL .
Water<br />
authorities<br />
Prohibiting the use of public water for<br />
gardening without promoting<br />
alternatives (e.g. wells, rain water<br />
harvesting, grey water use)<br />
Inhibiting Prohibiting use of public water for<br />
gardening without promoting<br />
alternatives (e.g. wells, rain water<br />
harvesting, grey water use)<br />
Inhibiting<br />
Credits Generally not available Inhibiting Generally not available Inhibiting<br />
Extension<br />
Services<br />
Generally not available, rarely in the<br />
context of development projects<br />
Markets Mostly bartering with neighbors;<br />
production too small<br />
Markets and<br />
market<br />
information<br />
Direct marketing, bartering, household<br />
networks<br />
No market information system in place<br />
Inhibiting Generally not available, sometimes in<br />
the context of development projects<br />
Supporting social<br />
interaction<br />
Sometimes<br />
supporting<br />
Sometimes<br />
inhibiting<br />
Access to markets difficult, direct<br />
marketing possible but not supported<br />
Direct marketing, bartering,<br />
household networks<br />
No market information system in<br />
place<br />
Inhibiting<br />
Inhibiting<br />
Sometimes<br />
supporting<br />
Sometimes<br />
inhibiting<br />
1 By non-supporting structures we mean those institutions and bodies, which do not have policies on mechanisms to directly support gardens,<br />
and may even impede their development. Supporting structures in contrast clearly support the development of gardens.<br />
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS GG<br />
335
336<br />
A.W. DRESCHER ET AL .<br />
• Extension strategies should be elaborated in close <strong>coop</strong>eration among all<br />
stakeholders, especially smallholders and extension officers.<br />
• Strong advocacy for the multifunctional role of trees in both gardens and the<br />
broader urban context is required. Integration of this concept into extension<br />
programs aimed at both homegardens and allotment gardens is needed.<br />
ENDNOTES<br />
1. Velez C. 1997. Homegardening as a strategy for food and nutrition security: a<br />
case study of selected households in Lantapan, Bukidnon. MSc paper in<br />
Applied Nutrition, submitted to the Faculty of the Graduate School, University<br />
of the Philippines, Los Baños.<br />
2. Urbina C.D., Miso A.U., Holmer R.J. 2005. Socio-economic impact of the<br />
allotment garden project in Cagayan de Oro. Paper presented at the 6th<br />
PUDSEA Network Conference. July 11-15, 2005, Cagayan de Oro, Philippines.<br />
3. Segne J.B., Salcedo J.M. and Guiral H. 2004. Implementation of an integrated<br />
solid waste management system in two Sitios of Cagayan de Oro. Proceedings<br />
of the 16th NOMCARRD Regional symposium on research and development<br />
highlights, August 5-6, 2004, Central Mindanao University, Musuan, Bukidnon,<br />
Philippines (in print).<br />
4. Saverino J.L. 1995. ‘Domani Ci Zappa`’: Italian immigration and ethnicity in<br />
Pennsylvania. In: Pennsylvania folk life. 45 (Autumn): pp. 2-22.<br />
5. Abalimi, pers. comm., 2000; see also in Motion Magazine 2002, interviews<br />
related to the United Nations World Summit on Sustainable Development in<br />
Johannesburg, South Africa, August 26 - September 4, 2002.<br />
6. Food and Agriculture Organization (FAO) 2001. Proceedings of the expert<br />
consultation on urban horticulture in Southern Africa. Stellenbosch, South Africa.<br />
REFERENCES<br />
Agbayani A.L.P., Holmer R.J., Potutan G.E. and Schnitzler W.H. 2001. Quality and quantity<br />
requirements for vegetables by private households, vendors and institutional users in a<br />
Philippine urban setting. Urban Agr Mag 5: 56 – 57, Leusden.<br />
Arias G. 2000. Análisis de las políticas publicas en la agricultura urbana caso Texcoco, México.<br />
Dirección de Desarrollo Rural, Ayuntamiento de Texcoco, México, 20p. Programa de Gestão<br />
Urbana para a América Latina e o Caribe – PGU-ALC (CNUAH-HABITAT/PNUD)<br />
http://www.ipes.org/au /estudioc/texcoco.pdf (last accessed: January 2006).<br />
Boncodin R., Prain G. and Campilan D. 2000. Dynamics in tropical homegardens. Urban Agr<br />
Mag 1: 19 – 20, Leusden.<br />
Chambers R. 1989. Editorial: introduction: vulnerability, coping, and policy. IDS Bull 20(2): 1 – 7.<br />
Christanty L. 1990. Home gardens in tropical Asia with special reference to Indonesia. In:<br />
Landauer K. and Brazil M. (eds), 1990. <strong>Tropical</strong> home gardens, pp. 9 – 20. United<br />
Nations University Press, Tokyo.<br />
Cruz M.C. and Medina R.S. (eds). 2003. Agriculture in the city. IDRC, Ottawa, 244p.<br />
Drescher A.W. 1996. Management strategies in African homegardens and the need for new<br />
extension approaches. In: Heidhues F. and Fadani A. (eds), Food security and<br />
innovations: Successes and lessons learned, pp 231 – 246. Peter Lang, Frankfurt.
URBAN U HOMEGARDENS AND ALLOTMENT GARDENS G<br />
337<br />
Drescher A.W. 1998. Hausgärten in Afrikanischen Räumen – Bewirtschaftung nachhaltiger<br />
Produktionssysteme und Strategien der Ernährungssicherung in Sambia und Simbabwe.<br />
Sozioökonomische Prozesse in Asien und Afrika, 4. Centaurus, Pfaffenweiler, 290p.<br />
Drescher A.W. 2001. The integration of urban agriculture into urban planning – An analysis of the<br />
current status and constraints. In: Annotated bibliography on urban agriculture, pp 343 – 357.<br />
ETC Urban Agriculture Programme and Swedish International development Agency (SIDA),<br />
Leusden. http://www.ruaf.org/bibliography/annotated/014.pdf (last accessed: January 2006).<br />
Drescher A.W. and Iaquinta D.L. 2003. Urbanization – linking development across the changing<br />
landscape. Economic and social department publication (ESAC), FAO, Rome, 121p. http://<br />
www.fao.org/fcit/docs/sofa_01.pdf (02.pdf – 03.pdf) (last accessed: January 2006).<br />
Drescher A.W., Hagmann J. and Chuma E. 1999. Home gardens - a neglected potential for<br />
food security and sustainable land management in the communal lands of Zimbabwe. In:<br />
Der Tropenlandwirt, 2/1999, pp 163–180. Kassel-Witzenhausen.<br />
Food and Agriculture Organization (FAO) 1998. Food security and community nutrition, No.<br />
22, Food and Agriculture Organization of the UN, Rome, 72p.<br />
Food and Agriculture Organization (FAO) 2001. FAO warns of increasing malnutrition<br />
among urban poor. FAO Press release 01/36. 1p. http://www.fao.org/WAICENT/OIS/<br />
PRESS_NE/PRESSENG /2001/pren0136.htm (last accessed: January 2006).<br />
Food and Agriculture Organization (FAO) 2002. Declaration of the World Food Summit: five<br />
years later. In: Report of the World Food Summit: five years later. FAO, Rome,<br />
http://www.fao.org/DOCREP/MEETING/005/Y7106E/Y7106E09.htm (last accessed: January<br />
2006).<br />
Frankenberger T.R. and McCaston M.K. 1998. The household livelihood security concept. In:<br />
FAO 1998. Food Nutr Agr 22: 30-35 http://www.fao.org/docrep/X0051T/X0051T00.htm<br />
(last accessed: January 2006).<br />
Guanzon Y.B. and Holmer R.J. 2003. Basic cultural management practices for vegetable<br />
production in urban areas of the Philippines. Urban Agr Mag 10: 14 – 15, Leusden.<br />
Hetterschijt T. 2004. Our daily realities: Urban organic homegardens in Lima, Peru. Urban<br />
Agr Mag (Gender and Urban Agriculture, May 2005) 12: 10-11, Leusden.<br />
Holmer R.J., Clavejo M.T., Dongus S. and Drescher A.W. 2003. Allotment gardens for<br />
Philippine cities. Urban Agr Mag 11: 29 – 31, Leusden.<br />
Hoogerbrugge I. and Fresco L.O. 1993. Homegarden systems: Agricultural characteristics and<br />
challenges. Gatekeeper, No. 39, International Institute for Environment and Development,<br />
London, 23p.<br />
Iaquinta D.L. and Drescher A.W. 2000. Defining periurban: towards guidelines for<br />
understanding rural-urban linkages and their connection to institutional contexts, pp 8 –<br />
27. Land Reform 2000/2, FAO, Rome.<br />
Iaquinta D.L. and Drescher A.W. 2001. More than the spatial fringe: An application of the<br />
periurban typology to planning and management of natural resources in the periurban,<br />
DPU International Conference, “Rural-urban encounters: Managing the environment of<br />
the periurban interface”, College of London, 9 – 10 November 2001. http://www.ucl.<br />
ac.uk/dpu/pui/events/EPM_conf_ abstracts.htm# DefiningthePeri-Urban (last accessed:<br />
January 2006).<br />
Iaquinta D.L. and Drescher A.W. 2002. Food security in cities – A new challenge to<br />
development. In: Brebbia C.A., Matrin-Duque J.F., and Wadhwa L.C. (eds), The<br />
sustainable city II – Urban regeneration and sustainability. Advances in Architecture, pp<br />
983 – 994. Wessex Institute of Technology, WIT Press, Wessex.<br />
Iaquinta D.L. and Drescher A.W. 2005. Defining periurban: A framework for transportation<br />
planning in India, periurban workshop, Leeds (April 11 – 12, 2005). https://www.periurban.<br />
org/pub/hom / me/events/0504_leeds/documents/PRESENTATION- DefiningPeriurban-Iaquinta.<br />
pdf (last accessed: January 2006).
338<br />
A.W. DRESCHER ET AL .<br />
Innenministerium B-W. 2004. Climate booklet for urban development references for zoning<br />
and planning. Amt für Umweltschutz, http://www.staedtebauliche-klimafibel.de/<br />
Climate_Booklet/index-2.htm (last accessed: January 2006).<br />
Jacobi P., Drescher A.W. and Amend J. 2000. Urban agriculture: Justification and planning<br />
guidelines. GTZ, Eschborn, 70p, http://www.cityfarmer.org/uajustification.html (last<br />
accessed: January 2006).<br />
Jarlöv L. 2000. Urban Agriculture in South Africa. In: Hoffmann H. and Mathey K. (eds),<br />
Urban agriculture and horticulture: the linkage with urban planning. International<br />
Symposium (July 2000), Berlin (on cd-rom). http://www.ruaf.org/conference/<br />
info_market/econf_papers/14jarlov.doc (last accessed: January 2006).<br />
Kasch G. 2001. Deutsches Kleingärtnermuseum in Leipzig: Deutschlands Kleingärtner vom 19.<br />
zum 21. Jahrhundert. Band 4, Sächsische Landesstelle für Museumswesen, Chemnitz, 128p.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst<br />
61/62: 135 – 152.<br />
Landeshauptstadt D. 1998. Umweltbericht. Stadtklima von Dresden. Umweltamt, 39p. http://<br />
www. dresden.de/pdf /infoblaetter/umweltbericht_text.pdf (last t accessed: Janua ary<br />
2006).<br />
Landon-Lane C. 2004. Livelihoods grow in gardens: Diversifying rural incomes through<br />
homegardens. FAO Diversification booklet 2. Agricultural Support Systems Division, FAO,<br />
Rome. 58p.<br />
MacNair E. 2002. The garden city handbook: How to create and protect community gardens<br />
in Greater Victoria. Polis Project on Ecological Governance. University of Victoria,<br />
Victoria BC, 34p. http:// www.polisproject.org/polis2/PDFs/the%20garden%20city%20<br />
handbook.pdf (last t accessed: January 2006).<br />
Mougeot L.J.A. (ed.). 2005. AGROPOLIS: The social, political, and environmental<br />
dimensions of urban agriculture. CRDI, Earthscan. 308p.<br />
Pinderhughes R. 2004. Alternative urban futures: Planning for sustainable development in<br />
cities throughout the world. Rowman & Littlefield Pub Inc. Lanham, MD, 271p.<br />
Prosterman R. and Mitchell R. 2002. Concept for land reform on Java. Paper presented at the<br />
seminar: Rethinking land reform in Indonesia, Jakarta (8 May 2002). On file with the<br />
Rural Development Institute, Seattle. http://www.rdiland.org/PDF/RDI_LandReform<br />
OnJava.pdf (last accessed: February 2006).<br />
Ranasinghe T.T. 2005. Family business gardens: Agricultural options in remodelling and<br />
modernising tsunami devastated townships in Sri Lanka. Cityfarmer, 9p.<br />
www.cityfarmer.org/subsouthasia.html (last accessed: 27 January 2006).<br />
Swift J. 1989. Why are rural people vulnerable to famine? In: Chambers R. (ed.), IDS Bull<br />
20(2): 8 – 15.<br />
United Nations Centre for Human Settlements (UNCHS) 2001. The state of the world’s cities<br />
report 2001.United Nations Centre for Human Settlements (UNCHS), Nairobi, 126p.<br />
Vasey D.E. 1985. Household gardens and their niche in Port Moresby, Papua New Guinea.<br />
Food Nutr. Bull 7(3): 37 – 52.<br />
Vélez-Guerra A. 2004. Multiple means of access to land for urban agriculture: A case study<br />
of farmers’ groups in Bamako, Mali. Cities feeding people report series, December 2004,<br />
International Development Research Centre (IDRC), Ottawa, 88p.<br />
Yamada M. and Osaqui H.M.L. 2006. The role of homegarden for agroforestry development:<br />
lessons from a Japanese-Brazilian settlement in the Amazon. In: Kumar B.M. and Nair<br />
P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry,<br />
pp 299 – 316. Springer Science, Dordrecht.<br />
Zalilah M.S. and Khor G.L. 2004. Indicators and nutritional outcomes of household food<br />
insecurity among a sample of rural Malaysian women. Pakistan J Nutr 3: 50 – 55.
CHAPTER 19<br />
ARE TROPICAL HOMEGARDENS<br />
SUSTAINABLE? SOME EVIDENCE FROM<br />
CENTRAL SULAWESI, INDONESIA<br />
K. KEHLENBECK AND B.L. MAASS*<br />
Institute for Crop and Animal Production in the Tropics, Georg-August-University<br />
Göttingen, Grisebachstr. 6, D-37077 Göttingen, Germany; *E-mail:<br />
<br />
Keywords: Biodiversity, In situ conservation, Soil fertility, Sustainability indicators,<br />
Vegetation dynamics.<br />
Abstract. <strong>Homegardens</strong> are regarded as sustainable agricultural production systems, although<br />
support for this statement by quantitative data has been rare. Out of the suggested<br />
indicators/descriptors for assessing sustainability, plant diversity has been frequently studied.<br />
However, species diversity is not static: it varies with time and according to ecological and<br />
socioeconomic factors and/or characteristics of the gardens and gardeners. In order to evaluate<br />
sustainability of the homegarden system, we assessed soil fertility parameters and changes in<br />
diversity of useful plants over time during 2001 – 2004 in 30 homegardens from three villages<br />
adjacent to the Lore Lindu National Park in Central Sulawesi, Indonesia. Soil carbon (C) and<br />
nitrogen (N) contents decreased over time. In large gardens with different production zones, soil<br />
of vegetable zones contained less C and N than that of cacao (Theobroma cacao) zones.<br />
Richness of useful plant species was high and increased over time, from 149 species in 2001 to<br />
168 in 2003. Species composition of homegardens from one village, mainly inhabited by<br />
migrants, contrasted strongly with those from the other two, inhabited by native farmers.<br />
Diversity of useful plants was lower in the migrant village, where soil fertility was low, too.<br />
Plant diversity appeared to be influenced to varying extent by a combination of factors such as<br />
garden size/age, soil fertility, ethnicity and age of gardener, and market access. The surveyed<br />
homegardens did not seem to be managed appropriately to ensure sustainability in terms of soil<br />
fertility although they had a high diversity of useful plants.<br />
1. INTRODUCTION<br />
<strong>Tropical</strong> homegardens are generally regarded as sustainable production systems<br />
(Christanty, 1990; Landauer and Brazil, 1990; Soemarwoto and Conway, 1991;<br />
339<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 339–354.<br />
© 2006 Springer. Printed in the Netherlands.
340<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
Torquebiau, 1992; Abdoellah et al., 2001; Kumar and Nair, 2004). However,<br />
quantitative support for this statement is mostly lacking, particularly because of the<br />
difficulties in measuring sustainability (Kumar and Nair, 2004). Therefore,<br />
researchers rely on indirect evidences using certain sustainability descriptors and/or<br />
indicators (Torquebiau, 1992; Huxley, 1999).<br />
Among the available indicators, perhaps the criterion most used in homegarden<br />
research is biodiversity, particularly plant species diversity. The wide spectrum of<br />
useful plants creates a multilayered vegetation structure in homegardens, which is<br />
responsible for many benefits and advantages of the system. This diversity results in<br />
favorable microclimate, reduced risk of pests and diseases, efficient use of<br />
resources, year-round availability of products, and soil fertility maintenance. Thus,<br />
plant diversity is considered as contributing substantially to the sustainability of the<br />
system (Soemarwoto and Conway, 1991; Torquebiau, 1992).<br />
Because of their diversity, homegardens are also regarded as an ideal production<br />
system for in situ conservation of plant genetic resources (Watson and Eyzaguirre,<br />
2002), crucial for long-term sustainability. However, crop diversity is influenced by<br />
different factors such as size and age of homegardens or age of gardeners<br />
(Abdoellah et al., 2001; Gutiérrez et al., 2004). Besides, environmental and<br />
socioeconomic characteristics are known to influence homegarden diversity<br />
(Michon and Mary, 1994; Wezel and Bender, 2003; Gutiérrez et al., 2004).<br />
Nevertheless, the suitability of biodiversity as a sustainability indicator needs to be<br />
critically examined because there is no threshold value for an ideal number of<br />
species in a sustainable system. In addition, diversity seems to be highly variable<br />
over time, and the homegarden research so far has neglected to quantify such<br />
changes.<br />
Another sustainability indicator generally accepted is soil fertility (Torquebiau,<br />
1992; Huxley, 1999; Kumar and Nair, 2004). In homegardens, soil fertility is said to<br />
be maintained due to the closed nutrient cycling and low nutrient-export through<br />
harvested products (Gajaseni and Gajaseni, 1999; Kumar and Nair, 2004). Dense<br />
layers of litter and undergrowth are supposed to prevent or at least reduce soil<br />
erosion in homegardens (Karyono, 1990; Soemarwoto and Conway, 1991).<br />
Investigation of soil fertility parameters is common in homegarden research (Jensen,<br />
1993; Gajaseni and Gajaseni, 1999), whereas soil erosion has rarely been assessed<br />
(Torquebiau, 1992). Usually, statements on sustainable soil fertility management in<br />
homegardens are supported only by a single ‘snapshot’ of the status quo without any<br />
further consideration on soil fertility variation over space and time. The role of<br />
different management practices leading to this variation in the long-term is not<br />
sufficiently investigated.<br />
In association with the multidisciplinary German-Indonesian collaborative<br />
research program STORMA (Stability of Rainforest Margins in Indonesia, SFB<br />
552), this study aimed at assessing the sustainability of selected homegardens on the<br />
island of Sulawesi with the help of selected sustainability indicators. A first<br />
assessment from a comprehensive dataset is presented here, focusing on aspects of:<br />
• Stability/dynamics in diversity of useful plants over time<br />
• Changes in soil fertility over time
341<br />
• Specific influences of selected factors on diversity of useful plants<br />
The ecological indicators ‘diversity of useful plants’ and ‘soil fertility’ were<br />
chosen because data from a previous study of the same homegardens were available<br />
(Kehlenbeck and Maass, 2004) and both indicators are essential for assured<br />
productivity of the system. Besides ecological indicators, social and economic<br />
indicators (e.g., labor requirement, cash input and biophysical output) as suggested<br />
by Torquebiau (1992) and Kumar and Nair (2004) have been assessed under the<br />
overall project, but those results will be presented elsewhere.<br />
2.1. Study area<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
2. MATERIALS AND METHODS<br />
The study was conducted from March to November 2001 and from June 2003 to<br />
June 2004 in the Napu Valley (1°23’ to 37’S, 120°18’ to 20’E), located on the<br />
eastern margins of the Lore Lindu National Park in Central Sulawesi (Indonesia),<br />
about 100 km south of the city of Palu. Elevation is around 1100 m above sea level;<br />
annual precipitation is about 2000 mm with a mean temperature of 21°C. Natural<br />
vegetation is classified as lower montane rainforest (Whitten et al., 1987); soils are<br />
mostly Cambisols (FAO; USDA: Tropepts, Inceptisols) and Fluvisols (Fluvents,<br />
Entisols).<br />
The initially low human population density has been increasing in the region,<br />
especially since the 1980s, due to migration. Most inhabitants are farmers, and offfarm<br />
employment opportunities are scarce. Agricultural production is mainly based<br />
on paddy rice (Oryza sativa) production for subsistence, agroforestry with cacao<br />
(Theobroma cacao) and coffee (Coffea spp.) as cash crops, and rain-fed annual<br />
crops (Kehlenbeck and Maass, 2004). Large areas of the Napu Valley are under<br />
fallow or degraded grasslands.<br />
Table 1. Characteristics of three villages studied in the Napu Valley, Central Sulawesi,<br />
Indonesia.<br />
Parameters Wuasa Rompo Siliwanga<br />
Year of foundation 1892 1915 1992<br />
Inhabitants (no.) 2600 (2003) 400 (2004) 600 (2004)<br />
Ethnicity mixed >75% indigenous >75% migrants<br />
Distance to paved road 0 km 5 km 0 km<br />
Market access good poor medium<br />
Source: Zeller et al. (2001) and Kehlenbeck (unpublished data).<br />
For this research, three villages, which differed in their market access and origin<br />
of inhabitants, were chosen (Table 1). Wuasa is the administrative center of the<br />
Napu Valley with a junior and senior high school, a small hospital, many shops and<br />
offices as well as a market place. Rompo is a small village surrounded by forest,
342<br />
accessible by a dirt road. Siliwanga was founded only recently for settling migrant<br />
families, mostly from Bali, in the context of the transmigration program of the<br />
Indonesian government (Mayor of Siliwanga, pers. comm., 2001). For convenience,<br />
the three villages were labeled as ‘market village’ (Wuasa), ‘forest village’<br />
(Rompo), and ‘migrant village’ (Siliwanga).<br />
2.2. Data collection<br />
Ten households with homegardens were randomly selected from each village.<br />
Information about local knowledge and management of the same homegardens was<br />
gathered in 2001 and 2003/2004, except for one garden in the migrant village that<br />
was abandoned in 2002. Gardeners were individually interviewed using an<br />
unstructured questionnaire with questions on age and functions of the homegarden,<br />
inputs and outputs, and the use of homegarden products, among others. Data<br />
concerning household characteristics, such as age, formal education, ethnic group, or<br />
occupation of the household members were also gathered through interviews, partly<br />
within larger surveys of the STORMA project.<br />
Homegarden size was measured, excluding the area occupied by the house.<br />
Complete inventories were carried out in 2001 (July – October) and 2003 (July –<br />
August) to assess number of species and abundance of crops and ornamentals. In<br />
this study, the term ‘crops’ is applied to all useful plant species, including planted<br />
and spontaneously occurring except the ornamentals. Presence of weeds, defined as<br />
undesired plants from the gardener’s view, was documented but not quantified.<br />
Plants were recorded with local and/or scientific names. Crop species were classified<br />
into different use categories (Kehlenbeck and Maass, 2004).<br />
In 2001, 20 soil samples per garden were randomly collected from 0 – 15 cm<br />
depth and mixed, except for four large gardens, where soil was sampled separately<br />
according to production zone (vegetables, coffee/cacao, or fruit trees). In 2003/2004,<br />
five soil samples per garden were randomly collected at 0 – 15 cm depth and mixed,<br />
2<br />
distinct production zones, five samples per zone were collected and mixed. Due to<br />
these different sampling strategies, soil fertility change over time was analyzed only<br />
in a subgroup of homegardens with comparable soil sampling in both years, i.e.,<br />
gardens with one mixed vegetation zone only (n = 10) as well as large gardens<br />
(n = 4) already sampled by zones in 2001. Total C and N were quantified by C/N-<br />
Autoanalyser and pH with an electrode (soil: water ratio, 1:2.5). Bulk density was<br />
determined in 2003 only by assessing the dry weight of soil samples with known<br />
field volume.<br />
2.3. Data analysis<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
if the garden was small (< 350 m<br />
) or planted uniformly. In large gardens with<br />
Species density (no. of spp./100 m 2 ), Shannon index (H’), and Pielou evenness index<br />
(E = H’/Hmax H ) were calculated for every garden (Magurran, 1988). To compare<br />
floristic similarity between the three villages, Sørensen’s coefficient was computed<br />
(Magurran, 1988). Data were analyzed using the statistical package SPSS 11.0.
343<br />
Differences between means were determined by Mann-Whitney U-Test U or Kruskal<br />
Wallis H-Test. Changes over time as well as spatial differences of soil fertility<br />
parameters between production zones within one garden were analyzed as ‘paired<br />
samples’ using the Wilcoxon-test. Influence of relevant factors on crop diversity was<br />
determined by correlation analysis (Spearman).<br />
3. RESULTS<br />
In 2003/2004, size of the homegardens ranged from 240 to 2400 m 2 , and they had<br />
been established 4 to 41 years ago. <strong>Homegardens</strong> in the migrant village were<br />
significantly younger than those in the market village, and were managed by<br />
younger families (Table 2). In all three villages, homegarden size, farm size, and<br />
homegarden proportion in relation to the overall farm size were highly variable.<br />
Compared to 2001 (Kehlenbeck and Maass, 2004), farm size increased significantly<br />
only in the migrant village due to purchase or clearing of additional land. Therefore,<br />
the proportion of the homegarden in relation to overall farm size as well as its<br />
importance for staple food production recently decreased in the migrant village.<br />
Table 2. Characteristics of households and homegardens surveyed in three villages of the<br />
Napu Valley, Central Sulawesi, Indonesia, 2003.<br />
Parameters Market village<br />
(Wuasa)<br />
Forest village<br />
(Rompo)<br />
Migrant village<br />
(Siliwanga)<br />
Median Range Median Range Median Range<br />
Age of household<br />
head (years)<br />
55 a<br />
34 – 69<br />
ab<br />
50 25 – 89<br />
b<br />
35 30 – 50<br />
Gardener’s age<br />
(years)<br />
48 a<br />
32 – 67<br />
a<br />
40 20 – 60<br />
a<br />
34 28 – 50<br />
Household members 8<br />
(no.)<br />
a<br />
3 – 14<br />
a<br />
5 1– 11<br />
a<br />
4 3 – 6<br />
Farm size (ha) 2.6 a<br />
0.9 – 11.1<br />
a<br />
5.9 1.7 – 11.5<br />
a<br />
3.1 1.5 – 5.5<br />
Homegarden size<br />
(m 2 )<br />
720 a<br />
236 – 1134<br />
a<br />
610<br />
a<br />
287 – 1450 820 471 – 2383<br />
Homegarden size/<br />
farm size (%)<br />
2.2 a<br />
0.5 – 10.0<br />
a<br />
1.2 0.5 – 4.3<br />
a<br />
2.3 1.2 – 11.9<br />
Age of homegarden<br />
(years)<br />
28 a<br />
14 – 37<br />
ab<br />
16 4 – 41<br />
b<br />
10 6 – 11<br />
Medians in a row followed by different superscripts are significantly different at p≤0.05.<br />
3.1. Crop diversity and its changes<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
Crop species richness was high and increased markedly over time both per village<br />
and per garden (Fig. 1). In the three villages, a combined total of 149 and 168 crop<br />
species were identified in 2001 and 2003 respectively. Distribution of crops into<br />
different use categories was comparable in different sampling years (Kehlenbeck
344<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
and Maass, 2004). Out of the 168 crop species grown in homegardens, about 35<br />
were wild species (mainly used as fuelwood/timber or medicine) and about 44 were<br />
classified as underutilized species (mainly used as vegetable). In addition to the 168<br />
crop species, 99 ornamental and 62 weed species were found in the homegardens<br />
surveyed in 2003.<br />
Figure 1. Changes of total and mean crop species richness in homegardens per village,<br />
studied in the Napu Valley, Central Sulawesi, Indonesia, 2001 (n = 30) and 2003 (n = 29).<br />
Figure 2. Changes of mean species density in homegardens of three villages studied in the<br />
Napu Valley, Central Sulawesi, Indonesia, 2001 and 2003.<br />
Mean species density increased significantly over time, particularly in the market<br />
village (Fig. 2). However, in the migrant village, species density continued to be<br />
significantly lower than that in the forest village in 2001 or the market village in<br />
2003. Changes in Shannon diversity and Pielou evenness indices were not so clear<br />
apart from the migrant village, where both indices were significantly higher in 2003<br />
than in 2001 (Fig. 3). In the market village, Shannon and Pielou indices showed a<br />
slight tendency to decrease because in 2003 some gardeners started to grow spring
345<br />
onion (Allium ( fistulosum)<br />
for sale in relatively large plots, which dramatically<br />
reduced the indices. For example, in one homegarden an area of about 190 m 2 out of<br />
865 m 2 was planted with a mixture of vegetables and spices in 2001, but only spring<br />
onion during 2003. This resulted in a decrease of Shannon index from 2.1 to 1.2 and<br />
Pielou index from 0.59 to 0.31. However, the total number of crop species increased<br />
in this particular garden from 35 to 47 during the same period.<br />
Figure 3. Changes of mean Shannon diversity and mean Pielou evenness indices in<br />
homegardens of three villages studied in the Napu Valley, Central Sulawesi, Indonesia, 2001<br />
and 2003.<br />
Crop species composition was clearly different among the three villages in both<br />
years. Sørensen’s coefficients showed a higher similarity between the market and<br />
the forest villages (0.71) than between these two and the migrant village (market vs.<br />
migrant village: 0.63; forest vs. migrant village: 0.58). Compared to 2001<br />
(Kehlenbeck and Maass, 2004), Sørensen’s coefficient decreased slightly in all<br />
cases. The species common to all three villages remained rather stable over time,<br />
while obvious changes occurred in those crop species unique for one village and,<br />
hence, not found in the other two. Particularly in the market village, 22 unique crops<br />
were recorded in 2003 instead of 15 in 2001.<br />
3.2. Soil properties<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
In large gardens with different production zones, soil fertility was obviously<br />
different among these zones. Across all 12 gardens where distinct vegetable and<br />
cacao zones existed, soil of the vegetable zone contained significantly less N and C<br />
than soil of the adjacent cacao zone (Table 3). Soil pH did not differ among<br />
vegetable and cacao production zones. Bulk density was significantly higher in<br />
vegetable zones than in adjacent cacao zones.<br />
Because of these large differences in soil fertility between production zones<br />
within one single homegarden, it did not appear meaningful to compare mean values<br />
of the homegardens investigated in the three villages. Instead, soil fertility of cacao
346<br />
zones only was compared among the villages as this particular zone existed in most<br />
of the homegardens (n = 16), apart from the very small ones. In five homegardens,<br />
the cacao zone was even the only obvious production zone.<br />
Table 3. Properties of homegarden topsoil (0–15 cm) from different production zones in three<br />
villages of the Napu Valley, Central Sulawesi, Indonesia, 2003/2004.<br />
Soil attributes Vegetable zone Cacao zone<br />
Mean Range Mean Range<br />
Ctotal (%) 1.64 b<br />
0.93 – 2.96 2.31 a<br />
Ntotal (%) 0.13<br />
1.40 – 3.42<br />
b<br />
0.06 – 0.21 0.18 a<br />
pH (H2O) 5.87<br />
0.10 – 0.27<br />
a<br />
4.65 – 6.88 5.62 a<br />
Bulk density (g/cm<br />
5.24 – 5.83<br />
3 ) 1.17 a<br />
0.90 – 1.48 1.03 b<br />
0.77 – 1.17<br />
Means in a row followed by different superscripts are significantly different at p ≤ 0.05.<br />
Table 4. Properties of topsoil (0–15 cm) from cacao production zones of 21 homegardens in<br />
three villages of the Napu Valley, Central Sulawesi, Indonesia, 2003/2004.<br />
Soil attributes Market village<br />
(Wuasa; n = 8)<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
Forest village<br />
(Rompo; n = 7)<br />
Migrant village<br />
(Siliwanga; n = 6)<br />
Mean Range Mean Range Mean Range<br />
C total (%) 2.02 a 1.43 – 2.95 2.31 a 1.40 – 3.21 2.83 a N total (%) 0.17<br />
2.32 – 3.42<br />
a 0.12 – 0.21 0.19 a 0.10 – 0.27 0.19 a pH (H2O) 5.65<br />
0.15 – 0.24<br />
a 5.24 – 5.84 5.48 a 5.16 – 5.80 5.63 a Bulk density<br />
(g/cm<br />
5.21 – 5.85<br />
3 )<br />
1.10 ab 0.92 – 1.24 0.98 b 0.77 – 1.16 1.16 a 1.08 – 1.24<br />
Means in a row followed by different superscripts are significantly different at p ≤ 0.05.<br />
Table 5. Changes of soil fertility parameters of topsoil (0–15 cm) from 14 homegardens in<br />
three villages of the Napu Valley, Central Sulawesi, Indonesia, 2001 and 2003.<br />
Soil attributes 2001 2003<br />
Mean Range Mean Range<br />
C total (%) 2.35 a 1.20 – 3.58 2.12 b 0.92 – 3.21<br />
N total (%) 0.19 a 0.11 – 0.29 0.16 b 0.07 – 0.27<br />
pH (H2O) 5.72 a 4.70 – 6.50 5.75 a 4.88 – 6.75<br />
Means in a row followed by different superscripts are significantly different at p ≤ 0.05.<br />
Soil C and N contents of cacao production zones were highly variable in all<br />
villages, although these values were slightly lower in the market village (Table 4).<br />
Soil pH was relatively similar in all villages. Only soil bulk density was significantly<br />
higher in the migrant village and lower in the forest village. When comparing soil
fertility over time, C and N contents decreased significantly from 2001 to 2003,<br />
whereas soil pH did not change (Table 5).<br />
3.3. Influence of selected factors on crop diversity<br />
347<br />
To detect factors that possibly influence crop diversity, correlations between crop<br />
diversity parameters and several variables describing characteristics of homegardens<br />
(e.g., age, size, soil fertility parameters), the gardener (e.g., age, education), or<br />
socioeconomics (e.g., wealth status of household, size of paddy rice fields, market<br />
access) were analyzed (Table 6). Socioeconomic characteristics of the gardeners or<br />
households did not play an important role in determining crop diversity.<br />
Table 6. Spearman correlation coefficients between crop diversity parameters and different<br />
characteristics of homegardens, gardeners, and households in three villages of the Napu<br />
Valley, Central Sulawesi, Indonesia, 2001 and 2003.<br />
Parameters Species<br />
richness<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
Species density Shannon<br />
index<br />
Pielou index<br />
2001 2003 2001 2003 2001 2003 2001 2003<br />
Garden age 0.45* 0.41* ns ns ns ns ns ns<br />
Garden size 0.45* 0.52** –0.83*** –0.81*** ns ns ns ns<br />
Soil pH value ns –0.40* 0.43* 0.40* ns ns ns ns<br />
Soil N content ns ns –0.50** –0.38* ns ns ns ns<br />
Soil C content ns ns –0.58** ns –0.43* ns –0.42* ns<br />
Gardener’s age 0.47** 0.49** ns ns 0.53** ns 0.41* ns<br />
Gardener’s<br />
education<br />
ns ns ns ns ns ns ns ns<br />
HH members<br />
(no.)<br />
ns ns ns ns ns ns ns ns<br />
Wealth status<br />
of HH<br />
ns ns ns ns ns ns ns ns<br />
Size of HH’s<br />
rice fields<br />
ns ns ns ns ns ns ns ns<br />
Garden<br />
size/farm size<br />
ns ns –0.68*** –0.43* ns ns –0.47* ns<br />
Market access ns ns ns ns ns ns ns ns<br />
*: p ≤ 0.05; **: p ≤ 0.01; ***: p ≤ 0.001; ns = not significant; HH = household.<br />
Crop diversity was mainly influenced by the gardener’s age and by variables<br />
describing homegarden characteristics such as size, age, or soil fertility parameters.<br />
In large and old homegardens, higher crop species richness could be expected than<br />
in small and young homegardens. Furthermore, the older the gardener, the higher<br />
was the species richness, diversity, and evenness. However, the influence of all<br />
variables was rather weak, particularly of soil parameters. Within the tested
348<br />
socioeconomic variables, only the ratio of homegarden-size to farm-size showed a<br />
weak but significant negative influence on evenness index. Ethnicity of the gardener<br />
probably was linked with crop diversity because mean species richness and density<br />
were significantly higher in gardens of local families than that of the migrants. No<br />
differences in crop diversity were observed by grouping gardeners into male and<br />
female subgroups. However, direct influence of these two nominal variables on crop<br />
diversity could not be assessed by the correlation analysis.<br />
4.1. Changes of crop diversity<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
4. DISCUSSION<br />
In 2001 and 2003 total crop species richness as well as the mean per garden were<br />
rather high, but comparable to the data reported from other regions in Indonesia or<br />
even from the tropics as a whole (Kumar and Nair, 2004). Crop diversity in the<br />
homegardens surveyed was not only maintained, but even increased over time.<br />
Seasonal effects could not be made responsible for this because in both years species<br />
inventories were carried out in the same season. Partly, the increase in diversity can<br />
be explained by interventions of development projects. For instance, in all villages,<br />
seedlings of mandarin trees (Citrus reticulata) were provided to most gardeners in<br />
2002/2003. Another project promoted the cultivation of medicinal plants in<br />
homegardens at the same time. As a result, the Mayor of the market village pushed<br />
gardeners to grow these recommended plants. This led to an increase of medicinal<br />
species from a total of 16 in 2001 to 21 in 2003 and a mean per garden from 3.4 to<br />
5.4, respectively. However, in the other two villages the impact of these<br />
development projects on homegarden diversity seemed to be rather low.<br />
Additionally, research activities in 2001 (Kehlenbeck and Maass, 2004) have<br />
possibly stimulated interest of the gardeners in crop diversity. As a result, gardeners<br />
might have revived the networks of seed and plant exchange within their<br />
neighborhoods, and were more open for experimental cultivation of new crops.<br />
At the same time, gardeners stopped to grow some crop species (a mean of six<br />
species per garden). According to the gardeners, many of these species died during<br />
an unusual dry period in 2002. Another reason for decrease of diversity in<br />
homegardens could be that production became more market-oriented, as described<br />
by Soemarwoto and Conway (1991). However, in this study, the market-oriented<br />
production of spring onions in the market village resulted only in a slight decrease of<br />
Shannon and evenness indices but not richness or density of crop species. Besides,<br />
in 2004 it was observed that the gardeners already stopped growing spring onions<br />
for sale due to a decline in prices and problems with diseases.<br />
It can, therefore, be concluded that crop diversity in homegardens is very<br />
dynamic and every species inventory reflects only the diversity at the very instant of<br />
assessment. Thus, the temporal dynamics observed in this study might not reflect<br />
long-term trends. Nevertheless, the suitability of homegardens for in situ<br />
conservation of plant genetic resources needs to be critically revised based on these<br />
results. For this purpose, specific target groups of crops or even key species instead
349<br />
of the overall diversity should be emphasized m (e.g., Watson and Eyzaguirre, 2002).<br />
Furthermore, it is crucial to make gardeners active stakeholders of such conservation<br />
efforts by sharing both responsibility and benefits. Finally, crop diversity should not<br />
be used as the only sustainability indicator of this system because of its changes with<br />
time.<br />
4.2. Soil fertility<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
According to Landon (1991), soil of vegetable and cacao zones surveyed had low to<br />
very low mean C and N contents, whereas mean pH values were classified as<br />
medium. Therefore, the current situation with limited N available in the soil most<br />
likely restricts the level of production, particularly for N-demanding vegetables.<br />
Considering the significant decrease in soil C and N contents over time, crop<br />
production may become more constrained in the near future, particularly in the<br />
market and forest villages, where C and N contents were already very low in many<br />
garden soils. Insufficient soil fertility management by the gardeners caused this<br />
alarming situation. For example, only about 30% of the gardeners used farmyard<br />
manure as a fertilizer, although it was available to all of them. Many gardeners<br />
removed weeds including their roots for burning or depositing in garbage pits<br />
instead of using them for compost preparation. Use of compost or mulch was<br />
virtually unknown, and industrial N fertilizer was available to only 15% of the<br />
gardeners, an overall situation that has not changed since 2001 (Kehlenbeck and<br />
Maass, 2004). Deterioration was accelerated also by the habit of gardeners to<br />
remove the litter layer by daily sweeping and regular burning. Typical reasons given<br />
by the gardeners for this practice were keeping away snakes and insects from the<br />
house. Sweeping and total weeding was carried out in all front gardens, in most<br />
vegetable and ornamental zones and in some cacao or fruit tree zones, which led to<br />
severe soil erosion (Fig. 4).<br />
In general, soil fertility is said to be maintained in homegardens in the long-term<br />
(Gajaseni and Gajaseni, 1999; Kumar and Nair, 2004). Only few reports<br />
(Soemarwoto, 1987; Soemarwoto and Conway, 1991; Hvoslef, 1994; Benjamin<br />
et al., 2001) stated problems of soil deterioration and erosion due to insufficient<br />
management practices similar to those identified here. Soil management in the<br />
present study, however, needs also to be seen in the context of changing traditional<br />
land use in the Napu Valley. The dominant shifting cultivation was replaced by<br />
permanent agriculture only about 10 to 30 years ago (Burkhard, 2002). Therefore,<br />
indigenous as well as newly arrived migrant farmers may not be familiar with<br />
appropriate sustainable land management practices. Negative environmental<br />
consequences have similarly been documented for other cases of resettlement, e.g.,<br />
in Ethiopia (Wood, 1993) and Tanzania (Charnley, 1997).<br />
Spread of household waste materials in homegardens might cause a new problem<br />
affecting long-term soil fertility and, consequently, system productivity. This has<br />
never been mentioned in the homegarden literature. Due to lack of opportunities for<br />
waste disposal, many gardeners in the research area spread all garbage on the soil of<br />
the backyard, including non-biodegradable items such as glass and plastic bottles,<br />
tins, plastic bags, and old batteries. Mixed with organic wastes from the kitchen, this
350<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
garbage formed the ‘litter’ layer in many backyard gardens. This practice will<br />
probably cause soil contamination; the spread of biodegradable waste on soil<br />
will, however, contribute to better nutrient cycling and reduced soil erosion.<br />
Figure 4. Example of soil erosion in the front yard of a homegarden in the forest village<br />
Rompo, Napu Valley, Central Sulawesi, Indonesia, 2004. The broken line indicates soil<br />
surface during planting of the ornamentals along the fence; the dotted line shows the present<br />
surface.<br />
To achieve sustainable soil fertility management in the study region, the existing<br />
extension service should not exclusively focus on paddy rice production but also on<br />
agroforestry systems (including homegardens) with their great significance for cash<br />
income generation (Maertens et al., 2002). Advantages of using compost, mulch,<br />
and farmyard manure should be explained. Growing N2-fixing cover crops ought to<br />
be promoted, not only in the homegardens, but also in other cropping systems.<br />
Besides, villagers should be enlightened about disadvantages and risks of soil and<br />
water contamination in order to preserve the resources on which they rely.
4.3. Factors influencing crop diversity<br />
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
351<br />
Within the major factors influencing crop diversity, garden size is one of the<br />
frequently analyzed. Among others, Abdoellah et al. (2001) and Gutiérrez et al.<br />
(2004) reported a positive relationship between garden size and crop species<br />
richness. Results from the present study (Table 6) showed a slightly positive, but<br />
non-linear relationship. In very large gardens, crop species richness tended to reach<br />
a plateau. On the other hand, the larger the garden the lower was crop species<br />
density because of more uniform planting patterns in very large gardens. A positive<br />
influence of garden age on species richness was also stated by Gutiérrez et al.<br />
(2004). In this study, however, garden age had a highly significant positive<br />
correlation with gardener’s age because, generally, young families establish a new<br />
homegarden, starting with a rather small set of crop species.<br />
Besides age and size of homegarden, soil fertility is another factor describing<br />
garden features, but its influence on crop diversity has not yet been studied in detail.<br />
Hodel et al. (1999) assumed an influence of soil factors on diversity without<br />
quantifying this. In forest gardens, Kaya et al. (2002) reported lower species<br />
diversity on marginal soils compared to soils that are more fertile. Many crop<br />
species, particularly vegetables and spices, do not give adequate yield under<br />
unfavorable soil conditions. Therefore, gardeners stop cultivating these species<br />
while switching to a reduced set of crops that can cope with low soil fertility. In the<br />
migrant village Siliwanga with its rather poor soil conditions, for example, acidtolerant<br />
species such as tea (Camellia sinensis), cassava (Manihot esculenta) and<br />
cashew ( (Anacardium occidentale)<br />
were found in many homegardens, whereas<br />
vegetable cultivation was rare (Kehlenbeck and Maass, 2004). However, influence<br />
of soil fertility parameters on crop diversity in this study must be seen in the context<br />
of the significant correlations between garden size and soil pH (negative) as well as<br />
between garden size and soil C and N contents (positive) that probably biased the<br />
results of analysis (see Table 6).<br />
Gardener’s age can influence crop diversity positively, possibly because, over<br />
the years, gardeners try to cultivate new crops while they continue to plant well-tried<br />
species (Gutiérrez et al., 2004). Besides, older gardeners often have more time for<br />
homegardening and are supported by their grown-up children. A higher timeallocation<br />
to homegardening leads to a higher diversity of useful plants (Hodel et al.,<br />
1999; Gutiérrez et al., 2004). In the present study, however, the positive relationship<br />
between gardener’s age and plant diversity was rather weak.<br />
Within gardener’s characteristics, ethnicity also may be a factor explaining<br />
variation in crop diversity (Soemarwoto and Conway, 1991; Hodel et al., 1999).<br />
Contrary to the findings of Soemarwoto and Conway (1991), crop diversity in the<br />
present study was lower in homegardens of migrants as compared to locals.<br />
Admittedly, migrant gardeners brought various useful species from their home<br />
regions. Due to the unfavorable soil and climate conditions of the lands assigned to<br />
them, a large part of these plants did not establish. Another reason for the low crop<br />
diversity in the migrant village might be the socioeconomic status of the gardeners.<br />
After arrival, young migrant families focused strongly on paddy rice production,<br />
with a resulting shortage of labor for homegarden management. Furthermore, field
352<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
crop failures and poor access to suitable agricultural land might have led to<br />
cultivation of additional staple crops in the migrants’ homegardens. The result of<br />
correlation analysis that a high portion of homegarden size to overall farm size was<br />
related with a low Pielou evenness index (Table 6) support this statement. A<br />
reduction in diversity of homegardens is known to be caused by a high proportion of<br />
staple food crops (Soemarwoto and Conway, 1991) as well as by labor shortage<br />
(Hodel et al., 1999; Gutiérrez et al., 2004).<br />
Among socioeconomic factors, the negative influence of market proximity and<br />
commercially oriented production on crop diversity has frequently been recorded<br />
(Christanty, 1990; Soemarwoto and Conway, 1991; Michon and Mary, 1994;<br />
Abdoellah et al., 2001; Gutiérrez et al., 2004). In the study area, however, this effect<br />
was only slightly recognized (Kehlenbeck and Maass, 2004). Nevertheless, there<br />
seemed to be a high risk of decreasing crop diversity with an associated loss of plant<br />
genetic resources, if production of cash crops such as spring onions were to be<br />
successful. In summary, our results suggest that diversity is not only influenced by<br />
clearly identifiable single factors but rather by a complex interaction among several<br />
factors studied and probably others. This interaction is not yet understood, and<br />
additional intrinsic characteristics of gardeners, such as individual preferences and<br />
practices might play an overriding role. Obviously, further research is needed for a<br />
better understanding of these interrelationships and the processes leading to them.<br />
This would help assessing the sustainability of the system as well as its suitability<br />
for in situ conservation of plant genetic resources.<br />
5. CONCLUSIONS<br />
Although the homegardens surveyed maintained high crop diversity over time, their<br />
management at present in the study region was not conducive to sustainability in<br />
terms of soil fertility management. The set of the two common sustainability<br />
indicators chosen was found to be adequate only for a temporary assessment of<br />
homegardens. Nevertheless, an estimation of soil erosion as an additional indicator<br />
of sustainability should be considered, particularly where soil fertility monitoring is<br />
not practicable over time. <strong>Homegardens</strong> can play an important role in in situ<br />
conservation of plant genetic resources as long as gardeners participate in the whole<br />
process.<br />
ACKNOWLEDGEMENTS<br />
Many thanks to all the people in Wuasa, Rompo and Siliwanga, especially to the 30<br />
respondent families, for their <strong>coop</strong>eration and ready help during the field survey.<br />
The support provided by members and staff of STORMA (SFB 552) in Bogor, Palu<br />
and Göttingen is also gratefully acknowledged, particularly the subproject<br />
‘Economic Analysis of Land Use Systems of Rural Households’ (A4) for making<br />
available their survey data set and subproject ‘Central Laboratory Unit’ (Z3) for<br />
carrying out soil analyses. The German Academic Exchange Service (DAAD) and<br />
German Science Foundation (DFG) provided financial support.
ARE TROPICAL T HOMEGARDENS SUSTAINABLE?<br />
REFERENCES<br />
353<br />
Abdoellah O.S., Takeuchi K., Parikesit, Gunawan B. and Hadikusumah H.Y. 2001. Structure<br />
and function of homegarden: a revisited. Proc. Seminar ‘Toward harmonisation between<br />
development and environmental conservation in biological production’ (21-23 February<br />
2001), pp 167 – 185. University of Tokyo, Tokyo.<br />
Benjamin T.J., Montañez P.I., Jiménez J.J.M. and Gillespie A.R. 2001. Carbon, water and<br />
nutrient flux in Maya homegardens in the Yucatán peninsula of México. Agroforest Syst<br />
53: 103 – 111.<br />
Burkhard G. 2002. Natural resource management in Central Sulawesi: Past experience and<br />
future prospects. STORMA Discussion Paper Series No. 8 (online). Universities of<br />
Goettingen and Kassel, Germany, Institut Pertanian Bogor and Universitas Tadulako,<br />
Indonesia. http://www.storma.de /DPS/pdf/SDP8.pdf (last accessed: July 29, 2004).<br />
Charnley S. 1997. Environmentally displaced peoples and the cascade effect: Lessons from<br />
Tanzania. Hum Ecol 25: 593 – 618.<br />
Christanty L. 1990. Home gardens in tropical Asia, with special reference to Indonesia. In:<br />
Landauer K. and Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 9 – 20. The United Nations<br />
University, Tokyo.<br />
Gajaseni J. and Gajaseni N. 1999. Ecological rationalities of the traditional homegarden<br />
system in the Chao Phraya Basin, Thailand. Agroforest Syst 46: 3 – 23.<br />
Gutiérrez M., Quiróz C., Pérez D., Rodríguez D., Pérez T., Marques A. and Pacheco W. 2004.<br />
Conservación in situ de diversas especies vegetales en ‘conucos’ (home gardens) en los<br />
estados Carabobo y Trujillo de Venezuela. Plant Gen Resour Newsl 137: 1 – 8.<br />
Hodel U., Gessler M., Cai H.H., Thoan V.V., Ha N.V., Thu N.X. and Ba T. 1999. In situ<br />
Conservation of plant genetic resources in home gardens of Southern Vietnam.<br />
International Plant Genetic Resources Institute, Rome, 106p.<br />
Hvoslef H. 1994. <strong>Homegardens</strong> of Javanese transmigrants in Seberida subdistrict:<br />
Description, agroecological constraints and evaluation of potential solutions to declining<br />
productivity. In: Sandbukt O. and Wiriadinata H. (eds), Rain forest and resource<br />
management. Proceedings NORINDRA seminar, pp 127 – 136. Indonesian Institute of<br />
Sciences (LIPI), Jakarta.<br />
Huxley P.A. 1999. <strong>Tropical</strong> agroforestry. Blackwell Science, Oxford, 371p.<br />
Jensen M. 1993. Soil conditions, vegetation structure and biomass of a Javanese homegarden.<br />
Agroforest Syst 24: 171 – 186.<br />
Karyono 1990. Home gardens in Java. Their structure and function. In: Landauer K. and<br />
Brazil M. (eds), <strong>Tropical</strong> home gardens, pp 138 – 146. The United Nations University,<br />
Tokyo.<br />
Kaya M., Kammesheidt L. and Weidelt H.-J. 2002. The forestt garden system of Saparua<br />
island, Central Maluku, Indonesia, and its role in maintaining tree species diversity.<br />
Agroforest Syst 54: 225 – 234.<br />
Kehlenbeck K. and Maass B.L. 2004. Crop diversity and classification of homegardens in<br />
Central Sulawesi, Indonesia. Agroforest Syst 63: 53 – 62.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Landauer K. and Brazil M. (eds). 1990. <strong>Tropical</strong> home gardens. The United Nations<br />
University, Tokyo, 257p.
354<br />
K. KEHLENBECK K AND B.L. MAASS M<br />
Landon J.R. (ed.). 1991. Booker tropical soil manual: A handbook for soil survey and<br />
agricultural land evaluation in the tropics and subtropics. Longman Scientific &<br />
Technical, Essex, 474p.<br />
Maertens M., Zeller M. and Birner R. 2002. Explaining agricultural land use in villages<br />
surrounding the Lore Lindu National Park in Central Sulawesi, Indonesia. STORMA<br />
Discussion Paper Series No. 4 (online). Universities of Goettingen/Kassel, Germany;<br />
Institut Pertanian Bogor/Universitas Tadulako, Indonesia. http://www.storma.de/DPS/<br />
index. htm (last accessed: July 27, 2004).<br />
Magurran A.E. 1988. Ecological diversity and its measurement. Croom Helm, London, 179p.<br />
Michon G. and Mary F. 1994. Conversion of traditional village gardens and new economic<br />
strategies of rural households in the area of Bogor, Indonesia. Agroforest Syst 25:<br />
31 – 58.<br />
Soemarwoto O. 1987. <strong>Homegardens</strong>: A traditional agroforestry system with a promising<br />
future. In: Steppler H.A. and Nair P.K.R. (eds), Agroforestry: A decade of development.<br />
pp 157 – 170. ICRAF, Nairobi.<br />
Soemarwoto O. and Conway G.R. 1991. The Javanese homegarden. J Farm Syst Res-Ext 2:<br />
95 – 118.<br />
Torquebiau E. 1992. Are tropical agroforestry home gardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Watson J.W. and Eyzaguirre P.B. (eds). 2002. Home gardens and in situ conservation of plant<br />
genetic resources in farming systems. Proceedings of the second international home<br />
gardens workshop (17 – 19 July 2001), Witzenhausen, Germany. International Plant<br />
Genetic Resources Institute, Rome, 184p.<br />
Wezel A. and Bender S. 2003. Plant species diversity of homegardens of Cuba and its<br />
significance for household food supply. Agroforest Syst 57: 39 – 49.<br />
Whitten A.J., Mustafa M. and Henderson G.S. 1987. The Ecology of Sulawesi. Gadjah Mada<br />
University Press, Yogyakarta, 777p.<br />
Wood A.P. 1993. Natural resource conflicts in south-west Ethiopia: State, communities, and<br />
the role of the National Resource Strategy in the search for sustainable development. Nord<br />
J Afr Stud 2: 83 – 99.<br />
Zeller M., Schwarze S. and van Rheenen T. 2001. Statistical sampling frame and methods<br />
used for the selection of villages and households in the scope of the research program on<br />
stability of rainforest margins in Indonesia (STORMA). STORMA Discussion Paper<br />
Series No. 1 (online). Universities of Goettingen and Kassel, Germany, Institut Pertanian<br />
Bogor and Universitas Tadulako, Indonesia. http://www.storma.de/DPS/pdf/SDP1.pdf<br />
(last accessed: January 20, 2003).
CHAPTER 20<br />
WHITHER HOMEGARDENS?<br />
P.K.R. NAIR<br />
School of Forest Resources and Conservation, University of Florida, Gainesville,<br />
Florida 32611, USA; E-mail: <br />
Keywords: Commercialization, Species diversity, Sustainability, Urban homegardens.<br />
Abstract. Although homegardens provide sustenance to millions of households in the tropics,<br />
their underlying scientific foundations have not been fully explored, and therefore they are not<br />
a part of development agendas. While their integrated and complex nature are a challenge to<br />
scientific investigations that are often compartmentalized, these very same attributes form the<br />
bases of the ecological, economic, and social sustainability of homegardens. In the wake of<br />
recent trend towards commercialization and consequent conversion of homegardens to<br />
produce market-oriented crops, concerns have been raised about the future of traditional<br />
homegardens. Lack of rigorous scientific evidence makes it difficult to make predictions.<br />
Nevertheless, experiences about the role and value of homegardens from around the world<br />
suggest that homegardens are not on the path to extinction. They will continue to be an<br />
essential part of the way of life, but their nature and functions will change in tune with the<br />
rapid changes happening all over. The concept of homegardens will increasingly be adopted<br />
in urban and periurban areas, not only in the tropics, but also in industrialized societies,<br />
reflecting the society’s increasing appreciation of traditional values and ecosystem functions.<br />
1. INTRODUCTION<br />
“… that whoever could make two ears of corn, or two blades of grass, to grow on a spot<br />
of ground where one grew before, would deserve better of mankind and do more<br />
essential service to his country …”<br />
Jonathan Swift<br />
The above quote that I included at the beginning of my first book nearly three<br />
decades ago (Nair, 1979) is as apt now as it was then. The subject matter of that<br />
book “Intensive Multiple Cropping with Coconuts in India,” written before the<br />
355<br />
B.M. Kumar and P.K.R. Nair (eds.), <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example of<br />
Sustainable Agroforestry, 355–370.<br />
© 2006 Springer. Printed in the Netherlands.
356<br />
P.K.R. NAIR N<br />
advent – or just at the beginning – of “modern” agroforestry, is not very different<br />
from the subject matter of this book, i.e., homegardens: multiple cropping with<br />
coconuts (Cocos nucifera) and other tree crops, now commonly referred to as<br />
multistrata agroforestry, is a distinguishing feature of (most) tropical homegardens.<br />
What Jonathan Swift envisioned in making two ears of corn, or two blades of grass,<br />
to grow on a spot of ground where one grew before is exactly what homegardeners<br />
have been practicing, especially in the warmer biomes, for centuries, i.e., growing an<br />
array of herbaceous species, shrubs, vines, and trees, all in intimate association on<br />
the same piece of land around their homes. Yet, these magnificent farming practices<br />
and intriguing plant associations are seldom recognized as worthy of consideration<br />
in development paradigms and ecological studies, nor are their practitioners treated<br />
as “… better of mankind doing more essential service to their countries …”<br />
In spite of this apparent neglect of homegardens and homegardeners, the reasons<br />
for which have been discussed in several previous writings (Nair, 2001; Kumar and<br />
Nair, 2004), the appeal, relevance, and lessons to be learned from this time-tested<br />
practice are so overwhelming and fascinating that time and again it attracts the<br />
attention of some researchers. For example, publications on homegardens can be<br />
found in almost all volumes of Agroforestry Systems. While some of them are at best<br />
scientific descriptions of a set pattern (characteristics of systems at specific<br />
locations), some deal with examining homegardens in the context of current trends<br />
and issues in land use systems, such as environmental integrity, carbon<br />
sequestration, biodiversity conservation, economic valuation of intangible benefits,<br />
and social equity, to name a few. Only very few of these are scientific analyses,<br />
however. Nevertheless, all such publications – old and new – on homegardens have<br />
had only “good things” to say about the practice: irrespective of its focus – be it C<br />
sequestration, biodiversity, soil fertility, or whatever – the study will have the<br />
inevitable conclusion that homegardens are “great” on that score.<br />
Other than these occasional researcher-motivated efforts – and, of course, the<br />
incessant individual efforts of the homegardeners – there has been no organized<br />
institutional initiative to promote homegardens either locally anywhere or<br />
internationally. That is hard to understand: if homegardens have all these desirable<br />
characteristics, why have they not earned a rightful place as a development vehicle?<br />
If homegardens are the “epitome of sustainability” (Torquebiau, 1992), how is it that<br />
they “defy” scientific explanation, or is it that homegardens are just a “backyard”<br />
activity with little prospects as a development tool and therefore not worthy of any<br />
serious scientific investigation? No answer has yet been found to the question that<br />
was posed five years ago: “Do homegardens defy science or is it the other way<br />
around?” (Nair, 2001). In the meanwhile, commercialization seems to make its way<br />
to homegardens that have traditionally been known as anything but commercial.<br />
Two chapters in this book report the recent tendency for growing crops in<br />
homegardens mainly for commercial use, in Java, Indonesia (Abdoellah et al., 2006)<br />
and Kerala, India (Peyre et al., 2006), the two best-known bastions of traditional<br />
homegardens. Is this an indication of the heralding of a new genre of homegardens<br />
and possibly the demise of the traditional ones? Is such an “evolution” of<br />
homegardens good or bad? In other words, what does the future hold for<br />
homegardens?
WHITHER W HOMEGARDENS?<br />
357<br />
In order to address the above key question, we need to discuss why homegardens<br />
(especially their species diversity) have traditionally been important to the<br />
households and what the relevance is of the much-acclaimed sustainability attributes<br />
of homegardens to the current context and future prospects.<br />
2. SPECIES DIVERSITY IN HOMEGARDENS AND HOUSEHOLD FOOD<br />
SECURITY<br />
The most distinguishing and possibly important characteristic of all homegardens is<br />
their species diversity: the intimate admixture of plants of all types – herbs, shrubs,<br />
vines, trees, other perennials, and so on – on the same small parcel of land (Fig. 1).<br />
From the homegardener’s point of view, the primary objective of growing all these<br />
plants together is to produce food, often as a supplementary source. In order to<br />
appreciate the role of these plants grown in apparent disarray, we have to first of all<br />
recognize the fact that ‘he’, the traditional homegarden practitioner, is a ‘she’:<br />
Figure 1. A “typical” rural homegarden in Kerala, India, showing a large number of<br />
economic species in intimate association around the home (Photo: B. Mohan Kumar).<br />
women have primary responsibilities, or are as involved as men, for homegarden<br />
maintenance. This is common wherever homegardening is practiced. Considering
358<br />
P.K.R. NAIR N<br />
that it is primarily the woman’s responsibility in many societies to feed the families,<br />
it is perhaps a combination of both inspiration and desperation that prompt them to<br />
grow food around their homesteads: inspiration from experience and innovative<br />
instinct, and desperation from the lack of other avenues for finding food for the<br />
family. Species diversity in these systems may be a consequence of the interplay of<br />
these forces of inspiration and desperation. Mixing annual food crops with<br />
frequently harvestable tree crops that provide food and sometimes cash income to<br />
the family represents a confluence of human ingenuity with ecological ambience,<br />
such that the opportunity offered by year-round growing seasons and the<br />
amenability of the various species to grow in mixed stands makes it a “win – win”<br />
situation. Tracing the historical development of homegardens, Wiersum (2006)<br />
observes that in the most widely studied homegarden systems in South- and<br />
Southeast Asia, homegardens are used to produce products with high nutritional<br />
va1ue (proteins, vitamins, minera1s), medicina1 plants and spices, firewood, and<br />
sometimes a1so forage crops and construction wood, and homegardening is always<br />
combined with field-crop cultivation often in the form of wetland rice (Oryza sativa)<br />
in South- and Southeast Asia. These regions with good farming conditions and high<br />
population densities contributed to optimal development of the complementary<br />
system of staple food cultivation in open fields and supplementary diversified<br />
homegarden production for the family’s self-sufficiency and trade.<br />
Whatever be the reason for species diversity, and irrespective of whether it will<br />
continue to be a conspicuous feature of future homegardens in the wake of the push<br />
to commercialization, researchers seem to be quite obsessed (perhaps more than the<br />
practitioners) with species diversity of homegardens. Cataloging of species lists is<br />
such a common feature of most homegarden literature to the extent that many<br />
authors believe that a paper on any aspect of homegarden is incomplete without a<br />
species list (Nair and Kumar, 2006). An interesting point that comes out of such<br />
species lists is that, irrespective of the geographical focus of the study, the species<br />
that dominate such lists are the same from similar ecological regions. This is evident<br />
from the species listed in four chapters of this book, summarized in Table 1, from<br />
homegardens in Kerala, India (Mohan et al., 2006); Peruvian Amazon (Wezel and<br />
Ohl, 2006); and two locations in the Pacific islands (Lamanda et al., 2006; and<br />
Thaman et al., 2006). The situation may not be different if the study is extended to<br />
all the 135 case studies included in Fig.1 of Nair and Kumar (2006), with the<br />
exception that in some locations, the locally important species that are not common<br />
outside their limited geographical areas of distribution will be common in<br />
homegardens as well. Examples of this category include the peach palm (Bactris<br />
gasipaes) and various other palm species in Central and South America, fruit trees<br />
such as durian (Durio zibethynus) in Southeast Asia and breadfruit (Artocarpus (<br />
altilis) in the Pacific islands, and various fruit trees in West Africa (Cola spp.,<br />
Dacroydes edulis, Pterocarpus spp., Treculia africana: Okafor and Fernandes,<br />
1987). Similarly, in the tropical highlands, the dominant species in homegardens<br />
will be different from those in tropical lowlands (e.g., Fernandes et al., 1984; and<br />
Soini, 2005; for the Chagga homegardens of Tanzania and Tesfaye Abebe et al.,<br />
2006, for the homegardens of Ethiopian highlands).
359<br />
The bottom line is, dominant food crops, both herbaceous and woody, that are<br />
locally adapted have been the dominant species of homegardens in different<br />
ecological regions. The easy access to these crops in the backyard and the<br />
opportunity offered by many of them for staggered harvesting as needed (e.g., tuber<br />
crops, vegetables, plantain) make them quite attractive to the women who take it on<br />
themselves as their obligation and responsibility to find food for the family.<br />
Nutritional security (rather than food security) of the homegarden is another<br />
important benefit of homegardens. It is well known that several of the tree fruits in<br />
the gardens (Table 1) are nutritionally richer than the common, carbohydrate-rich<br />
grain crops, and are indeed the main sources of vitamins and minerals to the family<br />
(Niñez, 1984; Okafor and Fernandes, 1987; Kumar and Nair, 2004; Nair, 2006). The<br />
cash-income opportunity offered by saleable products (especially tree products)<br />
from the homegardens make it an attractive proposition for men too. Social and<br />
cultural value of the species in the homegardens is yet another important factor to be<br />
considered (discussed later). Species diversity of homegardens is thus quite an<br />
appealing feature to the homegardeners for a variety of reasons, and has been a<br />
major driving force in the maintenance of the gardens over centuries.<br />
Table 1. Commonly reported plants in homegardens of humid tropical lowlands.<br />
Category Species in homegardens<br />
Root and<br />
tuber crops<br />
Other food<br />
crops<br />
Fruit and nut<br />
yielding<br />
perennials<br />
Spices, Social<br />
beverages,<br />
and<br />
stimulants<br />
WHITHER W HOMEGARDENS?<br />
Colocasia esculenta (taro), Dioscorea alata (greater yam),<br />
Dioscorea esculenta (sweet yam), Ipomoea batatas (sweet potato),<br />
Manihot esculenta (cassava), Xanthosoma spp. (tannia or<br />
cocoyam)<br />
Ananas comosus (pineapple), Arachis hypogaea (peanuts), Cajanus<br />
cajan (pigeon pea), Passiflora edulis (passion fruit), Phaseolus,<br />
Psophocarpus and Vigna spp. (beans and other legumes),<br />
Saccharum officinarum (sugarcane), Zea mays (corn = maize), and<br />
various vegetables<br />
Anacardium occidentale (cahew nut), Annona spp. (soursop and<br />
sweetsop), Averrhoa carambola (carambola), Artocarpus<br />
heterophyllus (jackfruit), A. altilis (breadfruit), Carica papaya<br />
(papaya), Citrus spp. (lemon, lime, orange, tangerin), Cocos<br />
nucifera (coconut), Ficus spp. (edible figs), Mangifera indica<br />
(mango), Musa spp. (bananas and plantains), Persea americana<br />
(avocado), Psidium guajava (guava), Spondias dulcis (vi apple,<br />
hogplum), Syzygium malaccense (Malay apple), Tamarindus indica<br />
(tamarind)<br />
Areca catechu (betel nut), Cinnamomum zeylanicum (cinnamon),<br />
Curcuma longa (turmeric), Cymbopogon citratus (lemon grass),<br />
Piper betle (betel vine), Piper methysticum (kava), Zingiber<br />
officinale (ginger).
360<br />
P.K.R. NAIR N<br />
3. SUSTAINABILITY AND HOMEGARDENS<br />
Sustainability is perhaps the most widely discussed, yet least well-defined, term<br />
across disciplines in contemporary agricultural and land use literature. Even before<br />
publication of the much-acclaimed and so-called Brundlandt Commission report<br />
(WCED, 1987), sustainability has been a cornerstone of many traditional land use<br />
systems and it used to figure prominently in the early debates on agroforestry (Bene<br />
et al., 1977). Without going into any discussion on this much-discussed issue,<br />
suffice it to say that sustainability is about meeting today’s needs without<br />
compromising the ability of future generations to satisfy their needs; it is not a new<br />
concept, simply the retrieval of ancient wisdom dictating that “you don’t eat your<br />
seed corn”; and it strives to achieve a balance between ecological preservation,<br />
economic vitality, and social justice.<br />
Much of the discussion on ecological sustainability of homegardens is linked to<br />
their species diversity. While dealing with species of various forms, life cycle, and<br />
nature of products, the number or frequency of occurrence of a species in the<br />
homegarden is not a sufficient indicator of the importance or dominance of the<br />
species. Ecological parameters and indices that are commonly used to express<br />
population complexity and diversity such as Sorenson’s index of similarity,<br />
Shannon-Weiner and Margalef Indices of species diversity, and Importance Value<br />
Index, have lately been reported in homegarden studies (Kumar and Nair, 2004 – for<br />
literature until then; Mohan et al., 2006; Abdoellah et al., 2006; Kehlenbeck and<br />
Maass, 2006). Some authors have also used statistical procedures such as cluster<br />
analysis and correspondence analysis to group descriptive characteristics of<br />
homegardens, and to find out factors that may play a significant role in explaining<br />
patterns of floristic composition of the complex system; one such study is reported<br />
by Tesfaye Abebe et al. (2006) in this volume.<br />
The rationale is to use these indices as a basis for comparing homegardens with<br />
nearby natural vegetation – usually forests – on the assumption that in terms of<br />
species abundance and diversity, homegardens are in between natural systems and<br />
managed systems. <strong>Homegardens</strong> are perhaps the most diverse agroforestry practice,<br />
and among all agroforestry practices, they are at one end of the spectrum, twospecies<br />
(a tree and a crop) associations such as alleycropping being at the other end<br />
(Nair, 1993; Rao et al., 1988). Species abundance and diversity of homegardens<br />
should not, however, be equated with ecological succession that is characteristic of<br />
natural systems and the benefits of which are exploited in some traditional low-input<br />
agricultural systems such as shifting cultivation. The fact that natural systems are<br />
more diverse than agricultural systems has been known for long, one of the most<br />
widely cited articles on the subject being that of Odum (1969). In the very few<br />
examples of low-input agriculture that take advantage of the process of succession,<br />
the species are all carefully selected, but are not random successional species that<br />
seed-in naturally. In homegardens too, the species are selected carefully, and are<br />
therefore similar to such systems. <strong>Homegardens</strong> start off from one particular stage of<br />
the natural successional process, but keep natural succession from carrying the<br />
community to a so-called “climax” community. On the other hand, agroforestry<br />
practices such as alleycropping that are at the “other end” of the species-diversity
WHITHER W HOMEGARDENS?<br />
361<br />
spectrum have little similarity with the natural systems and do not fit into the realm<br />
of successional processes. Thus, in terms of complexity and species diversity,<br />
homegardens represent a unique set of ecological sustainability characteristics of<br />
natural systems as well as production benefits of agricultural systems. Another<br />
aspect of ecological sustainability in homegardens is the benefit of nutrient cycling<br />
experienced in multistrata systems, which is again a consequence of the species<br />
diversity (Nair et al., 1999).<br />
It needs to be pointed out in this context that the premise that diversity provides<br />
stability to ecosystems, which is the basis of the concept of ecological sustainability<br />
of homegardens, is being debated by ecologists: the so-called “diversity – stability<br />
debate” (e.g., McCann, 2000). Although the consensus of this debate as of now is<br />
that diversity can be expected, on average, to give rise to ecosystem stability,<br />
diversity is not the driver of this relationship; rather, ecosystem stability depends on<br />
the ability of communities to contain species, or functional groups, that are capable<br />
of differential responses. At present, in ecological studies, the role of keystone<br />
species is receiving increasing attention; this concept has hardly been used in<br />
homegarden studies yet, but seems to offer scope for further studying the diversity –<br />
stability issue in homegardens (see Tesfaye Abebe, 2006). If simplified communities<br />
are more vulnerable to invasion by other communities/species, then the trend<br />
towards commercialization of homegardens (discussed later) should result in higher<br />
frequency of invader species as well as pests and diseases in homegardens. The<br />
profit-oriented commercial homegarden enterprises will then resort to keeping such<br />
invading species under check through use of chemicals, which will inevitably<br />
disrupt the harmonious biodiversity and species associations (including microorganisms<br />
and species other than plants) that have been so characteristic of<br />
traditional homegardens.<br />
Economic and social sustainability attributes of homegardens are even less well<br />
studied than ecological-sustainability attributes. A common problem seen mentioned<br />
in most attempts to study economic benefits of homegardens is, again, lack of<br />
widely accepted procedures to measure economic benefits of intangible benefits and<br />
services. Alavalapati and Mercer (2004) described some procedures for economic<br />
valuation of agroforestry systems. Most attempts at economic valuation have two<br />
common features: first, they acknowledge the importance and need for “proper”<br />
evaluation of the intangible benefits of homegardens, such as aesthetics and<br />
ornamentation, nutritional security, food quality, and empowerment of women; then<br />
they highlight the difficulties involved in collecting realistic data and therefore<br />
caution about the error-prone nature of such analyses. The two chapters on<br />
economic analysis presented in this volume are no exception to this general trend:<br />
Torquebiau and Penot (2006) articulate the importance of including valuation of<br />
such benefits in homegarden evaluation, but stop short of suggesting any new<br />
procedures; and, Mohan et al. (2006), following a study applying conventional and<br />
some “non-conventional” economic procedures in some Kerala homegardens,<br />
confirm that the results are along expected lines and caution that their study<br />
procedure will need considerable “fine-tuning” to adapt to local conditions before it<br />
is applied elsewhere. Thus, economic sustainability of homegardens remains another
362<br />
attribute, the importance of which can only be felt qualitatively and intuitively, but<br />
is difficult to quantify.<br />
The same can be said about social sustainability. All social studies on<br />
homegardens exclaim the social attributes of homegardens, ranging from their role<br />
in ensuring gender equality and nutritional security to societal harmony and cultural<br />
heritage. Several chapters in this book touch upon these issues. Howard (2006)<br />
presents a well researched account of the major role of women in homegardens in<br />
Latin America: the presence of a garden rich in a variety of plants epitomizes the<br />
woman’s exertions on behalf of kin and her proficiency as primary provider of food,<br />
health, and overall well-being of the family, and demonstrates her freedom from<br />
dependence on products from neighbors and commercial vendors. Abdoellah et al.<br />
(2006) describe how the tendency towards conversion of homegardens to produce<br />
commercially valuable crops for market in Indonesia has disrupted the community’s<br />
equality, sharing, and harmonious living (rukun) that used to be built around<br />
traditional homegardens, and decreased the number of common grounds (buruan) in<br />
front of homes that serve as playground for children, and as a place for socializing<br />
with neighbors and for children to learn cultural and social values from their elders.<br />
The strength of these threads that are woven together in the fabric of social<br />
sustainability of homegardens cannot be expressed in quantitative terms.<br />
4. HOMEGARDENS AND SOME CURRENT LAND USE ISSUES<br />
4.1. Biodiversity<br />
P.K.R. NAIR N<br />
Biodiversity (short form for biological diversity) is often used as a synonym for<br />
species diversity. The importance of maintaining biodiversity in sustaining food<br />
production and protecting human and ecosystem health is now universally<br />
recognized, and land use systems that promote biodiversity are considered to be<br />
quite desirable from that perspective. A classification based on the production<br />
systems and species diversity ranked homegardens top with its highest biological<br />
diversity among all manmade agroecosystems (Swift and Anderson, 1993). Species<br />
richness and extent of biodiversity in homegardens depend, however, on ecological<br />
and socioeconomic factors and household preferences. Gajaseni and Gajaseni (1999)<br />
have reported, for example, the existence of non-commercial indigenous varieties of<br />
durian (Durio sp.) and rare varieties of mango (Mangifera indica) in homegardens<br />
of Thailand. Large numbers of cultivars of banana (Musa paradisiaca), coconut, and<br />
breadfruit have been reported in the homegardens of Micronesia (Falanruw, 1990;<br />
Thaman et al., 2006). Indeed, as already mentioned, most publications on<br />
homegardens from around the world (see Fig. 1: Nair and Kumar, 2006) report the<br />
large numbers of species present. The role of homegardens as repositories of plant<br />
biodiversity is thus indisputable. In a recent study from seven New- and Old-World<br />
tropical forest dynamic plots, Wills and 33 collaborators from 21 institutions around<br />
the world reported that an erosion of an ecological community's species diversity<br />
(that tends to happen as a result of stochastic extinction, competitive exclusion, and<br />
unstable host-enemy dynamics) can be prevented over the short-term through
363<br />
preferential introduction of rare species (Wills et al., 2006). They found that when<br />
species were rare in a local area, they had a higher survival rate than when they were<br />
common, resulting in enrichment for rare species and increasing diversity with age<br />
and size class in these complex ecosystems. Thus, it can be surmised that the<br />
preferential introduction of rare species such as medicinal plants (Rao and<br />
Rajeswara Rao, 2006) and fruit trees that homegardeners have been practicing for<br />
centuries around the world contributes to species biodiversity even if economic and<br />
social gains are the primary motivations for such introductions.<br />
4.2 Genetic-diversity conservation and species domestication<br />
In addition to the wide array of plants grown in homegardens for a variety of<br />
reasons, homegardens have high potential for in situ conservation of genetic<br />
resources (Watson and Eyzaguirre, 2002; McNeely, 2004; Schroth et al., 2004). An<br />
important issue, the significance of which is seldom recognized in the extant<br />
species-listing-dominated literature on homegardens, is the continuous interaction of<br />
homegardeners with these large groups of plants and the resultant contribution to<br />
species domestication. Simons and Leakey (2004) describe the deliberate selection<br />
and management of trees (domestication) by humans that has been going on for<br />
millennia in agroforestry systems. For example, Leakey et al. (2004) present<br />
evidence that subsistence farmers have domesticated locally popular indigenous<br />
fruits (Dacroydes edulis and Irvingia gabonensis) in Cameroon and Nigeria. It is<br />
reasonable to assume that much of this in situ domestication has taken place in<br />
homegardens. It is also likely that similar patterns of domestication have happened<br />
for other plant species in homegardens around the world, especially in those with<br />
long history as in South- and Southeast Asia (Wiersum, 2004).<br />
4.3. Carbon sequestration<br />
WHITHER W HOMEGARDENS?<br />
Most discussions on carbon sequestration potential of homegardens – and, indeed<br />
agroforestry systems in general – are based more on hypothetical considerations<br />
than empirical results. The argument is that these systems have high carbon storage<br />
(sequestration) potential in their multiple plant species, especially in woody<br />
perennial species, and soil; they help in conservation of C stocks in existing forests<br />
by alleviating the pressure on natural forests (Schroth et al., 2004); and, to some<br />
extent, in C substitution by reducing fossil-fuel burning through promotion of wood<br />
fuel production. Most reports indicate that the addition of a large proportion of the<br />
relatively high quantity of plant materials produced in a system will increase C stock<br />
in soils (Lal, 2004); therefore it is reasonable to surmise that homegardens will help<br />
substantially in C sequestration. All reports on C sequestration potential of<br />
homegardens (e.g., Montagnini and Nair, 2004; Kumar, 2006), however, are related<br />
to aboveground biomass. In the case of soils, C stored in surface soils has received<br />
some mention. But C exists in soils in labile (mobile) or recalcitrant (stable) form;<br />
the latter is more important for C sequestration; and, no study has been reported on<br />
this “real” form of C sequestration within soil profiles in homegardens. Most C
364<br />
sequestration reports also have disclaimers and caveats that lack of reliable<br />
inventories/estimates and uncertainties in the methods of estimation present serious<br />
difficulties. Thus, as in the case of other intangible and difficult-to-measure benefits<br />
and services, C sequestration benefit of homegardens remains one of the “potential<br />
benefits” that has not been even quantified, let alone exploited.<br />
5. NEW DIMENSIONS OF HOMEGARDENS<br />
5.1. Commercialization of homegardens<br />
Consequent to liberalizations in many formerly tightly controlled economies, agricultural<br />
enterprises, just as other production enterprises, are becoming increasingly subject to<br />
market pressures. A direct consequence of this is development and adoption of new<br />
strategies to promote commercialization of even traditional operations such as<br />
homegardens. Abdoellah et al. (2006) describe a case study of such a transformation<br />
in a West Java village in Indonesia, where some villagers, attracted by economic<br />
possibilities, have transformed their homegardens in such a way that they have<br />
become dominated by few plant species or are approaching even monocultures; the<br />
dominant species are cash crops such as vegetables that are in high demand in<br />
nearby urban markets. Similar examples are also prevalent in the Pacific islands as<br />
described by Thaman et al. (2006), where promotion of a wide range of export cash<br />
crops in rural areas has led to the clearing of diverse agroforests. Increasing trend<br />
towards commercialization has also been reported from Kerala homegardens<br />
(Kumar and Nair, 2004).<br />
This so-called commercialization is, however, not new to homegardens. It has<br />
been in existence to varying degrees in most well-known homegardens (of South<br />
and Southeast Asia). Perennial species that produce commercial products such as<br />
spices, fruits and nuts, medicinal plants, and even timber have been a component in<br />
many of these systems. As Kumar and Nair (2004) have pointed out, although<br />
interest in homegardens has been primarily focused on producing subsistence items,<br />
its role in generating additional cash income has been quite substantial in many<br />
places. Considerable variations from place to place have also been reported in the<br />
proportion of homegarden products that are used for household consumption as<br />
opposed to sale, and the contribution of the net income derived from sale of products<br />
to the total household income. Conversion of homegardens to intensive production<br />
units of market-oriented systems as described by Abdoellah et al. (2006) is not a<br />
totally new phenomenon; similar trends have occurred in several rapidly urbanizing<br />
and periurban centers. A case in point is the conversion of the traditional shamba<br />
gardens of Kenya’s highlands to produce vegetables for sale in Nairobi, the capital<br />
city, and for export to Europe (author’s personal experience).<br />
5.2. Urban homegardens<br />
P.K.R. NAIR N<br />
Another relatively new trend related to commercialization of homegardens is the<br />
extension of the homegarden practice from its conventional rural settings to urban
WHITHER W HOMEGARDENS?<br />
365<br />
environments. Two chapters in this book (Drescher et al., 2006; and Thaman et al.,<br />
2006) describe such developments; while the former includes examples from several<br />
places around the world representing both developing and developed countries, the<br />
latter deals primarily with such developments in the Hawaiian Islands, USA. These<br />
urban homegardens are often the “modern” cousins of their traditional relatives in<br />
the sense that while they maintain the species diversity that is characteristic of the<br />
traditional homegardens, their aesthetic and recreational value is as important as – if<br />
not more than – their nutritional role. As Fig. 2, a photograph of an urban<br />
homegarden in Kona, Hawaii, USA, shows, the gardens with manicured lawns and<br />
hedges, well tended fruit trees, and attractive ornamentals surrounding a “modern”<br />
home look more like tourist resorts, in sharp contrast to the “natural” look of the<br />
subsistence-oriented homegardens and the type of “traditional” homes they surround<br />
(Fig. 1).<br />
Figure 2. An urban homegarden with fruit trees such as avocado (Persea americana), litchi<br />
(Litchi chinensis), mango (Mangifera indica), papapya (Carica papaya, a and various<br />
ornamentals, in Kona, Hawaii, USA (Photo: Craig Elevitch).<br />
This trend towards urban homegardening may be seen in the context of other<br />
similar activities such as urban forestry and organic agriculture that have gained<br />
considerable prominence in urban and periurban areas during the recent past. These<br />
activities constitute a substantial portion of the green space and are considered to be<br />
the lungs of the cities. For example, the role of urban vegetation in mitigating<br />
atmospheric greenhouse gas concentrations and improving air quality in Santiago,<br />
Chile (a city of more than 4 million inhabitants), was illustrated in a recent study
366<br />
P.K.R. NAIR N<br />
(F. Escobedo, personal communication; January 2006). Gaston et al. (2005) reported<br />
that the ‘domestic gardens’ with mean area of only 151 m 2 per garden covered<br />
approximately 33 km 2 or 23% of the predominantly urban area of the city of<br />
Sheffield, U.K., and provided tremendous opportunities for maintenance of<br />
biodiversity and provision of ecosystem services in urban areas. Furthermore, there<br />
is a revival of appreciation of recreational and social values of ornamental and other<br />
types of homestead gardening in the industrialized world such as the United States<br />
(Westmacott, 1992) and Europe (Vogl and Vogl-Lukasser, 2003). An increasing<br />
number of gardeners are now finding pleasure in growing plants for various uses and<br />
deriving satisfaction from agrarian life-style, self-reliance, and private ownership – a<br />
clear expression of the appreciation of the aesthetic, cultural, and landscape values<br />
of such integrated systems, and perhaps the bygone days.<br />
6. FUTURE OF HOMEGARDENS<br />
Prompted by the lack of appreciation of the value of homegardens in development<br />
paradigms and the trends towards commercialization of homegardens and urban<br />
homegardens, the question has been posed “are homegardens becoming extinct?”<br />
(Kumar and Nair, 2004). Wiersum (2006) argues that this illustrates that “the notion<br />
of socioeconomic sustainability of homegardens should be interpreted as referring<br />
not only to their ability to contribute towards the livelihood needs of traditional rural<br />
dwellers, but also to their ability to adjust to the process of rural change.”<br />
Obviously, no one can accurately predict the future of an activity such as<br />
homegardening that is deeply rooted in ecological, socioeconomic, and cultural<br />
milieu of the land and its people. Some of the well-known predictions such as the<br />
200-year-old Malthusian theory are even better known today for their failures to<br />
hold up in a changing world. As the old adage goes, change is the only constant<br />
thing. <strong>Homegardens</strong> are no exception; they will certainly be affected by the changes<br />
happening in the local ecology, economics, and culture. The rate and extent of the<br />
impact of such changes will depend on a myriad of factors. Economic and cultural<br />
forces often pull the society and people’s attitudes in opposite directions. If some<br />
farmers in periurban centers are attracted by the forces of economics to convert their<br />
homegardens or sections of them to growing crops that can fetch money in the<br />
market, there will be an equally strong (if not stronger) section of farmers who are<br />
not attracted by the lure of money to abandon their age-old traditions. When, rather<br />
than if, some genetically modified crops find their way to homegardens, that may<br />
not necessarily mean a proliferation of transgenic homegardens – at least in the near<br />
future. In fact, homegardens are “testing grounds” of many innovations of the<br />
gardeners, and today’s gardens of long standing are a result of such continuous<br />
innovation and improvement. The migration of the youth to urban and even overseas<br />
centers in search of jobs and cash income, a common feature in many homegardendominated<br />
societies, naturally raises concerns about the future of homegardens,<br />
particularly the scope for bringing any technological innovations to the practice of<br />
homegardening. What is seldom recognized, however, is the reverse migration of<br />
older workforce who, after long stays in industrialized urban centers get<br />
disenchanted and seek to return to their roots in increasing numbers and take up
WHITHER W HOMEGARDENS?<br />
367<br />
hobby farming and homegardening for the pure pleasure of doing something they<br />
have grown up with and to which they possess a cultural bondage; this reverse<br />
migration seldom gets the media attention of out-migration of youth.<br />
What conclusion can, then, be drawn on the future of homegardens? Will they<br />
survive or will they become extinct? It is anybody’s guess. I, for one, have<br />
relentlessly argued for quantitative and measurable evidence in support of a<br />
conclusion. But I don’t have much evidence of that nature to draw upon in this case.<br />
So, I would rather make no prediction. Nevertheless, my intuition is that<br />
homegardens will not become extinct. Because of the difficulties in quantitative<br />
valuation of the sustainability attributes of homegardens, it is unlikely that<br />
homegardens will become a part of the development bandwagon; therefore it is<br />
unlikely that there will be any “big push” towards research on homegardens. But<br />
that will not lead to the demise of homegardens. I have only my personal<br />
experiences of interactions with homegardeners around the world to support this<br />
intuitive prediction: the innovative spirit of the Japanese settler farmer in Tomé-Açu<br />
(Brazil), the sentimental attachment to ancestral land and way of life of the<br />
homegardeners in Kerala (India), the tenacity of the farmers who maintain<br />
economically attractive Kandyan homegardens (Sri Lanka), the community’s<br />
commitment to traditional life style of the homegardeners in Nakhon Sawan<br />
(Thailand), the intuitive skills of the industrious and tradition-bound homegardeners<br />
of Java even after they were transmigrated under government pressure to unfamiliar<br />
and distant lands in Kalimantan (both in Indonesia), the friendliness and confidence<br />
of the ecotourism-oriented homegardeners of the Blue Mountain region (Jamaica),<br />
the hope and aspirations built around homegardens of the hapless rural folks in<br />
Koutiala (Mali) and Cap Haitien (Haiti), the satisfaction of the gardeners in being<br />
able to produce a variety of food and other essential needs in their homegardens in<br />
mountainous landlocked terrains in Mount Hagen (Papua New Guinea) and waterlocked<br />
Gizo (Solomon Islands), the pride and self-confidence effused by the female<br />
gardeners in the shambas of the Kikuyuland (Kenya) and the chagga in Arusha<br />
region (Tanzania), the ingenuity of the farmers who have successfully introduced<br />
rearing in captivity through stall-feeding of the African grasscutter (Thryonomys<br />
swinderianus, a herbivorous rodent that is harvested for delicious and pricy bush<br />
meat) in Kumasi (Ghana), … – the list can be long – all point to continuation of the<br />
homegardens, of sorts, in perpetuity. So, my submission is, homegardens will<br />
undergo changes; but they will not become extinct; they will continue to exist with<br />
their mysterious, enigmatic charm to provide sustenance, satisfaction, income, and<br />
aesthetic appeal to many, and fascination to scientists who care to look at them.<br />
ACKNOWLEDGMENTS<br />
I thank Drs. Michael Bannister, Vimala Nair, and Freerk Wiersum for comments<br />
and suggestions on the manuscript.
368<br />
P.K.R. NAIR N<br />
REFERENCES<br />
Abdoellah O.S., Hadikusumah H.Y., Takeuchi K., Okubo S. and Parikesit. 2006.<br />
Commercialization of homegardens in an Indonesian village: vegetation composition and<br />
functional changes. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A<br />
time-tested example of sustainable agroforestry, pp 233 – 250. Springer Science,<br />
Dordrecht.<br />
Alavalapati J.R.R. and Mercer D.E. (eds). 2004. Valuing agroforestry systems: Methods and<br />
applications (Advances in Agroforestry # 2). Kluwer, Dordrecht, 314p.<br />
Bene J.G., Beall H.W. and Côte A. 1977. Trees, food and people. International Development<br />
Research Centre, Ottawa, Canada. 52p.<br />
Drescher A.W., Holmer R.J. and Iaquinta D.L. 2006. Urban homegardens and allotment<br />
gardens for sustainable livelihoods: Management strategies and institutional<br />
environments. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A timetested<br />
example of sustainable agroforestry, pp 317 – 338. Springer Science, Dordrecht.<br />
Falanruw M.V.C. 1990. The food production system of the Yap Island. In: Landauer K. and<br />
Brazil B. (eds), <strong>Tropical</strong> home gardens, pp 94–104. United Nations University Press,<br />
Tokyo.<br />
Fernandes E.C.M., O’Kting’ati A. and Maghembe J. 1984. Chagga homegardens: a<br />
multistory agroforestry cropping system on Mt. Kilimanjaro, northern Tanzania.<br />
Agroforest Syst 2: 73 – 86.<br />
Gajaseni J. and Gajaseni N. 1999. Ecological rationalities of the traditional homegarden<br />
system in the Chao Phraya Basin, Thailand. Agroforest Syst 46: 3 – 23.<br />
Gaston K.J., Warren P.H., Thompson K and Smith R.M. 2005. Urban domestic gardens (IV):<br />
the extent of the resource and its associated features. Biodivers Conserv 14: 3327 – 3349.<br />
Howard P.L. 2006. Gender and social dynamics in swidden and homegardens in Latin<br />
America. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 159 – 182. Springer Science, Dordrecht.<br />
Kehlenbeck K. and Maass B.L. 2006. Are tropical homegardens sustainable? Some evidence<br />
from Central Sulawesi, Indonesia. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 339 – 354. Springer<br />
Science, Dordrecht.<br />
Kumar B.M. 2006. Carbon sequestration potential of tropical homegardens. In: Kumar B.M.<br />
and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 185 – 204. Springer Science, Dordrecht.<br />
Kumar B.M. and Nair P.K.R. 2004. The enigma of tropical homegardens. Agroforest Syst 61:<br />
135 – 152.<br />
Lal R. 2004. Soil carbon sequestration impacts on global change and food security. Science<br />
304: 1623-1627.<br />
Lamanda N., Malézieux E. and Martin P. 2006. Structure and dynamics of coconut-based<br />
agroforestry systems in Melanesia: a case study from Vanuatu archipelago. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 105 – 121. Springer Science, Dordrecht.<br />
Leakey R.R.B., Tchoundjeu Z., Smith R.I., Munro R.C., Fondoun J-M., Kengue J., Anegbeh<br />
P.O., Atangana A.R., Waruhiu A.N., Asaah E., Usoro C. and Ukafor V. 2004. Evidence<br />
that subsistence farmers have domesticated indigenous fruits (Dacryodes edulis and<br />
Irvingia gabonensis) in Cameroon and Nigeria. Agroforest Syst 60: 101 – 111.<br />
McCann K.S. 2000. The diversity – stability debate. Nature 405: 228 – 233.<br />
McNeely J.A. 2004. Nature vs. nurture: Managing relationships between forests, agroforestry<br />
and wild biodiversity. Agroforest Syst 61: 155 – 165.
WHITHER W HOMEGARDENS?<br />
369<br />
Mohan S., Alavalapati J.R.R. and Nair P.K.R. 2006. Financial analysis of homegardens: A<br />
case study from Kerala State, India. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 283 – 296. Springer<br />
Science, Dordrecht.<br />
Mohan S., Nair P.K.R. and Long A.J. 2006. An assessment of homegarden diversity: A case<br />
study from Kerala State, India. J Sust Agric (in press).<br />
Montagnini F. and Nair P.K.R. 2004. Carbon sequestration: An under-exploited<br />
environmental benefit of agroforestry systems. Agroforest Syst 61: 281 – 298.<br />
Nair P.K.R. 1979. Intensive multiple cropping with coconuts in India: Principles,<br />
programmes and prospects. Verlag Paul Parey, Berlin, 149p.<br />
Nair P.K.R. 1993. An Introduction to Agroforestry. Kluwer, Dordrecht, 499p.<br />
Nair P.K.R. 2001. Do tropical homegardens elude science, or is it the other way around?<br />
Agroforest Syst 53: 239 – 245.<br />
Nair P.K.R. 2006. Agroforestry for sustainability of lower-input land use systems. J Sust.<br />
Crop Production (in press).<br />
Nair P.K.R. and Kumar B.M. 2006. Introduction. In: Kumar B.M. and Nair P.K.R. (eds),<br />
<strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp 1 – 10.<br />
Springer Science, Dordrecht.<br />
Nair P.K.R., Buresh R.J., Mugendi D.N. and Latt C.R. 1999. Nutrient cycling in tropical<br />
agroforestry systems: Myths and science. In: Buck L.E., Lassoie J.P., and Fernandes<br />
E.C.M. (eds), Agroforestry in sustainable agricultural system, pp 1 – 31. CRC Press, Boca<br />
Raton, FL.<br />
Niñez V.K. 1984. Household gardens: theoretical considerations on an old survival strategy.<br />
Potatoes in Food Systems Research Series Report No. 1, International Potato Center,<br />
Lima, Peru, 41p.<br />
Odum E.P. 1969. The strategy of ecosystem development. Science 164: 262 – 270.<br />
Okafor J.C. and Fernandes E.C.M. 1987. The compound farms of Nigeria: A predominant<br />
agroforestry homegarden system with crops and small livestock. Agroforest Syst 5: 153 –<br />
168.<br />
Peyre A., Guidal A., Wiersum K.F. and Bongers F. 2006. Homegarden dynamics in Kerala,<br />
India. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 87 – 103. Springer Science, Dordrecht.<br />
Rao M.R. and Rajeswara Rao B.R. 2006. Medicinal plants in tropical homegardens. In:<br />
Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of<br />
sustainable agroforestry, pp 205 – 232. Springer Science, Dordrecht.<br />
Rao M.R., Nair P.K.R. and Ong C.K. 1988. Biophysical interactions in tropical agroforestry<br />
systems. Agroforest Syst 38: 3 – 50.<br />
Schroth G., da Fonseca A.B., Harvey C.A., Gascon C., Vasconcelos H.L. and Izac N. (eds).<br />
2004. Agroforestry and biodiversity conservation in tropical landscapes. Island Press,<br />
Washington, DC. 523p.<br />
Simons A.J. and Leakey R.R.B. 2004. Tree domestication in tropical agroforestry. Agroforest<br />
Syst 61: 167 – 181.<br />
Soini E. 2005. Changing livelihoods on the slopes of Mt. Kilimanjaro, Tanzania: Challenges<br />
and opportunities in the Chagga homegarden system. Agroforest Syst 64: 157 – 167.<br />
Swift M.J. and Anderson J.M. 1993. Biodiversity and ecosystem function in agricultural<br />
systems. In: Schulze E.D. and Mooney H.A. (eds), Biodiversity and ecosystem function,<br />
pp 15 – 42. Springer, Berlin.<br />
Tesfaye Abebe, Wiersum K.F., Bongers F. and Sterck F. 2006. Diversity and dynamics in<br />
homegardens of southern Ethiopia. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong><br />
homegardens: A time-tested example of sustainable agroforestry, pp 123 – 142. Springer<br />
Science, Dordrecht.
370<br />
P.K.R. NAIR N<br />
Thaman R.R., Elevitch C.R. and Kennedy J. 2006. Urban and homegarden agroforestry in the<br />
Pacific islands: current status and future prospects. In: Kumar B.M. and Nair P.K.R. (eds),<br />
<strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp 25 – 41.<br />
Springer Science, Dordrecht.<br />
Torquebiau E. 1992. Are tropical agroforestry homegardens sustainable? Agric Ecosyst<br />
Environ 41: 189 – 207.<br />
Torquebiau E. and Penot E. 2006. Ecology versus economics in tropical multistrata<br />
agroforests. In: Kumar B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested<br />
example of sustainable agroforestry, pp 269 – 282. Springer Science, Dordrecht.<br />
Vogl C.R. and Vogl-Lukasser B. 2003. Tradition, dynamics and sustainability of plant species<br />
composition and management in homegardens on organic and non-organic small scale<br />
farms in Alpine Eastern Tyrol, Austria. Biol Agric Hortic 21: 349 – 366.<br />
Watson J.W. and Eyzaguirre P.B. (eds). 2002. Home gardens and in situ conservation of plant<br />
genetic resources in farming systems. Proceedings of the Second International Home<br />
Gardens Workshop (17–19 July 2001), Witzenhausen, Germany. International Plant<br />
Genetic Resources Institute (IPGRI), Rome, Italy, 184p.<br />
World Commission on Environment and Development (WCED) 1987. Our common future.<br />
Oxford University Press, Oxford, 370p.<br />
Wills C, Harms K.E., Condit R., King D., Thompson J., He J., Muller-Landau H.C., Ashton<br />
P., Losos E., Comita L., Hubbell S., LaFrankie J., Bunyavejchewin S., Dattaraja H.S.,<br />
Davies S., Esufali S., Foster R., Gunatilleke N., Gunatilleke S., Hall P., Itoh A., John R.,<br />
Kiratiprayoon S., de Lao S.L., Massa M., Nath C., Noor M.N.S., Kassim A.R., Sukumar<br />
R., Suresh H.S., Sun I-F., Tan S., Yamakura T. and Zimmerman, J. 2006. Nonrandom<br />
processes maintain diversity in tropical forests. Science 311: 527 – 531.<br />
Westmacott R.N. 1992. African-American gardens and yards in the rural south. University of<br />
Tennessee Press, Knoxville, TN, 198p.<br />
Wezel A. and Ohl J. 2006. Homegarden plant diversity in relation to remoteness from urban<br />
centers: a case study from the Peruvian Amazon region. In: Kumar B.M. and Nair P.K.R.<br />
(eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable agroforestry, pp<br />
143 – 158. Springer Science, Dordrecht.<br />
Wiersum K.F. 2004. Forest gardens as an ‘intermediate’ land use system in the nature –<br />
culture continuum: characteristics and future potential. Agroforest Syst 61: 123 – 134.<br />
Wiersum K.F. 2006. Diversity and change in homegarden cultivation in Indonesia. In: Kumar<br />
B.M. and Nair P.K.R. (eds), <strong>Tropical</strong> homegardens: A time-tested example of sustainable<br />
agroforestry, pp 13 – 24. Springer Science, Dordrecht.
Adaptability 66, 67, 87, 123, 179, 275<br />
Adaptive management 137, 185, 283,<br />
293<br />
Adaptive research 299<br />
Aesthetic garden18<br />
African-American yards and gardens 6,<br />
7<br />
Agricultural landscape 65, 203, 235,<br />
271<br />
Agrobiodiversity (see also biodiversity)<br />
46, 55, 103, 117, 170, 173, 174,<br />
179, 200, 269, 280<br />
Agrodeforestation 25, 26, 34, 36, 39,<br />
41, 118<br />
Allotment gardens 9, 229, 317–319,<br />
321–323, 325–327, 329–337,<br />
368<br />
Amazon 10, 23, 43–60, 62, 64, 73, 83,<br />
84, 134, 139, 143, 144, 149,<br />
154–158, 160, 161, 163,<br />
165–167, 169, 170, 173, 175,<br />
179, 180–182, 189, 201, 204,<br />
206, 212, 213, 224, 225, 230,<br />
231, 267, 299–301, 304,<br />
309–312, 314–316, 338, 358,<br />
370<br />
Amazonia 23, 43–60, 62, 83, 144,<br />
156–158, 160, 163, 165–167,<br />
169, 175, 179–181, 189, 201,<br />
204, 230, 231, 267, 299, 301,<br />
314–316<br />
Amerindians 75, 84, 160, 165, 175<br />
Andes 145, 167, 170, 171, 173–175,<br />
181<br />
Animal husbandry 34, 247, 249<br />
Aromatherapy 221, 223, 228<br />
Aromatic plants 205, 208, 213, 226,<br />
226–231<br />
Benefit-cost analysis 283<br />
Biodiversity (see also<br />
Agrobiodiversity, Crop diversity<br />
and Species diversity) 8, 27, 39,<br />
41, 61–64, 69, 73, 74, 77, 80, 82,<br />
SUBJECT INDEX<br />
83, 87, 88, 101, 134, 137, 139,<br />
158, 181, 193, 199, 201, 203,<br />
231, 270–274, 279, 280, 284,<br />
320, 330, 339, 340, 356,<br />
361–363, 366, 368, 369<br />
Biodiversity conservation (see also<br />
Convention on Biological<br />
Diversity) 8, 77, 137, 199,<br />
270–274, 279, 280, 356, 369<br />
Biofactory 228<br />
Biopesticides 214<br />
Biopiracy 227<br />
Budget garden 20<br />
Caboclo 43, 44, 46–48, 50–52, 54, 55,<br />
58, 167, 182<br />
Canopy area 258<br />
Canopy conductance 258, 263, 264<br />
Canopy layers 251, 252, 260, 261<br />
Carbon sequestration 9, 61, 80–83,<br />
117, 119, 185–187, 189, 191,<br />
193, 195, 197, 198, 200–203,<br />
272, 274, 278, 281, 282, 356,<br />
363, 368, 369<br />
Carbon stocks 185, 188, 189, 191, 201,<br />
203<br />
Cash crops 9, 22, 38, 39, 90, 97, 100,<br />
102, 123, 124, 127, 128, 131,<br />
132, 134–138, 159, 162, 170–173,<br />
214, 233, 235, 236, 244–247,<br />
341, 352, 364<br />
Catalonia 3, 4, 6, 8, 211, 212, 224,<br />
228<br />
Cattle grazing 106, 111, 112, 117<br />
Chagga 2, 3, 9, 134, 135, 138, 139,<br />
140, 207, 213, 229, 358, 367–369<br />
Children 54, 57, 117, 162, 178, 181,<br />
227, 244, 248, 301, 311, 319,<br />
320, 325, 351, 362<br />
Classification, homegarden 23, 89, 90,<br />
92, 99, 108, 109, 112, 113, 127,<br />
229, 253, 320, 333, 353, 362<br />
Clean Development Mechanisms<br />
198–200, 272, 279
372 SUBJECT INDEX<br />
Cluster analysis 69, 87, 89, 90, 92, 360<br />
Commercial crops 18, 19, 47, 48, 50,<br />
55, 98, 123, 124, 133, 233, 235,<br />
240, 284, 293, 314<br />
Commercialization 7, 8, 15, 21, 22, 37,<br />
40, 60, 88, 101, 123, 124, 132,<br />
133, 135, 137, 138, 159, 165,<br />
173, 179, 214, 233–235, 237,<br />
239, 241, 243–249, 355, 356,<br />
358, 361, 364, 366, 368<br />
Commoditization 179<br />
Commodity production 94, 171, 172,<br />
186<br />
Community gardens 318, 319, 322,<br />
322–324, 326, 330, 331, 338<br />
Composition, homegarden 8, 9, 13, 15,<br />
17–22, 35, 40, 51, 53, 58, 61, 66,<br />
68, 69, 74, 81, 87, 88, 90,<br />
100–102, 123–132, 135–139,<br />
147, 148, 158–160, 165, 168–170,<br />
173, 176, 180, 186, 194, 197–199,<br />
201, 213, 224, 230, 231, 233,<br />
235, 240, 245, 248–250, 253,<br />
293, 311, 339, 345, 360, 368,<br />
370<br />
Contract farming 226, 228<br />
Convention on Biological Diversity<br />
187<br />
Correspondence analysis 69, 129, 130,<br />
360<br />
Cosmology 54, 161, 166, 167, 170,<br />
181<br />
Crop diversity (see also Species<br />
diversity and Biodiversity) 18,<br />
19, 23, 45, 124, 132, 136, 162,<br />
176, 229, 340, 343, 347–349,<br />
351–353<br />
Cropping system dynamics 105<br />
Diversity index (see also Shannon<br />
index, Shannon-Weaver index,<br />
and Margalef index) 94, 236–238,<br />
245, 246, 345<br />
Division of labor 159–162, 165–168,<br />
171, 172, 177<br />
Domestic animals (see also Livestock)<br />
1, 2, 20, 32, 48, 61, 76, 77, 81,<br />
206, 301<br />
Domestication 43–46, 54, 56, 57, 59,<br />
60, 73, 80, 82, 363, 369<br />
Dynamics, homegarden 9, 13, 15, 20–23,<br />
40, 82, 87–89, 91, 93, 95, 97, 99,<br />
101–103, 105–110, 112, 115,<br />
118, 123, 124, 126, 132, 135,<br />
137, 139, 158–161, 163, 165,<br />
167, 169, 171, 173, 175, 177,<br />
179–181, 197, 201, 229, 235,<br />
265, 275, 280, 300, 315, 336,<br />
339, 340, 348, 362, 368, 369, 370<br />
Ecological instability 247<br />
Ecological succession 360<br />
Ecological sustainability 78, 87, 88,<br />
101, 102, 135–137, 271, 274,<br />
280, 360, 361<br />
Economic alternatives 284, 285, 291<br />
Economic resilience 274, 284, 285,<br />
290<br />
Economic utility 283, 285, 287, 295,<br />
296<br />
Economic valuation (see also Financial<br />
worth) 356, 361<br />
Ecosystem services 194, 198, 272, 203,<br />
366<br />
Ecotourism 225, 228, 367<br />
Environmental economics 278, 279<br />
Environmental services (see also<br />
Ecosystem services) 62, 81, 198,<br />
269, 273, 278, 279, 281<br />
Ethnobiodiversity 39<br />
Ethnobotany 84, 180–182<br />
Ethnohistory 23, 43, 44, 46, 83<br />
Extension 10, 17, 21, 40, 43, 44, 56,<br />
57, 106, 108, 110, 111, 118, 119,<br />
173, 185, 273, 294, 299, 313,<br />
314, 317, 318, 326, 330, 331,<br />
333, 335, 336, 350, 364<br />
External inputs 87, 88, 91, 95, 98, 100,<br />
101, 135–137, 237, 243, 244,<br />
247, 248
Externalities 198, 269, 270, 272, 273,<br />
276, 278–280<br />
Extractivism 278<br />
Fallow 17, 26, 33, 46, 54, 55, 58, 65,<br />
84, 105, 106, 109, 111, 112, 115,<br />
117, 144, 145, 156, 162, 165,<br />
188, 201, 207, 225, 300, 341<br />
Farm size 123, 132, 133, 137, 343,<br />
347, 348, 353<br />
Farmer innovation 299<br />
Farmer-explorers 311<br />
Farmers’ practices 106, 112<br />
Farming systems 2, 9, 13, 15, 17, 19,<br />
21, 23, 103, 106, 124, 139, 158,<br />
230, 269, 273, 280, 311, 329,<br />
354, 370<br />
Financial worth (see also Economic<br />
valuation) 285, 287, 288, 290<br />
Floodplains 51, 143, 144, 156, 158,<br />
225<br />
Floristic composition 69, 81, 180, 245,<br />
249, 250, 360<br />
Food security 3, 25, 26, 39–41, 55, 61,<br />
62, 74, 75, 77, 81, 117, 119, 139,<br />
173, 202, 203, 266, 294, 317,<br />
326, 329, 336, 337, 357, 359, 368<br />
Forest fallow 188, 225, 300<br />
Forest garden 2, 17, 23, 24, 62, 139,<br />
140, 182, 226, 231, 252, 270,<br />
281, 282, 351, 353, 370<br />
Forest rent 273, 279, 280<br />
Fragmentation of holdings 133, 186,<br />
275<br />
Fruit trees 2, 3, 6, 7, 17, 26, 31, 35,<br />
43–47, 49, 51, 53–56, 61, 62, 65,<br />
66, 68, 72, 74, 75, 80, 94, 95, 98,<br />
110–113, 115, 143, 144, 148,<br />
149, 155, 172, 205, 224, 273,<br />
299, 300, 301, 312, 314, 320,<br />
342, 358, 363, 365<br />
Functional characteristics 91, 94, 99<br />
Functional differentiation 89<br />
Functions 6, 16, 18, 21, 23, 52, 69, 91,<br />
98, 117, 126, 129, 135, 136,<br />
158–160, 165, 168, 174, 176,<br />
SUBJECT INDEX 373<br />
234, 235, 240, 247, 248, 270–273,<br />
293, 319, 320, 342, 355<br />
Gender relations 159, 160, 181<br />
Genesis, homegardens 2<br />
Global distribution, homegardens 2–5<br />
Globalization 26, 227, 275,<br />
Handicraft plants 27, 30<br />
Health care 144, 177, 333<br />
Herbal medicine 206, 226, 227<br />
Hillside gardening 38<br />
Historic development, homegardening<br />
15, 17<br />
Historical accounts, homegardening<br />
45<br />
Homegarden management 53, 101,<br />
178, 351<br />
Homegarden model 318, 327–329<br />
Homegarden, types 2, 15–18, 20, 89,<br />
91, 92–100, 129–132, 237, 239,<br />
240, 245, 246, 330, 336<br />
<strong>Homegardens</strong>, extent 3, 4, 38, 186,<br />
207, 301, 325<br />
Importance value index 360<br />
In situ conservation 69, 103, 205, 339,<br />
340, 348, 352–354, 363, 370<br />
Incident radiation 61, 68, 80, 81, 251,<br />
252, 257, 260, 261, 263–265<br />
Indigenous knowledge 8, 41, 43, 48,<br />
62, 177, 180, 205, 225–227<br />
Indigenous people 46, 80, 143<br />
Institutional support 299, 313, 315<br />
Intangible benefits (see also nonmarket<br />
benefits) 8, 283, 284,<br />
286, 287, 293, 356, 361<br />
Integrated farming system 9, 16, 123,<br />
124, 129<br />
Intellectual property rights 227<br />
Intensification 2, 19, 20, 25, 101, 106,<br />
118, 195, 199, 201<br />
Intercropping 108, 118, 203, 229,<br />
300<br />
Internalization of externalities 269,<br />
280
374 SUBJECT INDEX<br />
Japanese-Brazilian 10, 60, 84, 300,<br />
302, 338<br />
Javanese 2, 15–18, 21, 35, 30, 41, 140,<br />
158, 187, 188, 201, 250, 271,<br />
275, 281, 302, 320, 353, 354<br />
Kandyan gardens 22, 266<br />
Keystone species 123, 136–139, 195,<br />
361<br />
Kitchen garden 18, 38, 40, 176, 319<br />
Knowledge transmission 177, 178<br />
Knowledge erosion 178<br />
Kyoto protocol 196, 198, 199, 272<br />
Land tenure 51, 53, 169, 170, 181, 272,<br />
274, 285, 325, 326<br />
Leaf area index 258<br />
Livelihood security 138, 198, 199, 327,<br />
337<br />
Livelihood strategies 15, 16, 24, 318<br />
Livestock (see also Domestic animals)<br />
4, 17, 26, 38, 53, 55, 62, 65, 76,<br />
125, 128–133, 135–137, 139,<br />
144, 161, 165, 200, 205, 207,<br />
213, 225, 230, 236, 237, 243, 244,<br />
270, 283, 286, 319, 324, 369<br />
Living edible pens 34<br />
Malnutrition 15, 41, 61, 66, 74, 77,<br />
328, 337<br />
Management intensity 17, 91, 95, 100<br />
Management practices 26, 56, 78–81,<br />
87, 90, 91, 95, 96, 100, 101, 137,<br />
180, 193, 199, 226, 249, 337,<br />
340, 349<br />
Management strategies 9, 61, 229, 282,<br />
283, 294, 317, 326, 330, 336, 368<br />
Margalef index 360<br />
Market access 19, 339, 341, 347<br />
Market garden 18, 20<br />
Market pressure 154, 233, 234, 364<br />
Market value 130, 275, 278, 284, 285<br />
Marketing 16, 19, 40, 56, 108, 118,<br />
136, 172, 173, 226, 274, 275,<br />
301, 314, 331, 335<br />
Market-oriented systems 364<br />
Matsiguenka 143, 145, 147–158<br />
Maya 61, 62, 64, 65, 68, 70, 72–74, 76,<br />
78, 80–84, 138, 157, 160, 162,<br />
163, 165, 169, 170, 172, 173,<br />
175, 176, 178–182, 199, 200,<br />
207, 226, 229–231, 250, 265,<br />
266, 353<br />
Medicinal plants 8, 16, 27, 28, 35, 36,<br />
46, 49, 68, 69, 91, 98, 142, 148,<br />
153, 155, 171, 175, 177, 205–207,<br />
211, 213–215, 224–231, 271,<br />
285–287, 301, 311, 320, 348,<br />
363, 364, 369<br />
Melanesian agriculture 105, 118<br />
Mesoamerica 2, 6, 44, 61–78, 80–83,<br />
135, 139, 160, 162, 163, 165,<br />
169, 170, 172, 182, 186, 202<br />
Mestizo 48, 158, 160, 170, 179<br />
Micro-economic approach 278<br />
Microgranjas 319<br />
Mixed gardens 17, 18, 235<br />
Modeling 252, 266, 269, 270, 276–278,<br />
280<br />
Modernization 22, 66, 87, 101<br />
Monocultures 187, 235, 245, 273, 300,<br />
364<br />
Multispecies systems 87<br />
Multistrata systems 1, 2, 68, 82, 88,<br />
105, 111, 116, 117, 138, 157,<br />
185, 187, 192–194, 196, 199,<br />
206, 225, 226, 229, 240, 245,<br />
250, 269–271, 273, 275, 277,<br />
279, 281, 294, 299, 313, 314,<br />
356, 361, 370<br />
Net primary productivity 185<br />
Nitrogen fixation 78, 82, 185, 196,<br />
294, 350<br />
Non-food plants 27, 29, 30<br />
Non-market benefits (see also<br />
Intangible benefits) 284<br />
Non-monetary benefits (see also<br />
Intangible benefits) 283, 291<br />
Nutrient cycling 55, 62, 67, 78, 80, 81,<br />
124, 135, 202, 269, 272, 274,<br />
280, 281, 340, 350, 361, 369
Nutrient outputs 194<br />
Nutritional security 7, 14, 117, 274,<br />
284, 287, 293, 294, 359, 361,<br />
362<br />
Nutritional value 16, 61, 117, 247,<br />
271<br />
Nutrition-related non-communicable<br />
diseases 26, 39<br />
Olympe 276, 280–282<br />
Opportunity costs 285, 288<br />
Organoponics 319<br />
Ornamentals 2, 18–20, 31, 36, 49, 71,<br />
90, 98, 99, 101, 124, 133, 134,<br />
162, 171, 211, 224, 342, 350,<br />
365<br />
Patrimonial analysis 277<br />
Patrimonial strategies 271, 277, 280<br />
Pekarangan 2, 3, 16, 17, 22, 186, 249,<br />
271<br />
Penman-Monteith equation 258, 263<br />
Periurban typology 337<br />
Physical environment 133, 234<br />
Phytochemicals 205, 228<br />
Phytoremediation 198, 274<br />
Pielou evenness index 342, 344, 345,<br />
352<br />
Plant diversity (see also Crop<br />
diversity, Biodiversity, and<br />
Diversity index) 17, 30, 62, 70,<br />
72, 143, 144, 158, 175, 181,<br />
193, 197, 202, 203, 232, 233,<br />
236, 237, 248, 339, 340, 351,<br />
370<br />
Plant genetic resources 55, 103, 340,<br />
348, 352–354, 370<br />
Policies 101, 199, 226, 249, 272, 275,<br />
335<br />
Population density 14, 27, 65, 66, 68,<br />
108, 127, 132, 133, 247, 275,<br />
341<br />
Poverty 25, 36, 39, 61, 65, 66, 74, 75,<br />
77, 80, 176, 198, 203, 312, 314,<br />
318, 328<br />
Profit 227, 283, 285, 287–293, 361<br />
SUBJECT INDEX 375<br />
Radiation interception 251<br />
Rainforest 59, 64, 83, 143–145, 149,<br />
156–158, 201, 315, 340, 341,<br />
354<br />
Regression 132, 133, 193, 258, 262,<br />
287, 290<br />
Residue management 61, 78, 81<br />
Resource utilization efficiency 196<br />
Ribereño 43, 44, 46, 47, 50–52, 54, 55,<br />
58<br />
Rights 32, 51, 168–171, 181, 227, 272,<br />
274, 326<br />
Rights to plants 170, 181<br />
Risk buffering 269, 271, 274, 276, 278,<br />
279<br />
Roadside gardening 69, 76, 326<br />
Rooftop gardening 319<br />
Rural development 22, 56, 61, 77, 90,<br />
102, 234, 299, 300, 314, 316,<br />
338<br />
Rural transformations 13, 15, 20, 21<br />
Sap flow gauges 251, 255, 265<br />
Sap flow rate 251, 256–259, 262<br />
Sapwood cross-sectional area 260<br />
Sensitivity analysis 283, 289, 290<br />
Shannon index 91, 93, 94, 342, 344,<br />
345, 347, 348<br />
Shannon-Weaver index /Shannon-<br />
Wiener index 105, 109, 110,<br />
111, 114, 116, 236–239, 360,<br />
Shifting cultivation 1, 2, 14, 16, 17, 20,<br />
21, 106, 143, 144, 158, 162, 225,<br />
228, 349, 360<br />
Slash and burn 75, 143–145, 188, 225<br />
Social and economic functions 235,<br />
320<br />
Social capital 249<br />
Social dynamics 82, 88, 124, 159, 229,<br />
368<br />
Social networks 175, 178, 179, 248<br />
Social status 159, 161, 174, 176, 179,<br />
272, 278<br />
Social sustainability 13, 22, 88, 101,<br />
102, 274, 355, 361, 362<br />
Social-ecological perspective 277, 281
376 SUBJECT INDEX<br />
Socioeconomic change 13, 14, 87–89,<br />
101, 123<br />
Socioeconomic sustainability 14, 15,<br />
87, 89, 135, 136, 366<br />
Soil biota 194, 195, 198<br />
Soil carbon sequestration 82, 197, 202,<br />
368<br />
Soil erosion 78, 203, 274, 340, 349,<br />
350, 352<br />
Soil fertility 19, 37, 56, 117, 118, 135,<br />
207, 270, 312, 339–343, 345–347,<br />
349–352, 356<br />
Soil organic matter 81, 185, 195, 197,<br />
274<br />
Sørensen’s Index 147, 148, 342, 345<br />
Spatial densification 318<br />
Species abundance 130, 174, 195, 197,<br />
330, 342, 360<br />
Species composition (see also Species<br />
diversity) 9, 17, 53, 58, 61, 66,<br />
68, 87, 100, 123, 128–130, 135,<br />
147, 148, 158, 186, 199, 213,<br />
224, 230, 240, 248, 311, 339,<br />
349, 370<br />
Species density 87, 90, 92, 342, 344,<br />
347, 351<br />
Species diversity (see also Species<br />
composition) 7, 57, 61, 69, 72,<br />
73, 84, 98, 101, 109, 111–113,<br />
116, 123, 128, 130, 131, 133–137,<br />
143, 154, 156–158, 160, 162,<br />
176, 185, 201, 203, 213, 225,<br />
231, 245, 250, 281, 282, 301,<br />
317, 330, 339, 340, 351, 353–355,<br />
357–362, 365<br />
Species introduction 49–51, 53, 56, 91,<br />
98–100, 214, 230, 233, 235, 300,<br />
302, 304, 306, 308, 310–313, 363<br />
Species richness 91, 109, 130, 132,<br />
133, 134, 137, 143, 154, 156,<br />
157, 186, 293, 343, 344, 347,<br />
348, 351, 362<br />
Staple crops 32, 38, 80, 165, 352<br />
Streuobst 6, 9<br />
Structural complexity 8, 61, 67<br />
Structure of homegardens (see also<br />
Vegetation structure) 13, 18, 22,<br />
66, 69, 89, 116, 173, 234, 237<br />
Structure, horizontal 61, 66–68, 69, 80,<br />
109, 111–113, 234,<br />
Structure, vertical 61, 66–68, 80, 89,<br />
105, 109, 111, 113, 116, 171,<br />
196,, 234, 241, 245, 252, 253,<br />
259, 274<br />
Subsistence crops 17, 57, 171, 271<br />
Subsistence garden 6, 18, 20<br />
Sundanese 16–18, 247<br />
Survival garden 20<br />
Sustainability indicators 339, 340, 352<br />
Sustainable production 72, 234, 311,<br />
315, 339<br />
Swidden agriculture 51, 143<br />
Swidden fallow/gardens 46, 58, 65, 84,<br />
144, 145, 160, 165–167, 174,<br />
176<br />
Talun-kebun 2, 16, 17, 22, 249<br />
Temporal dynamics 108, 110, 112,<br />
115, 348<br />
Thermal dissipation probes 251, 255,<br />
256<br />
Tokun 299, 311–315<br />
Totonac 70, 76, 79, 82, 171, 180, 181<br />
Traditional agroforestry 9, 13, 22, 23,<br />
25, 74, 83, 102, 103, 125, 139,<br />
231, 249, 250, 354<br />
Traditional homegardens 22, 52, 78–80,<br />
82, 87, 88, 100, 101, 103, 173,<br />
174, 180, 230, 234, 247–249,<br />
294, 355, 356, 361, 362, 365<br />
Traditional medicine 205, 206, 225,<br />
227<br />
Traditional vegetables 73, 247, 326<br />
Transmigrants 18, 353<br />
Transmigration 20, 342<br />
Transpiration 251–267<br />
Tree management 45, 69, 72, 73, 80,<br />
87, 89–91, 95–102, 137, 161,<br />
170, 176, 193, 195, 226, 228,<br />
274, 330, 331
Tree root systems 190, 193, 196, 197,<br />
201–203, 251, 252, 262, 264,<br />
366<br />
Tree tenure 170, 181, 280<br />
Urban homegardens 7, 8, 19, 229, 317,<br />
319–321, 323–325, 327, 329,<br />
331–338, 355, 364–366, 368<br />
Urban poor 317, 318, 328, 329, 331,<br />
337<br />
Urbanization 18, 25, 26, 179, 186, 235,<br />
318, 325, 333, 337<br />
Value addition 205, 225, 227, 228<br />
Vegetation dynamics 339<br />
SUBJECT INDEX 377<br />
Vegetation structure (see also<br />
Structure of homegardens) 17,<br />
90, 91, 98, 99, 105, 106, 108–110,<br />
112, 116, 124, 127, 137, 158,<br />
201, 240, 241, 248, 252, 340,<br />
353<br />
Whole-farm analysis 276<br />
Women 27, 74, 77, 159–182, 214, 216,<br />
227, 230, 274, 294, 319, 320,<br />
325, 338, 357, 359, 361, 362<br />
World Trade Organization 227<br />
Zonation 83, 103, 139, 158, 182, 230,<br />
250, 281, 294
1.<br />
Advances In Agroforestry<br />
P.K.R. Nair, M.R. Rao and L.E. Buck (eds.): New Vistas in Agroforestry: A Comp-<br />
st<br />
endium for the 1 World Congress of Agroforestry, 2004. ISBN 1-4020-2501-7<br />
2. Janaki R.R. Alavalapati and D. Evan Mercer (eds.): Valuing Agroforestry Systems:<br />
Methods and Applications, 2004.<br />
ISBN 1-4020-2412-6<br />
3.<br />
B.M. Kumar and P.K.R. Nair (eds.): <strong>Tropical</strong> <strong>Homegardens</strong>: A Time-Tested Example<br />
of Sustainable Agroforestry, 2006.<br />
ISBN 1-4020-4947-1<br />
springer.com